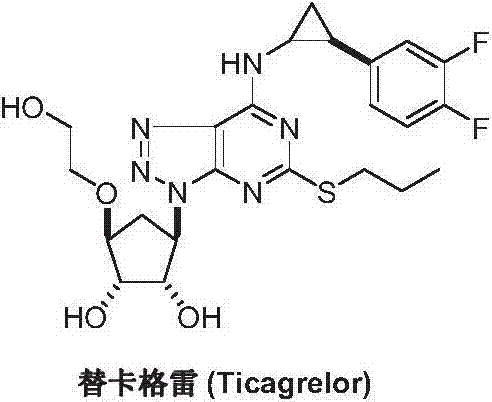
Advanced Synthesis of Ticagrelor Intermediate C: A Scalable Route for Global API Manufacturing
The pharmaceutical landscape for antiplatelet therapy has been significantly shaped by the introduction of Ticagrelor, a reversible P2Y12 receptor antagonist that offers superior efficacy in managing acute coronary syndromes. As global demand for this critical medication continues to rise, the efficiency and reliability of its supply chain become paramount for multinational pharmaceutical corporations. Patent CN103588750A introduces a groundbreaking methodology for synthesizing a pivotal precursor, specifically 2-[[(3aR,4S,6R,6aS)-6-aminotetrahydro-2,2-dimethyl-4H-cyclopenta-1,3-dioxan-4-yl]oxy]ethanol, commonly referred to as Intermediate C. This technical disclosure represents a strategic shift away from cumbersome traditional syntheses towards a more streamlined, cost-effective, and scalable chemical pathway. By leveraging a unique azide ring-opening strategy coupled with tandem ketalization, this innovation addresses long-standing bottlenecks in the production of high-purity pharmaceutical intermediates. For R&D directors and procurement leaders, understanding the nuances of this patented route is essential for securing a robust supply of active pharmaceutical ingredients (APIs) while optimizing manufacturing expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ticagrelor Intermediate C has relied heavily on complex multi-step sequences that pose significant challenges for industrial scale-up and cost management. Traditional routes often commence with D-ribose or involve Diels-Alder reactions using cyclopentadiene, both of which introduce substantial inefficiencies into the manufacturing process. For instance, pathways originating from D-ribose necessitate a rigorous series of protection and deprotection steps to manage the multiple hydroxyl groups inherent to the sugar backbone, leading to increased solvent consumption, extended reaction times, and lower overall yields. Furthermore, Diels-Alder approaches frequently suffer from issues related to regioselectivity and the requirement for harsh reaction conditions that can compromise safety and environmental compliance. These legacy methods often result in a proliferation of impurities that are difficult to remove, thereby increasing the burden on downstream purification processes and quality control laboratories. The cumulative effect of these drawbacks is a supply chain that is vulnerable to disruptions and characterized by elevated production costs, making it less attractive for large-scale commercial operations seeking reliable pharmaceutical intermediate suppliers.
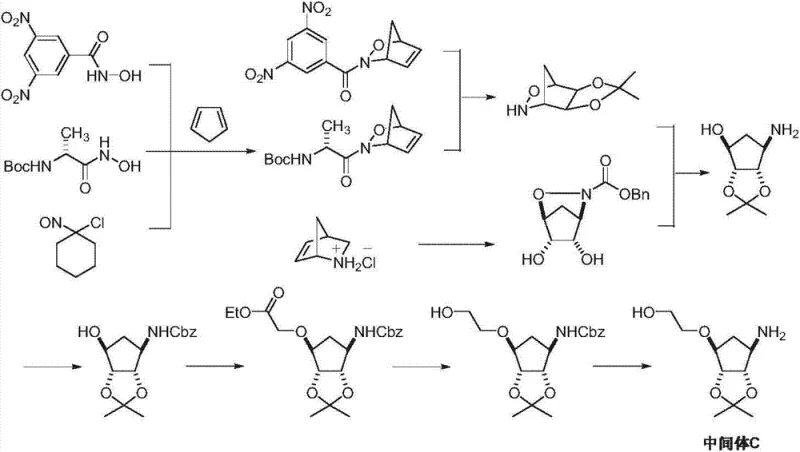
The Novel Approach
In stark contrast to these convoluted legacy pathways, the methodology disclosed in CN103588750A offers a remarkably concise and elegant solution by utilizing (1R,2R,3R,4R)-rel-1,2;3,4-diepoxy cyclopentane as the foundational building block. This innovative strategy capitalizes on the inherent reactivity of the bis-epoxide system to simultaneously achieve ring-opening and ketalization in a single operational step, effectively collapsing what were previously distinct stages into a unified transformation. By employing sodium azide in a mixed solvent system of acetone and water, the process not only installs the necessary nitrogen functionality but also establishes the critical acetonide protecting group with high stereochemical fidelity. This reduction in step count is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it directly correlates to reduced material handling, lower energy consumption, and minimized waste generation. Moreover, the subsequent etherification and reduction steps are designed to proceed under mild conditions using commercially available reagents, ensuring that the process remains robust and easily transferable to multi-tonne production scales without compromising product integrity or safety standards.
Mechanistic Insights into Azide-Mediated Ring Opening and Tandem Ketalization
The core chemical innovation of this patent lies in the sophisticated orchestration of nucleophilic attack and protecting group chemistry during the initial transformation of the diepoxide starting material. When (1R,2R,3R,4R)-rel-1,2;3,4-diepoxy cyclopentane is exposed to sodium azide in the presence of acetone and water, a highly selective nucleophilic ring-opening occurs at one of the epoxide rings. The azide ion acts as a potent nucleophile, attacking the less hindered carbon of the epoxide to generate an intermediate alcohol, which immediately participates in an acid-catalyzed ketalization with the acetone solvent. This tandem sequence is mechanistically fascinating because it locks the stereochemistry of the newly formed centers while simultaneously protecting the vicinal diol system as an acetonide, preventing unwanted side reactions in subsequent steps. The choice of solvent ratio, specifically an acetone-to-water volume ratio of approximately 1:1, is critical for balancing the solubility of the inorganic azide salt with the organic substrate, thereby maximizing the reaction rate and conversion efficiency. This precise control over reaction parameters ensures that the formation of the desired (3aR,4S,6R,6aS)-rel-6-azido intermediate proceeds with minimal formation of regioisomers or hydrolysis byproducts, setting a high standard for purity right from the outset of the synthesis.
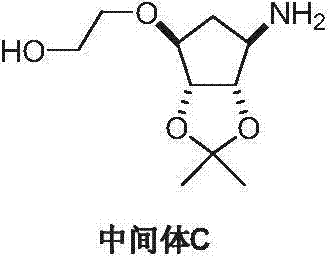
Following the initial ring construction, the maintenance of optical purity and the efficient installation of the hydroxyethoxy side chain are achieved through carefully optimized etherification and reduction protocols. The conversion of the azido-alcohol intermediate to the corresponding ether involves the use of strong bases such as sodium hydride or potassium tert-butoxide to deprotonate the hydroxyl group, facilitating a nucleophilic substitution with 2-haloethanol. This step is crucial for introducing the ethoxy linker that will eventually connect to the triazolopyrimidine core of the final drug molecule. Subsequently, the reduction of the azide moiety to the primary amine is performed using catalytic methods, such as palladium on carbon with Hantzsch ester, which offers a safer and cleaner alternative to stoichiometric metal reductions that generate heavy metal waste. The final resolution step, utilizing chiral acids like L-tartaric acid, serves as the ultimate gatekeeper for quality, ensuring that only the biologically active enantiomer proceeds to the final coupling stage. This rigorous approach to impurity control and stereochemical definition is essential for meeting the stringent regulatory requirements imposed by global health authorities on cardiovascular medications.
How to Synthesize Ticagrelor Intermediate C Efficiently
Implementing this advanced synthesis requires a disciplined adherence to the specific reaction conditions and stoichiometry outlined in the patent to ensure reproducibility and high yield. The process begins with the careful preparation of the reaction mixture, ensuring that the molar ratio of the diepoxide to sodium azide is maintained within the optimal range of 1:1.1 to 1:1.3 to drive the reaction to completion without excessive reagent waste. Operators must monitor the reaction progress closely, typically using gas chromatography or TLC, to determine the precise endpoint before proceeding to the extraction and workup phases. The subsequent etherification and reduction steps demand strict control over temperature and atmosphere, particularly the use of inert gas protection to prevent oxidation of sensitive intermediates. While the general workflow is straightforward, the devil is in the details regarding solvent selection, drying agents, and crystallization conditions, all of which contribute to the final quality of the product. For a comprehensive breakdown of the exact operational parameters, temperatures, and workup procedures, please refer to the standardized synthesis guide below.
- Perform simultaneous azide ring-opening and ketalization on (1R,2R,3R,4R)-rel-1,2;3,4-diepoxy cyclopentane using sodium azide in acetone/water to form the azido-ketal intermediate.
- Conduct etherification of the azido-ketal intermediate with 2-haloethanol using a strong base like sodium hydride or potassium tert-butoxide to install the hydroxyethoxy side chain.
- Reduce the azide group to an amine using catalytic hydrogenation or Hantzsch ester, followed by chiral resolution using L-tartaric acid to isolate the target enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound benefits for procurement managers and supply chain executives tasked with optimizing the cost structure and reliability of API production. The most immediate impact is seen in the drastic simplification of the manufacturing workflow, which eliminates the need for expensive chiral pool starting materials like D-ribose and removes multiple protection-deprotection cycles that traditionally inflate production costs. By shortening the synthetic sequence, manufacturers can significantly reduce the consumption of solvents, reagents, and utilities, leading to substantial cost savings in pharmaceutical intermediate manufacturing without sacrificing quality. Furthermore, the use of readily available and inexpensive raw materials, such as the rel-diepoxy cyclopentane and common inorganic salts, mitigates the risk of supply shortages and price volatility associated with specialty chemicals. This resilience in the supply base ensures a more predictable and stable flow of materials, allowing for better inventory management and long-term planning. Additionally, the greener nature of the process, characterized by aqueous workups and catalytic reductions, aligns with increasingly strict environmental regulations, reducing the liability and cost associated with waste disposal and environmental compliance.
- Cost Reduction in Manufacturing: The elimination of lengthy protection groups and the use of cheaper starting materials directly lowers the bill of materials, while fewer processing steps reduce labor and overhead costs significantly.
- Enhanced Supply Chain Reliability: Sourcing robust, commodity-grade chemicals instead of scarce chiral sugars ensures consistent availability and reduces the risk of production delays caused by raw material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced solvent load make this process inherently safer and easier to scale from pilot plant to commercial tonnage, while minimizing the environmental footprint.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific details provided in the intellectual property documentation. These inquiries address common concerns regarding raw material sourcing, stereochemical control, and the practical feasibility of scaling this route for industrial applications. Understanding these aspects is vital for technical teams evaluating the viability of integrating this intermediate into their existing supply chains. The answers provided are derived strictly from the experimental data and claims presented in the patent, ensuring accuracy and relevance for decision-makers.
Q: What is the primary starting material for this novel Ticagrelor Intermediate C synthesis?
A: The process utilizes (1R,2R,3R,4R)-rel-1,2;3,4-diepoxy cyclopentane as the key starting material, which undergoes a tandem azide ring-opening and ketalization reaction.
Q: How does this method improve upon traditional D-ribose based routes?
A: Unlike D-ribose routes that require extensive protection and deprotection steps, this method achieves ring formation and functionalization in fewer steps with readily available epoxide precursors, significantly simplifying the workflow.
Q: What resolution agent is preferred for isolating the target enantiomer?
A: The patent specifies L-tartaric acid or D-mandelic acid as preferred resolving agents to separate the racemic amine mixture and obtain the high-purity (3aR,4S,6R,6aS) enantiomer required for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Intermediate C Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and a proven track record in process development. Our team of seasoned chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising potential of this azide-mediated synthesis is fully realized in a GMP-compliant environment. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with a focus on safety, efficiency, and environmental stewardship, making us an ideal partner for bringing this advanced Ticagrelor intermediate to the global market. By leveraging our infrastructure and expertise, we can help you secure a stable supply of this critical building block while optimizing your overall production economics.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific volume requirements and quality standards. Request a Customized Cost-Saving Analysis today to understand the potential financial impact of switching to this more efficient manufacturing method. Our experts are ready to provide specific COA data and route feasibility assessments to support your internal evaluation processes. Let us collaborate to enhance the resilience and profitability of your pharmaceutical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
