Revolutionizing 3-Alkenyl Oxindole Production with Green Iron Catalysis for Commercial Scale-Up
Revolutionizing 3-Alkenyl Oxindole Production with Green Iron Catalysis for Commercial Scale-Up
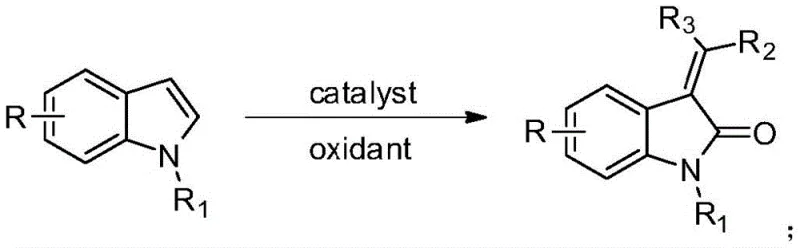
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways to access critical heterocyclic scaffolds, particularly those found in oncology therapeutics. Patent CN115925609A introduces a groundbreaking preparation method for 3-alkenyl oxindole derivatives that fundamentally shifts the paradigm from precious metal catalysis to earth-abundant iron chemistry. This technology leverages a visible-light-mediated oxidative coupling strategy, utilizing inexpensive ferrous chloride as the catalyst and molecular oxygen as the terminal oxidant. By replacing traditional stoichiometric oxidants and expensive transition metals, this process addresses key pain points in modern API manufacturing, including environmental impact, heavy metal contamination, and overall cost of goods sold. The method operates under remarkably mild conditions, utilizing a blue LED light source to drive the reaction efficiency, thereby offering a robust solution for the synthesis of complex intermediates used in drugs like Sunitinib and BRD4 inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-alkenyl oxindole core has relied heavily on classical condensation reactions or transition-metal-catalyzed cyclizations that present significant operational and economic hurdles. Traditional protocols often employ Lewis acid systems such as Zn-TiCl4 or Ti(OiPr)4/pyridine, which necessitate rigorous anhydrous conditions and generate substantial amounts of titanium-containing waste streams that are difficult to dispose of environmentally. Alternatively, noble metal-catalyzed approaches using Palladium or Rhodium have been explored for intramolecular aromatic C-H functionalization; however, these methods suffer from the prohibitive cost of the catalysts and the requirement for complex, pre-functionalized precursor compounds. These legacy processes often involve high temperatures and hazardous reagents, leading to safety concerns during scale-up and complicating the purification process due to the formation of metal-complexed impurities that are challenging to remove to ppm levels required for pharmaceutical grades.
The Novel Approach
In stark contrast, the methodology disclosed in CN115925609A utilizes a one-pot oxidative strategy that dramatically simplifies the synthetic workflow while enhancing sustainability. By employing a mixture of acetone and water as the solvent system, the process eliminates the need for toxic organic solvents and leverages acetone not just as a medium but potentially as a carbon source or participant in the oxidative transformation. The use of molecular oxygen (O2) at atmospheric pressure as the oxidant is a game-changer, as it replaces hazardous chemical oxidants and produces water as the only byproduct, aligning perfectly with green chemistry principles. This approach allows for the direct conversion of readily available indole substrates into valuable 3-alkenyl oxindoles in a single step, bypassing the multi-step sequences typically required to install the exocyclic double bond, thus improving overall atom economy and process mass intensity.
Mechanistic Insights into FeCl2-Catalyzed Photo-Oxidative Cyclization
The core innovation of this technology lies in the synergistic interaction between the ferrous chloride catalyst and visible light irradiation to activate molecular oxygen. Under 450nm blue LED illumination, the iron center likely undergoes a ligand-to-metal charge transfer (LMCT) or facilitates the generation of reactive oxygen species (ROS) such as superoxide radicals. These activated oxygen species then abstract hydrogen atoms or facilitate electron transfer from the indole substrate, generating a radical intermediate at the C3 position. This radical species subsequently couples with the acetone-derived fragment or undergoes oxidative dehydrogenation to form the exocyclic alkene moiety. The catalytic cycle is sustained by the continuous regeneration of the active iron species by oxygen, ensuring that only catalytic amounts (5 mol%) of the metal salt are required to drive the transformation to completion. This mechanistic pathway avoids the high-energy transition states associated with thermal condensation, allowing the reaction to proceed efficiently at room temperature or slightly elevated temperatures without degrading sensitive functional groups.
From an impurity control perspective, the mildness of this photochemical protocol offers distinct advantages over thermal methods. High-temperature reactions often promote polymerization, over-oxidation, or rearrangement side reactions that create structurally similar impurities which are difficult to separate chromatographically. By maintaining the reaction temperature near ambient conditions through LED cooling, the formation of thermal degradation products is minimized. Furthermore, the use of iron, a biocompatible metal, simplifies the downstream purification process compared to palladium or rhodium, as iron residues are easier to chelate and remove using standard scavengers or aqueous workups. This results in a cleaner crude profile, reducing the burden on final crystallization steps and ensuring that the final API intermediate meets stringent regulatory specifications for residual metals.
How to Synthesize 3-Alkenyl Oxindole Derivatives Efficiently
Implementing this synthesis requires careful attention to the light source and oxygen saturation to maximize yield and reproducibility. The patent outlines a straightforward procedure where the indole substrate is dissolved in the acetone-water mixture, and the catalyst is added prior to irradiation. Maintaining a steady stream of oxygen or an oxygen atmosphere is critical to sustain the catalytic cycle and prevent the accumulation of reduced iron species that could stall the reaction. The reaction progress is conveniently monitored by TLC, with typical completion times ranging from 5 to 10 hours depending on the electronic nature of the substituents on the indole ring. Following the reaction, a standard aqueous workup involving ethyl acetate extraction and drying over anhydrous sodium sulfate effectively isolates the product, which can be further purified by flash column chromatography if necessary.
- Charge indole substrate and ferrous chloride catalyst (5 mol%) into a reaction vessel equipped with magnetic stirring.
- Add a mixture of acetone and water (volume ratio 2.5: 1) as the solvent system and introduce oxygen gas at 1 atmosphere pressure.
- Irradiate the reaction mixture with 450nm blue LED light for 5-10 hours, then proceed to extraction and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed protocol presents a compelling value proposition centered on cost stability and raw material security. The shift away from precious metals like Palladium and Rhodium mitigates the risk associated with the volatile pricing of these commodities, which are subject to geopolitical supply constraints and mining limitations. By utilizing ferrous chloride, a commodity chemical produced in massive quantities globally, the manufacturing process becomes insulated from supply shocks, ensuring consistent availability of the catalyst at a fraction of the cost of noble metals. Additionally, the use of oxygen from the air or simple oxygen cylinders eliminates the need to purchase and store hazardous liquid oxidants, reducing both material costs and the regulatory burden associated with handling dangerous chemicals in the warehouse.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts directly lowers the variable cost per kilogram of the intermediate. Since the catalyst loading is low (5 mol%) and the metal is inexpensive, the contribution of the catalyst to the total COGS is negligible compared to Pd-catalyzed routes. Furthermore, the simplified one-pot nature of the reaction reduces labor hours and utility consumption, as there is no need for extended heating cycles or complex inert atmosphere setups beyond oxygen sparging. The ability to use acetone, a low-cost, high-volume solvent, instead of specialized anhydrous ethers or chlorinated solvents further drives down the solvent recovery and disposal costs, contributing to substantial overall economic efficiency.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain resilience by reducing the dependency on specialized reagents that may have long lead times. Ferrous chloride and acetone are available from multiple global suppliers, preventing single-source bottlenecks. The mild reaction conditions also mean that the process can be transferred between manufacturing sites with minimal re-validation of thermal safety parameters, facilitating a more agile supply network. This flexibility allows for decentralized production or rapid scaling at CDMO partners who may not have specialized high-pressure or high-temperature reactors, ensuring that production schedules are met even during periods of high demand.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but advancements in flow chemistry and thin-film reactors make this technology highly scalable for industrial production. The green nature of the process, producing water as the primary byproduct and avoiding heavy metal waste, simplifies environmental permitting and wastewater treatment. This alignment with ESG (Environmental, Social, and Governance) goals is increasingly important for pharmaceutical companies aiming to reduce their carbon footprint. The process avoids the generation of halogenated waste or heavy metal sludge, significantly lowering the cost and complexity of waste disposal and ensuring compliance with increasingly strict environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines for oncology drug development.
Q: What are the primary advantages of using FeCl2 over Palladium catalysts for oxindole synthesis?
A: The use of ferrous chloride (FeCl2) eliminates the reliance on expensive precious metals like Palladium or Rhodium, significantly reducing raw material costs. Furthermore, iron is earth-abundant and less toxic, simplifying heavy metal removal processes and ensuring stricter compliance with pharmaceutical impurity guidelines.
Q: How does the visible-light mediated process impact reaction safety and scalability?
A: Operating under blue LED irradiation at ambient temperature avoids the high thermal energy inputs required by traditional condensation methods. This mild condition reduces the risk of thermal runaway and decomposition, making the process inherently safer and more adaptable to continuous flow manufacturing for large-scale production.
Q: Can this synthetic route accommodate diverse functional groups on the indole scaffold?
A: Yes, the patent demonstrates high substrate tolerance, successfully synthesizing derivatives with halogens (Br, I), alkoxy groups, and various N-substituents (alkyl, allyl, benzyl). This versatility allows for the rapid generation of diverse libraries for SAR studies without needing protective group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of monitoring trace metal levels to meet stringent purity specifications required for clinical and commercial API supply. We are committed to leveraging this green chemistry innovation to deliver cost-effective, high-quality 3-alkenyl oxindole derivatives that support the global demand for next-generation cancer therapeutics.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this iron-catalyzed process for your specific molecule. We encourage you to contact us to obtain specific COA data for our reference standards and to receive detailed route feasibility assessments that demonstrate how we can accelerate your development timelines while reducing overall manufacturing costs.
