Scalable Metal-Free Photocatalytic Synthesis of 3-Aminomethylated Quinoxalinones for Commercial Production
Scalable Metal-Free Photocatalytic Synthesis of 3-Aminomethylated Quinoxalinones for Commercial Production
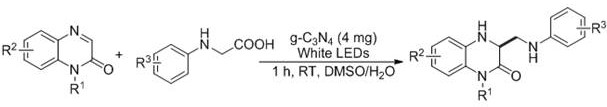
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes that minimize environmental impact while maximizing efficiency. A pivotal development in this domain is detailed in patent CN111689912A, which discloses a novel preparation method for 3,4-dihydro-2(1H)-quinoxalinone compounds. This technology leverages graphitic carbon nitride (g-C3N4) as a recyclable, heterogeneous photocatalyst to achieve C3-aminomethylation under mild visible-light irradiation. Unlike traditional methods that rely on harsh conditions, this approach utilizes a DMSO/H2O mixed solvent system without additional additives, representing a significant leap forward in green chemistry. For R&D directors and procurement specialists, this patent offers a compelling pathway to synthesize high-value heterocyclic intermediates with reduced operational complexity and enhanced safety profiles.
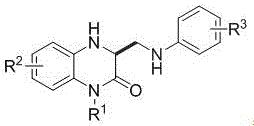
The core innovation lies in the ability to functionalize the C3 position of the quinoxalinone scaffold directly, a transformation that is crucial for accessing biologically active molecules with anticancer, antimicrobial, and anti-inflammatory properties. The structural versatility allowed by this method, accommodating various R1, R2, and R3 substituents such as methyl, allyl, propynyl, and halogens, ensures its applicability across a wide range of drug discovery programs. By establishing a reliable pharmaceutical intermediate supplier network based on this robust chemistry, manufacturers can secure a steady supply of critical building blocks while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C3-functionalized 2(1H)-quinoxalinone derivatives has been plagued by significant technical and economic hurdles that impede efficient commercial production. Traditional oxidative coupling reactions typically necessitate the use of stoichiometric amounts of expensive and hazardous oxidants, which generate substantial quantities of toxic waste streams requiring complex disposal protocols. Furthermore, these legacy processes often demand elevated temperatures and prolonged reaction times, leading to excessive energy consumption and potential thermal degradation of sensitive functional groups. The reliance on homogeneous transition metal catalysts in many conventional routes introduces another layer of difficulty, as removing trace metal residues to meet pharmaceutical purity standards requires costly scavenging steps and extensive chromatographic purification. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain.
The Novel Approach
In stark contrast, the methodology described in CN111689912A circumvents these challenges by employing a metal-free photocatalytic strategy that operates under ambient conditions. The use of g-C3N4, a stable and non-toxic polymeric semiconductor, eliminates the risk of heavy metal contamination entirely, thereby simplifying the downstream purification process and ensuring compliance with strict ICH guidelines for elemental impurities. The reaction proceeds efficiently at room temperature within just one hour under white LED irradiation, drastically reducing energy requirements compared to thermal methods. Additionally, the catalyst is heterogeneous, meaning it can be easily separated from the reaction mixture via filtration and potentially recycled, further enhancing the economic viability of the process. This streamlined workflow not only accelerates cost reduction in pharmaceutical intermediate manufacturing but also aligns perfectly with the principles of atom economy and waste minimization.
Mechanistic Insights into g-C3N4 Photocatalytic C-H Functionalization
The mechanistic foundation of this synthesis relies on the unique electronic properties of graphitic carbon nitride, which acts as a photosensitizer to drive the radical generation necessary for C-H bond activation. Upon absorption of visible light photons, the g-C3N4 catalyst undergoes excitation, promoting electrons from the valence band to the conduction band and generating electron-hole pairs. These photogenerated charge carriers facilitate the single-electron transfer (SET) oxidation of the N-arylglycine substrate, leading to the formation of an alpha-amino alkyl radical intermediate. This highly reactive species then attacks the electron-deficient C3 position of the 2(1H)-quinoxalinone ring, forging the new carbon-carbon bond with high regioselectivity. The entire catalytic cycle is sustained by the continuous regeneration of the catalyst surface, ensuring consistent turnover numbers without the degradation often seen with molecular photocatalysts.
From an impurity control perspective, the mildness of the radical mechanism prevents the formation of over-oxidized byproducts or polymerization side reactions that are common in harsh oxidative environments. The specific interaction between the catalyst surface and the substrates in the DMSO/H2O solvent system appears to stabilize the transition state, favoring the desired aminomethylation pathway. This precise control over the reaction trajectory results in cleaner crude reaction profiles, which translates directly to higher isolated yields and reduced solvent usage during purification. For process chemists, understanding this mechanism is vital for optimizing reaction parameters such as light intensity and catalyst loading to maximize throughput during commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Aminomethylated Quinoxalinones Efficiently
Implementing this photocatalytic protocol requires careful attention to the ratio of substrates and the specific configuration of the light source to ensure reproducible results. The standard procedure involves dissolving the 2(1H)-quinoxalinone and N-arylglycine starting materials in a 3:1 volume ratio of DMSO to water, creating a homogeneous medium that supports both organic solubility and catalyst dispersion. The addition of the g-C3N4 catalyst is critical, with the patent specifying a loading of approximately 20 grams per mole of substrate to maintain sufficient active surface area for photon absorption. Once the mixture is prepared, it is subjected to white LED irradiation with continuous stirring to keep the heterogeneous catalyst suspended and ensure uniform light exposure throughout the reaction vessel. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Combine 2(1H)-quinoxalinone and N-arylglycine substrates in a reaction vessel with a DMSO/H2O (3: 1) mixed solvent system.
- Add the heterogeneous g-C3N4 photocatalyst (20 g per mole of substrate) to the reaction mixture.
- Stir the mixture under white LED irradiation at room temperature for 1 hour, followed by extraction and purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this g-C3N4 mediated synthesis offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a major volatile cost component from the bill of materials, shielding the supply chain from fluctuations in the prices of palladium, rhodium, or iridium. Moreover, the simplified workup procedure, which avoids complex metal scavenging and extensive aqueous washes, significantly reduces the consumption of auxiliary chemicals and processing time. This operational efficiency allows for faster batch turnover rates, enabling manufacturers to respond more agilely to market demand fluctuations and reducing the inventory holding costs associated with long-cycle syntheses. The robustness of the method also implies a lower risk of batch failure, ensuring greater supply continuity for critical drug substances.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of expensive homogeneous catalysts with inexpensive, earth-abundant g-C3N4. By removing the need for transition metals, the process eliminates the capital and operational expenditures associated with metal removal technologies, such as specialized resin columns or activated carbon treatments. Furthermore, the ability to run the reaction at room temperature negates the need for heating or cooling infrastructure, leading to substantial savings in utility costs. The short reaction time of one hour maximizes reactor utilization, allowing existing facilities to produce significantly more material per annum without additional capital investment, thereby driving down the unit cost of the final intermediate.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically 2(1H)-quinoxalinones and N-arylglycines, are commercially available and structurally diverse, reducing the risk of single-source dependency. The use of a heterogeneous catalyst that can be potentially recovered and reused adds a layer of resilience to the supply chain by minimizing the consumption of consumable reagents. Additionally, the mild reaction conditions reduce the safety risks associated with high-pressure or high-temperature operations, lowering insurance premiums and minimizing the likelihood of unplanned shutdowns due to safety incidents. This stability is crucial for maintaining long-term contracts with global pharmaceutical partners who prioritize vendor reliability.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but the use of flow chemistry or optimized LED arrays can effectively address this for g-C3N4 systems. The process generates minimal hazardous waste, as the solvent system is relatively benign and the catalyst is non-toxic, simplifying wastewater treatment and disposal compliance. This environmental friendliness aligns with corporate sustainability goals and helps manufacturers avoid regulatory fines or restrictions related to VOC emissions and heavy metal discharge. Consequently, this technology facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology in an industrial setting. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses cover aspects ranging from catalyst handling to substrate compatibility, ensuring a comprehensive overview for decision-makers.
Q: What are the advantages of using g-C3N4 over traditional transition metal catalysts?
A: g-C3N4 is a metal-free, heterogeneous photocatalyst that eliminates the risk of heavy metal contamination in the final API, significantly reducing downstream purification costs and regulatory compliance burdens associated with residual metals.
Q: Can this photocatalytic method be scaled for industrial production?
A: Yes, the process operates at room temperature with short reaction times (1 hour) and uses inexpensive, recyclable catalysts, making it highly suitable for commercial scale-up without the need for high-pressure or high-temperature equipment.
Q: What is the substrate scope for this quinoxalinone synthesis?
A: The method demonstrates broad tolerance for various substituents including methyl, allyl, propynyl, ester, and benzyl groups on the quinoxalinone nitrogen, as well as electron-donating or withdrawing groups on the aryl ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-2(1H)-Quinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the g-C3N4 photocatalytic method for producing high-quality quinoxalinone derivatives. As a leading CDMO partner, we possess the technical expertise to adapt this laboratory-scale innovation into robust, large-scale manufacturing processes that meet the rigorous demands of the global pharmaceutical industry. Our facilities are equipped with advanced photochemical reactors and stringent QC labs capable of handling diverse synthetic pathways, ensuring that every batch meets stringent purity specifications. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to support your projects from early-stage development through to full commercial supply with unwavering consistency and quality assurance.
We invite you to collaborate with our technical team to explore how this green synthesis route can optimize your specific supply chain requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this technology can reduce your overall manufacturing expenses. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project, and let us help you secure a sustainable and cost-effective supply of these critical intermediates.
