Revolutionizing Agrochemical Intermediates: Advanced Catalytic Synthesis of cis-1,2,3,6-Tetrahydrophthalimide
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of critical intermediates, specifically cis-1,2,3,6-tetrahydrophthalimide (CAS 85-40-5), a pivotal building block for fungicides such as Captan and Captafol. A groundbreaking technical advancement detailed in patent CN115353481A introduces a highly efficient catalytic synthesis method that fundamentally redefines the production landscape for this essential compound. Unlike conventional approaches that struggle with energy intensity and impurity profiles, this novel protocol leverages a sophisticated one-pot strategy utilizing low-boiling aromatic solvents and organic acid catalysis to achieve unprecedented operational simplicity. The process initiates with the controlled ammoniation of cis-1,2,3,6-tetrahydrophthalic anhydride in a solvent environment, followed by a seamless transition to a dehydration ring-closing reaction that eliminates the need for intermediate isolation. By integrating these steps into a unified workflow, the technology not only mitigates the generation of hazardous waste gases but also ensures a product purity exceeding 99 percent with yields consistently surpassing 97.9 percent. For industry leaders seeking a reliable agrochemical intermediate supplier, this patent represents a paradigm shift towards greener, more cost-effective manufacturing capabilities that align perfectly with modern sustainability mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cis-1,2,3,6-tetrahydrophthalimide has been plagued by significant technical bottlenecks that compromise both economic efficiency and environmental safety. Prior art methods, such as those disclosed in CN108484475A, often involve batch feeding of phthalic anhydride at elevated temperatures, a practice that frequently leads to the formation of intractable polymeric jellies which drastically reduce selectivity and complicate downstream separation processes. Furthermore, alternative routes described in patents like CN111454196A necessitate the melting of solid anhydride at temperatures around 110°C followed by reaction with urea, a pathway that generates substantial volumes of waste gas and requires rigorous temperature control to prevent safety incidents. Other gas-solid phase reactions, as seen in CN110606819A, suffer from poor mixing efficiency and slow reaction kinetics due to the inherent limitations of introducing ammonia gas into a solid matrix, resulting in prolonged reaction times and inconsistent product quality. These legacy processes are characterized by high energy consumption, extensive wastewater generation requiring complex treatment, and operational risks associated with high-temperature exothermic events, making them increasingly untenable in a regulated global market.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the catalytic synthesis method outlined in CN115353481A offers a streamlined solution that addresses every major pain point of traditional manufacturing. By conducting the initial ammoniation in a liquid phase using a low-boiling aromatic solvent like toluene, the process ensures homogeneous mixing and precise temperature control between 30°C and 50°C, effectively suppressing side reactions and minimizing ammonia consumption. The subsequent dehydration ring-closing step is revolutionized by the introduction of an organic acid catalyst, such as p-toluenesulfonic acid, which dramatically accelerates the cyclization rate while allowing the reaction to proceed at the moderate reflux temperature of the solvent. This innovative approach eliminates the need for high-temperature melting or hazardous gas-solid interactions, thereby removing the risk of jelly formation and ensuring a smooth, continuous flow of materials. The result is a robust, scalable process that delivers superior yield and purity metrics while significantly reducing the environmental footprint through the elimination of waste gas and the minimization of wastewater discharge.
Mechanistic Insights into Acid-Catalyzed Dehydration Cyclization
The core chemical transformation in this advanced synthesis relies on a meticulously orchestrated two-stage mechanism that maximizes atomic economy and reaction efficiency. In the first stage, cis-1,2,3,6-tetrahydrophthalic anhydride undergoes nucleophilic attack by ammonia in the presence of toluene, forming an unstable ammonium salt intermediate that rapidly rearranges into the corresponding amide-acid species. The use of a solvent medium is critical here, as it solubilizes the anhydride and facilitates the diffusion of aqueous ammonia, ensuring that the molar ratio of anhydride to ammonia remains tightly controlled between 1:1.01 and 1:1.03 to prevent excess reagent accumulation. Following the formation of this intermediate, the reaction mixture undergoes a phase separation where the aqueous layer, containing excess water and ammonia, is removed, leaving the organic phase rich in the precursor ready for cyclization. This strategic removal of water prior to the high-temperature step is essential for driving the equilibrium forward and preventing hydrolysis of the sensitive intermediate.
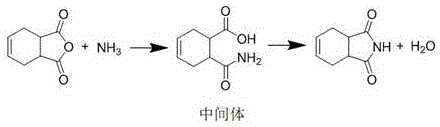
The second stage involves the acid-catalyzed dehydration ring-closure, where the addition of p-toluenesulfonic acid serves as the kinetic driver for imide formation. The catalyst protonates the carbonyl oxygen of the carboxylic acid group in the intermediate, increasing its electrophilicity and facilitating the nucleophilic attack by the adjacent amide nitrogen. This intramolecular cyclization releases a molecule of water, which is continuously removed via azeotropic distillation with the toluene solvent using a water separator, thus pushing the reaction to completion according to Le Chatelier's principle. The presence of the catalyst lowers the activation energy barrier sufficiently to allow the reaction to reach completion within just 0.5 to 1 hour at reflux, compared to several hours required in uncatalyzed thermal processes. This mechanistic precision ensures that the final product, cis-1,2,3,6-tetrahydrophthalimide, crystallizes with exceptional purity upon cooling, free from the polymeric byproducts that typically contaminate non-catalytic high-temperature runs.
How to Synthesize cis-1,2,3,6-Tetrahydrophthalimide Efficiently
Implementing this catalytic synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield on a commercial scale. The process begins with the dissolution of the anhydride starting material in toluene, followed by the controlled dropwise addition of 25wt% aqueous ammonia at temperatures maintained between 30°C and 50°C to manage the exotherm effectively. Once the ammoniation is complete and the water layer is separated, the critical cyclization step is initiated by adding the organic acid catalyst and heating the mixture to reflux, with continuous monitoring of the intermediate concentration via HPLC to determine the precise endpoint. The detailed standardized operating procedures, including specific stirring rates, cooling profiles for crystallization, and filtration protocols, are essential for maintaining the high-quality standards expected by downstream agrochemical manufacturers.
- Dissolve cis-1,2,3,6-tetrahydrophthalic anhydride in toluene and react with aqueous ammonia at 30-50°C to form the intermediate amide.
- Separate the aqueous layer from the organic phase to remove excess water and unreacted ammonia without filtration.
- Add p-toluenesulfonic acid catalyst to the organic phase and heat to reflux (110°C) for dehydration ring-closure to yield the final imide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic synthesis technology translates directly into tangible operational improvements and risk mitigation strategies that enhance the overall resilience of the supply network. The elimination of complex solid-state handling and the reduction of reaction times significantly streamline the production cycle, allowing for faster turnaround times and more flexible response to market demand fluctuations without compromising product integrity. Furthermore, the simplified one-pot design reduces the number of unit operations required, which inherently lowers the capital expenditure associated with equipment maintenance and the operational expenditure related to labor and energy consumption. By shifting away from hazardous gas-solid reactions and high-temperature melting processes, facilities can also reduce their insurance premiums and regulatory compliance burdens, creating a safer working environment that aligns with corporate social responsibility goals.
- Cost Reduction in Manufacturing: The implementation of this catalytic method drives down manufacturing costs through multiple mechanisms, primarily by eliminating the need for expensive high-energy heating cycles and complex waste treatment systems associated with traditional methods. The use of a recyclable low-boiling solvent like toluene, combined with the high selectivity of the acid catalyst, minimizes raw material loss and reduces the volume of waste solvent that requires disposal or regeneration. Additionally, the significant improvement in yield, consistently achieving levels above 97.9 percent, means that less starting material is required to produce the same amount of finished goods, directly lowering the cost of goods sold and improving margin potential for buyers seeking cost reduction in agrochemical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this liquid-phase process ensures a consistent and reliable supply of high-purity cis-1,2,3,6-tetrahydrophthalimide, mitigating the risks of production delays caused by equipment fouling or batch failures common in older technologies. The ability to operate at lower temperatures and pressures reduces the likelihood of unplanned shutdowns due to safety interlocks or thermal runaway events, thereby guaranteeing continuity of supply for critical downstream applications. Moreover, the simplified workflow reduces the dependency on specialized operators for complex solid-handling tasks, making the production line less susceptible to labor shortages and training bottlenecks that can disrupt delivery schedules.
- Scalability and Environmental Compliance: From a scalability perspective, the homogeneous nature of the reaction mixture allows for seamless translation from pilot scale to multi-ton commercial production without the mixing and heat transfer limitations often encountered in heterogeneous gas-solid systems. The process generates no waste gas and significantly less wastewater compared to prior art, facilitating easier compliance with increasingly stringent environmental regulations and reducing the need for costly end-of-pipe treatment infrastructure. This environmental compatibility not only future-proofs the supply chain against regulatory changes but also enhances the brand value of the final agrochemical products by associating them with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced catalytic synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a transparent view of the process capabilities and advantages over existing methods. Understanding these details is crucial for making informed decisions about sourcing strategies and process optimization initiatives within the agrochemical sector.
Q: How does this catalytic method improve upon traditional high-temperature melting processes?
A: Traditional methods often require melting solid anhydride at temperatures exceeding 110°C or using urea, which generates significant waste gas and poses safety risks. This patented catalytic method operates in a liquid phase at lower temperatures (reflux of toluene at 110°C with catalyst vs higher temps without), significantly reducing energy consumption and eliminating hazardous gas emissions while preventing the formation of difficult-to-handle polymeric jellies.
Q: What is the role of p-toluenesulfonic acid in the synthesis?
A: p-Toluenesulfonic acid acts as a potent organic acid catalyst that lowers the activation energy for the dehydration ring-closing reaction. Its presence allows the reaction to proceed efficiently at the reflux temperature of low-boiling solvents like toluene, reducing the reaction time from several hours to approximately 30 minutes and improving the overall yield to over 97.9%.
Q: Why is the separation of the water layer critical before the cyclization step?
A: Removing the water layer generated during the initial ammoniation step shifts the equilibrium towards the intermediate amide and prevents hydrolysis during the subsequent high-temperature cyclization. This phase separation simplifies the workflow by avoiding complex filtration steps required in solid-state reactions, thereby enhancing process continuity and throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-1,2,3,6-Tetrahydrophthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies requires a partner with deep technical expertise and a proven track record of scaling complex chemical pathways. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115353481A are fully realized in practical, large-volume deliveries. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of cis-1,2,3,6-tetrahydrophthalimide meets the exacting standards required for pesticide synthesis, guaranteeing consistency and performance for your final formulations.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements and operational context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make a data-driven decision that secures a competitive advantage in the global agrochemical market.
