Scaling Green Synthesis of UV Absorber Intermediates for Commercial Production
The chemical industry is currently witnessing a paradigm shift towards greener, safer synthetic routes for high-value intermediates, particularly in the realm of UV absorbers and fine chemicals. Patent CN102329207A introduces a robust and environmentally conscious methodology for the synthesis of 2,2'-dihydroxy-4,4'-dimethoxybenzophenone, a critical compound widely utilized in sunscreen formulations and polymer stabilization. This technical disclosure moves away from hazardous traditional reagents, opting instead for a controlled acylation strategy using oxalyl chloride coupled with a precise Lewis acid-mediated demethylation. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize supply chains by adopting a process that balances high purity standards with operational safety. The detailed reaction pathways outlined in the document provide a clear roadmap for transitioning from laboratory-scale experiments to multi-ton commercial production without compromising on quality or environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzophenone derivatives has heavily relied on the use of phosgene, a highly toxic and volatile gas that poses severe safety risks during industrial handling. The logistical burden of storing and transporting phosgene requires specialized infrastructure and rigorous safety protocols, which inevitably drives up capital expenditure and operational costs for manufacturers. Furthermore, the reactivity of phosgene can be difficult to control precisely, often leading to side reactions that complicate downstream purification and reduce overall yield. These traditional methods also generate significant hazardous waste, conflicting with modern green chemistry principles and increasing the environmental footprint of the manufacturing process. Consequently, there is an urgent industry demand for alternative acylating agents that offer similar reactivity profiles but with vastly improved safety and handling characteristics.
The Novel Approach
The innovative process described in the patent addresses these challenges by substituting phosgene with oxalyl chloride, a liquid reagent that is far easier to manage in a standard chemical plant setting. This substitution allows for a more controlled Friedel-Crafts acylation reaction between m-xylylene dimethyl ether and the acylating agent, facilitated by radical initiators such as azoisobutyronitrile or benzoyl peroxide. The reaction proceeds efficiently at moderate temperatures of 70-80°C, minimizing energy consumption while ensuring high conversion rates to the tetramethoxy intermediate. By eliminating the need for gaseous reagents, the process simplifies reactor design and reduces the risk of accidental exposure, thereby enhancing overall plant safety. This strategic shift not only aligns with regulatory trends favoring safer chemicals but also streamlines the procurement of raw materials, as oxalyl chloride is a widely available commodity chemical.
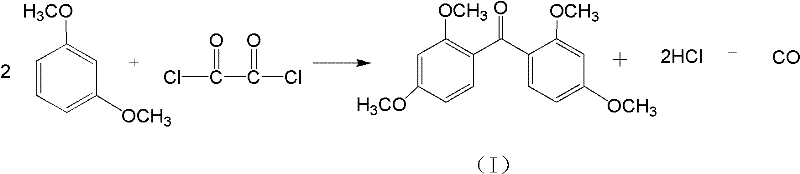
Mechanistic Insights into Oxalyl Chloride Acylation and Lewis Acid Demethylation
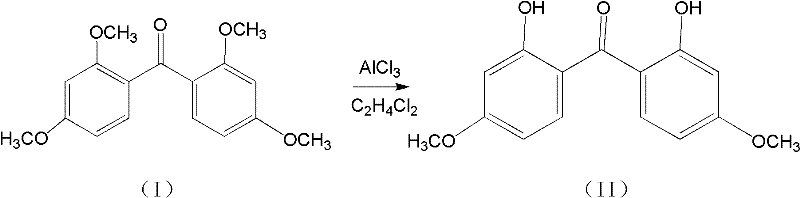
The core of this synthetic strategy lies in the dual-step mechanism that ensures both structural integrity and functional group transformation. In the first stage, the radical initiator decomposes to generate free radicals that activate the oxalyl chloride, promoting its electrophilic attack on the electron-rich aromatic ring of the m-xylylene dimethyl ether. This step is crucial for forming the carbon-carbon bond that links the two aromatic systems via the carbonyl bridge, creating the 2,2',4,4'-tetramethoxybenzophenone skeleton. The use of specific catalysts like AIBN ensures that the reaction kinetics are optimized, preventing polymerization or over-chlorination which could lead to impurities. Following this, the second stage involves a sophisticated Lewis acid-catalyzed demethylation where reagents like aluminum chloride coordinate with the methoxy oxygen atoms. This coordination weakens the carbon-oxygen bond, allowing for the selective cleavage of the methyl groups to reveal the phenolic hydroxyl groups essential for UV absorption activity.
Controlling the purity of the final product requires meticulous management of the demethylation conditions, particularly the temperature and the choice of Lewis acid. The patent specifies a reaction temperature of 50°C, which is low enough to prevent thermal degradation of the sensitive benzophenone core yet high enough to drive the demethylation to completion within a reasonable timeframe of 2-3 hours. The selection of the solvent, such as dichloroethane or chlorobenzene, plays a pivotal role in solubilizing the Lewis acid complex and facilitating the mass transfer required for efficient reaction. Post-reaction workup involves careful hydrolysis to quench the Lewis acid, followed by phase separation and recrystallization, which effectively removes inorganic salts and residual organic impurities. This rigorous purification protocol ensures that the final 2,2'-dihydroxy-4,4'-dimethoxybenzophenone meets the stringent purity specifications required for pharmaceutical and cosmetic applications.
How to Synthesize 2,2'-Dihydroxy-4,4'-dimethoxybenzophenone Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the reaction vessel under inert gas protection to prevent moisture interference, followed by the precise addition of the radical catalyst and oxalyl chloride. Operators must maintain strict temperature control during the exothermic acylation phase to avoid runaway reactions, ensuring the formation of the intermediate proceeds smoothly. Once the intermediate is isolated, the subsequent demethylation step demands accurate stoichiometry of the Lewis acid to ensure complete conversion without excessive reagent usage. Detailed standardized operating procedures for each unit operation, from filtration to recrystallization, are essential for reproducibility.
- React m-xylylene dimethyl ether with oxalyl chloride at 70-80°C using AIBN or benzoyl peroxide catalyst to form the tetramethoxy intermediate.
- Treat the intermediate with a Lewis acid (e.g., AlCl3) in an organic solvent like dichloroethane at 50°C for 2-3 hours.
- Quench the reaction with water, separate layers, remove solvent via rotary evaporation, and recrystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for UV absorber intermediates. The replacement of phosgene with oxalyl chloride eliminates the need for specialized gas handling infrastructure, resulting in significantly reduced capital investment and lower maintenance costs for production facilities. Additionally, the liquid nature of oxalyl chloride simplifies logistics and storage, reducing the risks associated with transportation and ensuring a more reliable supply of raw materials. The mild reaction conditions further contribute to cost efficiency by lowering energy consumption and extending the lifespan of reactor equipment due to reduced thermal stress. These factors collectively enhance the economic viability of the process, making it an attractive option for large-scale manufacturing.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling systems and the use of readily available liquid reagents lead to a drastic simplification of the production workflow. By avoiding the complex safety measures required for phosgene, manufacturers can allocate resources more efficiently towards quality control and process optimization. The high selectivity of the reaction minimizes the formation of by-products, reducing the burden on purification steps and lowering the overall cost of goods sold. Furthermore, the ability to recover and recycle solvents like dichloroethane adds another layer of economic benefit, contributing to long-term sustainability and profitability.
- Enhanced Supply Chain Reliability: Sourcing oxalyl chloride and standard Lewis acids like aluminum chloride is far more straightforward than managing the supply of regulated toxic gases. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages or regulatory hurdles. The robustness of the process allows for flexible production planning, enabling suppliers to respond quickly to fluctuations in market demand. Moreover, the stability of the intermediate and final products facilitates easier storage and distribution, reducing the risk of degradation during transit and ensuring consistent quality upon delivery to customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions do not impose severe limitations on reactor size or mixing efficiency. The use of common organic solvents and standard catalysts means that existing chemical plants can easily adapt their current infrastructure to accommodate this synthesis without major retrofits. From an environmental standpoint, the reduction in hazardous waste generation and the avoidance of persistent toxic reagents align with global sustainability goals. This compliance not only mitigates regulatory risks but also enhances the brand reputation of manufacturers as responsible stewards of the environment, appealing to eco-conscious clients in the personal care and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this UV absorber intermediate. These insights are derived directly from the patented methodology to provide clarity on process capabilities and product specifications. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this material into their supply chains.
Q: Why is oxalyl chloride preferred over phosgene for this synthesis?
A: Oxalyl chloride is a liquid at room temperature, making it significantly safer and easier to transport and store compared to toxic gaseous phosgene, while maintaining high reactivity for acylation.
Q: What represents the critical control point in the demethylation step?
A: Maintaining the reaction temperature at 50°C is critical to ensure selective demethylation without degrading the benzophenone core structure, ensuring high purity yields.
Q: Can this process be scaled for industrial production?
A: Yes, the mild reaction conditions (70-80°C and 50°C) and the use of standard organic solvents like dichloroethane make this process highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dihydroxy-4,4'-dimethoxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic technologies like the one described in CN102329207A to deliver high-performance chemical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest multinational corporations. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,2'-dihydroxy-4,4'-dimethoxybenzophenone performs consistently in your final formulations. Our dedication to green chemistry principles means that our manufacturing processes are not only efficient but also environmentally responsible, aligning with the sustainability targets of modern enterprises.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating the tangible economic advantages of switching to our supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-quality fine chemical intermediates.
