Advanced Bisulfate-Catalyzed Synthesis of N-Alkoxyoxalyl Alanine Ester for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic viability, particularly for critical intermediates like N-alkoxyoxalyl alanine esters. Patent CN101830821B introduces a transformative chemical synthesis method that addresses long-standing inefficiencies in producing these valuable compounds, which serve as pivotal precursors for Vitamin B6 and various agrochemical applications. This innovation leverages a bisulfate-catalyzed esterification process that operates under moderate thermal conditions, specifically between 50°C and 150°C, utilizing readily available alanine and oxalic acid as starting materials. By replacing corrosive mineral acids and expensive organic bases with inexpensive inorganic bisulfates, this technology offers a pathway to significantly enhance process safety and reduce the total cost of ownership for manufacturers. The method achieves reaction yields exceeding 80% through a continuous water-removal strategy, ensuring that the equilibrium is driven decisively toward the desired product while minimizing the formation of difficult-to-separate impurities. For R&D directors and procurement specialists alike, this patent represents a critical opportunity to optimize supply chains for high-purity pharmaceutical intermediates through a greener, more economically sustainable manufacturing protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this bisulfate-catalyzed technique, the industrial synthesis of N-alkoxyoxalyl alanine esters was plagued by significant technical and economic hurdles that hindered efficient commercial scale-up. Traditional methods often relied on the use of concentrated hydrochloric acid as a catalyst, which necessitated the use of specialized corrosion-resistant equipment and generated substantial amounts of acidic wastewater requiring complex neutralization and treatment protocols. Alternative approaches utilized expensive starting materials such as alanine esters combined with organic base catalysts like triethylamine, which not only inflated raw material costs but also introduced challenges in removing residual amines from the final product stream. Furthermore, existing non-acidic conditions frequently resulted in the formation of alanine mono-esterification by-products, creating a complex impurity profile that demanded rigorous and yield-loss-inducing purification steps. These legacy processes were characterized by long reaction times, harsh operating environments, and inconsistent product quality, making them increasingly untenable in a modern regulatory landscape that demands both environmental compliance and cost efficiency.
The Novel Approach
The innovative methodology described in the patent data fundamentally reengineers the synthesis landscape by employing sodium or potassium bisulfate as a highly active, solid acid catalyst. This approach allows for the direct reaction of free alanine with oxalic acid in C1-C8 alcohols, bypassing the need for pre-esterified amino acid derivatives and eliminating the reliance on volatile organic bases. The core of this advancement lies in the integration of azeotropic distillation using water-carrying agents such as toluene or benzene, which continuously removes the water generated during esterification, thereby shifting the chemical equilibrium towards completion without requiring excessive reagent loads. As illustrated in the reaction scheme below, this process converts alanine (Formula II) directly into the target N-alkoxyoxalyl alanine ester (Formula I) with exceptional selectivity.
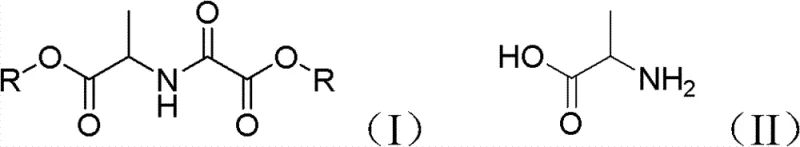
By maintaining reaction temperatures between 60°C and 100°C and optimizing the molar ratios of reactants, this novel route achieves high conversion rates while virtually eliminating the mono-esterification by-products that plague conventional methods. The result is a streamlined process that delivers high-purity products with simplified downstream processing, offering a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of both quality and manufacturability.
Mechanistic Insights into Bisulfate-Catalyzed Esterification
The efficacy of this synthesis route is rooted in the unique mechanistic properties of the bisulfate ion, which acts as a potent Brønsted acid catalyst to facilitate the nucleophilic attack of the alcohol on the carboxylic acid groups of oxalic acid and alanine. Unlike strong liquid mineral acids that can promote degradation or side reactions due to excessive acidity, the solid bisulfate provides a controlled acidic environment that activates the carbonyl groups for esterification while preserving the integrity of the amino acid backbone. The reaction mechanism involves the protonation of the carbonyl oxygen, increasing its electrophilicity and allowing the alcohol solvent to attack effectively, followed by the elimination of water molecules. Crucially, the presence of the water-carrying agent creates a heterogeneous azeotrope that allows for the physical removal of water from the reaction zone as it is formed, preventing the reverse hydrolysis reaction and driving the kinetics forward according to Le Chatelier's principle. This dynamic removal of by-product water is essential for achieving the high yields reported in the patent data, as it prevents the accumulation of water which would otherwise stall the reaction at an incomplete conversion stage.
From an impurity control perspective, the specific acidity and solubility profile of the bisulfate catalyst play a vital role in suppressing the formation of unwanted mono-esters. In conventional organic base-catalyzed routes, the basicity can lead to uneven acylation or transesterification issues, resulting in a complex mixture of species that are difficult to separate via standard crystallization or distillation. In contrast, the bisulfate system promotes a more uniform reaction progression where the di-esterification is favored thermodynamically and kinetically under the optimized conditions of 10 to 40 hours. The high selectivity ensures that the final crude product contains minimal levels of structural analogs, significantly reducing the burden on purification units and enhancing the overall mass balance of the process. This mechanistic precision is what allows manufacturers to consistently meet stringent purity specifications required for API intermediates, ensuring that the final material is suitable for sensitive downstream pharmaceutical synthesis without extensive reprocessing.
How to Synthesize N-Alkoxyoxalyl Alanine Ester Efficiently
The practical implementation of this synthesis route requires careful attention to reactor configuration and process parameters to maximize the benefits of the bisulfate catalysis system. Operators must utilize a reaction setup equipped with a rectifying column and a water separator to effectively manage the azeotropic removal of water, which is the driving force behind the high conversion rates. The process begins with the dissolution of alanine and oxalic acid in the chosen alcohol solvent, followed by the addition of the catalyst and the water-carrying agent, after which the mixture is heated to reflux. Detailed standardized synthesis steps, including specific molar ratios and temperature ramping profiles, are critical for reproducibility and are outlined in the technical guide below.
- Dissolve alanine and oxalic acid in a C1-C8 alcohol solvent within a reactor equipped with a rectifying column and water separator.
- Add sodium or potassium bisulfate catalyst and a water-carrying agent such as toluene or benzene to the mixture.
- Heat the reaction mixture to 60-100°C for 10-40 hours, continuously removing generated water via azeotropic distillation to drive equilibrium.
- Upon completion, wash the reaction liquid with water, separate the organic phase, dry with anhydrous sodium sulfate, and recover the product under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this bisulfate-catalyzed synthesis method presents a compelling value proposition centered on cost reduction in pharmaceutical intermediate manufacturing and enhanced operational stability. The shift away from corrosive hydrochloric acid and expensive organic amines to inexpensive, stable inorganic salts drastically simplifies the raw material sourcing strategy and reduces exposure to volatile pricing markets for specialty reagents. Furthermore, the elimination of severe corrosion risks extends the lifespan of standard stainless steel reactors, deferring capital expenditure on exotic alloy equipment and reducing maintenance downtime associated with acid damage. The simplified workup procedure, which involves basic washing and drying rather than complex neutralization and extraction sequences, translates directly into reduced labor hours and lower utility consumption per kilogram of product produced. These factors combine to create a manufacturing process that is not only chemically superior but also economically resilient, offering substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Cost Reduction in Manufacturing: The replacement of costly triethylamine and corrosion-prone hydrochloric acid with low-cost sodium or potassium bisulfate results in a dramatic decrease in direct material costs. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, improving the overall atom economy and reducing the cost per unit of the final active ingredient. The ability to recover and recycle the water-carrying agents and excess alcohol further contributes to a leaner cost structure, making the process highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as alanine, oxalic acid, and common alcohols as feedstocks, manufacturers can mitigate the risk of supply disruptions often associated with specialized or imported catalysts. The robustness of the reaction conditions allows for flexible production scheduling, as the process is less sensitive to minor fluctuations in temperature or mixing compared to more fragile catalytic systems. This reliability ensures consistent delivery timelines for downstream customers, strengthening the position of the manufacturer as a dependable partner in the global pharmaceutical supply network.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of hazardous volatile organic catalysts make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduction in acidic wastewater generation aligns with increasingly strict environmental regulations, lowering the costs associated with waste treatment and disposal. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for selection by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative performance data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement officers assessing the quality implications of the new method. The answers provided reflect the specific advantages of the bisulfate system over legacy technologies, focusing on yield, purity, and operational simplicity.
Q: What are the primary advantages of using bisulfate catalysts over traditional hydrochloric acid methods?
A: The use of solid bisulfate catalysts eliminates the severe equipment corrosion associated with concentrated hydrochloric acid, reduces hazardous waste generation, and simplifies the post-reaction neutralization process, leading to lower operational costs and higher product purity.
Q: How does this synthesis method control the formation of mono-esterification by-products?
A: The method utilizes precise molar ratios of alanine to oxalic acid and employs efficient azeotropic water removal. This drives the reaction equilibrium towards the desired di-ester product while the specific acidity of the bisulfate catalyst minimizes side reactions common with organic base catalysts.
Q: Is this process suitable for large-scale industrial production of Vitamin B6 intermediates?
A: Yes, the process operates at moderate temperatures (60-100°C) and uses readily available raw materials like alanine and oxalic acid. The robust nature of the catalyst and the straightforward workup procedure make it highly scalable for commercial manufacturing of key pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkoxyoxalyl Alanine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global fine chemical market. Our team of expert chemists has thoroughly analyzed the potential of the bisulfate-catalyzed method described in CN101830821B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity N-alkoxyoxalyl alanine ester that meets stringent purity specifications, leveraging our rigorous QC labs to ensure every batch conforms to the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific azeotropic distillation requirements of this process, guaranteeing consistent quality and supply continuity for your Vitamin B6 or agrochemical projects.
We invite you to collaborate with us to unlock the full commercial potential of this advanced synthesis technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
