Advanced Organolithium Synthesis of 4-Bromo-2,6-Difluorobenzoic Acid for Commercial Scale-Up
Advanced Organolithium Synthesis of 4-Bromo-2,6-Difluorobenzoic Acid for Commercial Scale-Up
The global demand for fluorinated aromatic intermediates continues to surge, driven by their critical role in the development of next-generation pharmaceuticals and agrochemicals. Patent CN100522920C introduces a transformative methodology for the preparation of 4-bromo-2,6-difluorobenzoic acid, a pivotal building block in the synthesis of liquid crystals, antitumor agents, and functional dyes. This technical disclosure addresses long-standing challenges in regioselective functionalization of polyfluorinated aromatics, offering a pathway that utilizes readily available industrial raw materials like 3,5-difluorobromobenzene. By shifting away from traditional oxidative or non-selective Grignard protocols, this innovation provides a robust framework for manufacturers seeking to optimize yield and minimize environmental impact. For R&D directors and procurement strategists, understanding the nuances of this organolithium-mediated carboxylation is essential for securing a competitive supply chain for high-value fluorinated intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a carboxyl group onto an aromatic nucleus has relied heavily on oxidation of alkyl side chains or Grignard reagent synthesis, both of which present significant drawbacks for complex fluorinated substrates. The oxidation style is strictly confined to substrates possessing an alkyl side chain and often necessitates strong oxidizers like potassium bichromate, which pose severe environmental pollution risks and are difficult to recover once destroyed. Furthermore, when attempting to synthesize 4-bromo-2,6-difluorobenzoic acid using the conventional Grignard reagent method, manufacturers typically start with 3,5-difluoro-1,4-dibromobenzene. As illustrated in the reaction pathway below, this approach is fundamentally flawed due to the presence of two bromine atoms on the phenyl ring.
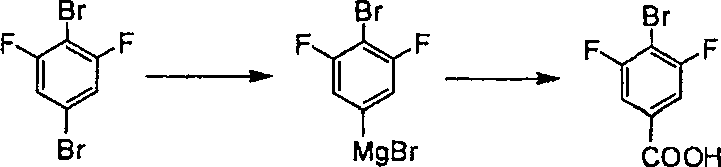
When generating the Grignard reagent from 3,5-difluoro-1,4-dibromobenzene, there is an equal probability of magnesium insertion at either bromine position, leading to a mixture of isomers and drastically reduced selectivity. This lack of regiocontrol results in difficult purification processes, lower overall yields, and the generation of substantial chemical waste, rendering the method unsuitable for efficient industrialization. The instability of the generated intermediates further complicates the process, often leading to decomposition before the desired carboxylation can occur.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique reactivity of organolithium reagents to achieve precise regioselective substitution. By utilizing 3,5-difluorobromobenzene as the starting material, the process bypasses the ambiguity of dual-halogen substrates entirely. The core innovation lies in the use of sterically hindered amine lithium reagents, which direct the lithiation specifically to the 4-position of the aromatic ring. This method operates under mild reaction conditions, typically ranging from -100°C to 50°C, and utilizes common ether solvents such as tetrahydrofuran (THF) or methyl tertiary butyl ether. The simplicity of the post-treatment process, involving straightforward hydrolysis and acidification, allows for easy isolation of the target acid. This shift in synthetic strategy not only simplifies the operational workflow but also significantly enhances the economic viability of producing high-purity fluorinated benzoic acids on a commercial scale.
Mechanistic Insights into Sterically Hindered Organolithium Lithiation
The success of this synthesis hinges on the precise mechanistic interaction between the substrate and the bulky lithium base. When 3,5-difluorobromobenzene is treated with a standard organolithium reagent, multiple lithiation sites (2, 4, and 6 positions) are theoretically possible, which would lead to a complex mixture of by-products. However, the patent specifies the use of sterically hindered amine lithium reagents, such as 2,2,6,6-tetramethylpiperidine lithium (LTMP) or N-Lithiodiisopropylamide (LDA). The substantial steric bulk of these reagents effectively blocks access to the more crowded ortho-positions relative to the fluorine atoms or the bromine, thereby kinetically favoring deprotonation at the less hindered 4-position. This steric guidance ensures that the resulting 4-substituted lithium salt is formed with exceptional regioselectivity, reported to exceed 99% in preferred embodiments.
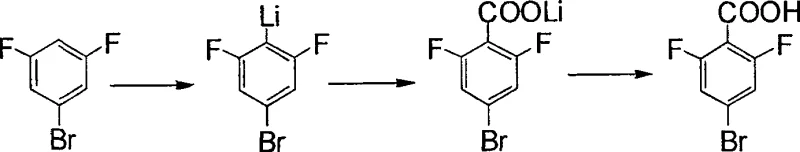
Furthermore, the stability of the intermediate lithium salt is crucial for the subsequent carboxylation step. The patent highlights that the specific combination of solvents and hindered bases prevents the decomposition of the organometallic intermediate prior to the introduction of carbon dioxide. Once the stable lithium salt is formed, the introduction of CO2 gas leads to the formation of the carboxylate salt, which is subsequently hydrolyzed to the free acid. This mechanism effectively controls the impurity profile by minimizing side reactions such as halogen-lithium exchange or multiple lithiations, which are common pitfalls in fluorinated aromatic chemistry. For quality control teams, this mechanistic clarity translates to a predictable and consistent impurity spectrum, facilitating easier validation and regulatory compliance for pharmaceutical applications.
How to Synthesize 4-Bromo-2,6-Difluorobenzoic Acid Efficiently
Implementing this synthesis requires careful attention to solvent selection and temperature control to maximize the benefits of the organolithium pathway. The process begins with the preparation of the organolithium reagent in an aprotic polar solvent, followed by the controlled addition of the fluorinated substrate. Detailed operational parameters, including specific molar ratios of solvent to substrate (4:1 to 15:1) and precise temperature ramps during the CO2 saturation phase, are critical for reproducing the high yields described in the patent examples. Manufacturers should note that while the reaction window is broad, optimizing the stoichiometry of the lithium reagent (preferably 1:1 to 1:1.05) is key to preventing excess reagent from causing side reactions. The standardized synthesis steps outlined below provide a foundational protocol for scaling this chemistry from laboratory benchtop to pilot plant operations.
- Prepare the reaction system by mixing an organic solvent (such as THF or ether) with a sterically hindered amine lithium reagent like LTMP or LDA at temperatures between -100°C and 50°C.
- Add 3,5-difluorobromobenzene to the mixture to effect regioselective lithiation at the 4-position, forming the stable lithium salt intermediate.
- Introduce carbon dioxide gas to the lithium salt intermediate followed by acidic hydrolysis to yield the final 4-bromo-2,6-difluorobenzoic acid product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organolithium-based methodology offers tangible strategic advantages beyond mere technical feasibility. The primary benefit lies in the utilization of 3,5-difluorobromobenzene, a commercially abundant and cost-effective starting material, compared to the specialized and potentially scarce 3,5-difluoro-1,4-dibromobenzene required for older methods. This shift in raw material sourcing mitigates supply risk and stabilizes input costs, ensuring a more resilient supply chain for downstream customers. Additionally, the simplified workup procedure eliminates the need for complex purification steps often associated with Grignard by-products, directly translating to reduced processing time and lower utility consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available ether solvents significantly lowers the direct material cost of goods sold. By achieving high regioselectivity without the need for protective group strategies or extensive chromatographic purification, the process reduces solvent waste and labor hours associated with downstream processing. The high molar yields, reaching up to 88.9% in optimized examples, mean that less raw material is required to produce the same amount of active intermediate, driving down the effective cost per unit. Furthermore, the ability to recycle solvents like THF or ether adds another layer of economic efficiency to the manufacturing cycle.
- Enhanced Supply Chain Reliability: Relying on industrial-grade raw materials that are easily purchasable ensures that production schedules are not held hostage by the availability of niche precursors. The robustness of the reaction conditions, which tolerate a relatively wide temperature range and utilize standard chemical equipment, means that production can be easily transferred between different manufacturing sites without significant requalification burdens. This flexibility is crucial for maintaining continuity of supply in the face of global logistical disruptions. The high purity of the crude product (often exceeding 99% HPLC content before recrystallization) also reduces the lead time for quality assurance testing and release.
- Scalability and Environmental Compliance: The process is inherently designed for industrialization, with reaction exotherms that are manageable through standard cooling systems. The avoidance of heavy metal oxidants like potassium bichromate aligns with increasingly stringent environmental regulations regarding wastewater treatment and hazardous waste disposal. The 'little pollution' characteristic mentioned in the patent abstract suggests a greener manufacturing footprint, which is a growing requirement for multinational corporations auditing their supplier base. Scaling from 100 kgs to multi-ton batches is facilitated by the homogeneous nature of the reaction mixture and the straightforward gas-liquid contact required for the carboxylation step.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN100522920C, providing a reliable reference for technical due diligence. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this intermediate into their own value chains.
Q: Why is the organolithium method superior to the Grignard route for this specific compound?
A: The conventional Grignard method using 3,5-difluoro-1,4-dibromobenzene suffers from poor selectivity because magnesium can react with either bromine atom randomly. The novel organolithium route using 3,5-difluorobromobenzene and sterically hindered bases achieves over 99% regioselectivity at the 4-position.
Q: What specific reagents are required to ensure high stability of the intermediate?
A: The patent specifies the use of sterically hindered amine lithium reagents such as 2,2,6,6-tetramethylpiperidine lithium (LTMP), N-Lithiodiisopropylamide (LDA), or Tetramethyl Ethylene Diamine lithium. These bulky groups prevent side reactions and stabilize the 4-position lithium salt.
Q: What yields and purity levels can be expected from this process?
A: Experimental data in the patent demonstrates molar yields ranging from 67% to nearly 89%, with HPLC purity consistently exceeding 99% after recrystallization, making it highly suitable for pharmaceutical grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2,6-Difluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex fluorinated intermediates requires more than just a patent; it demands deep process engineering expertise and rigorous quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. We are committed to delivering high-purity 4-bromo-2,6-difluorobenzoic acid that meets stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our capability to adapt the organolithium chemistry described in CN100522920C allows us to offer a product that balances cost-efficiency with the high quality demanded by the pharmaceutical and agrochemical sectors.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies translate into value for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a secure and competitive supply of this critical building block for your future innovations.
