Advanced Asymmetric Ligand Technology for Scalable Pharmaceutical Intermediate Production
Introduction to Next-Generation Asymmetric Catalysis
The landscape of chiral synthesis is undergoing a significant transformation driven by the need for more efficient and cost-effective catalytic systems. Patent CN101395165A introduces a groundbreaking class of asymmetric ligands that depart from the traditional reliance on the chiral pool, specifically sugar-derived scaffolds. This innovation presents a robust alternative for the production of high-value pharmaceutical intermediates, offering a streamlined synthetic pathway that addresses the inherent limitations of carbohydrate-based ligands. The core of this technology lies in a novel cyclohexyl-based phosphine oxide structure, which provides a rigid chiral environment essential for inducing high enantioselectivity in metal-catalyzed transformations.
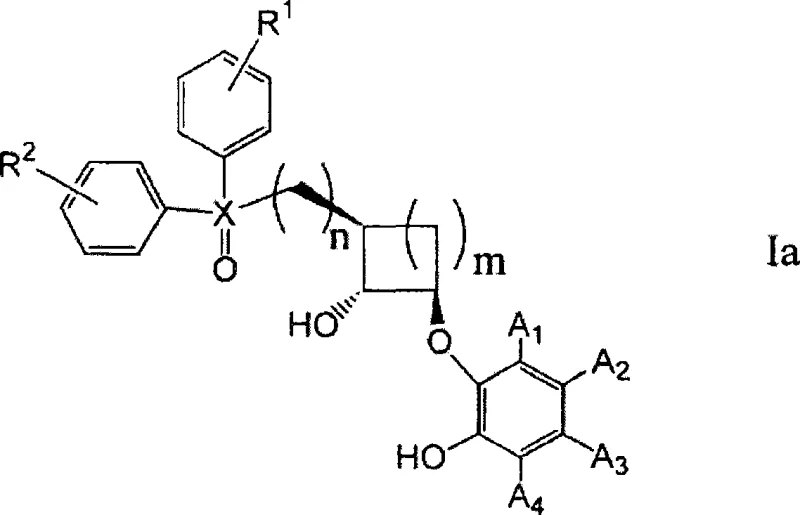
By leveraging this new structural motif, manufacturers can achieve superior catalytic activity and enantiomeric excess (ee) compared to existing state-of-the-art systems. The versatility of the ligand design allows for fine-tuning of electronic and steric properties through the variation of substituents on the aromatic rings, such as fluorine or chlorine atoms. This adaptability makes it a powerful tool for a reliable asymmetric ligand supplier aiming to serve diverse synthetic needs in the fine chemical and pharmaceutical sectors, ensuring consistent quality and performance across different reaction scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active compounds has heavily depended on ligands derived from natural chiral sources, particularly sugars. While effective, these sugar-derived ligands suffer from significant drawbacks that impact commercial viability. The primary issue is the complexity of their synthesis; they often require multistep sequences involving numerous protection and deprotection steps to mask reactive hydroxyl groups. This not only drives up the cost of goods sold (COGS) due to increased reagent consumption but also lowers the overall yield through cumulative losses at each stage. Furthermore, the supply chain for high-purity sugar derivatives can be volatile, subject to agricultural fluctuations and purification bottlenecks, creating risks for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The technology disclosed in CN101395165A offers a paradigm shift by utilizing a cyclohexane backbone that is readily accessible from petrochemical feedstocks. This approach eliminates the need for complex protecting group chemistry, drastically shortening the synthetic route. The novel ligand features a phosphine oxide moiety attached to a chiral cyclohexyl ring substituted with electron-deficient aromatic groups. This design enhances the Lewis acidity of the coordinated metal center, thereby accelerating reaction rates while maintaining strict stereocontrol. By simplifying the molecular architecture, the new method achieves higher atom economy and reduces waste generation, aligning with modern green chemistry principles and facilitating the commercial scale-up of complex pharmaceutical additives.
Mechanistic Insights into Gadolinium-Catalyzed Asymmetric Transformations
The efficacy of this ligand system is rooted in its ability to form stable and highly organized complexes with rare earth metals, particularly Gadolinium (Gd). When coordinated with Gd(OiPr)3, the ligand creates a chiral pocket that precisely orients the substrate for nucleophilic attack. In reactions such as the asymmetric ring-opening of aziridines or the cyano-Michael addition, the bulky phosphine oxide group and the fluorinated aromatic rings work synergistically to block one face of the substrate. This steric hindrance forces the incoming nucleophile to approach from the less hindered trajectory, resulting in the formation of a single enantiomer with exceptional purity.
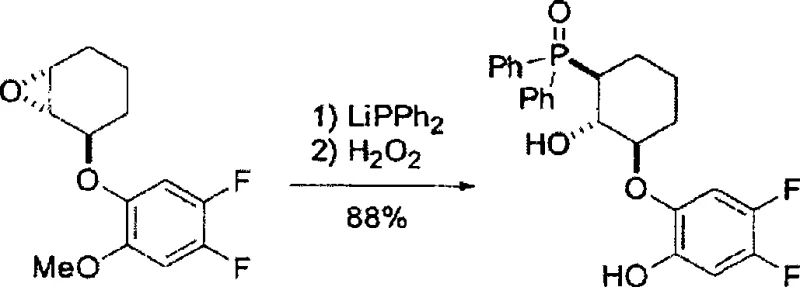
Furthermore, the electronic nature of the substituents plays a critical role in modulating the catalyst's activity. The presence of fluorine atoms on the aromatic ring increases the electrophilicity of the metal center, enhancing its ability to activate weakly nucleophilic species like trimethylsilyl cyanide (TMSCN). This mechanistic advantage allows the reaction to proceed under milder conditions, often at lower temperatures, which suppresses background racemic reactions. The result is a catalytic system that not only delivers high yields but also maintains optical purity levels exceeding 98% ee, a critical specification for regulatory compliance in API production.

How to Synthesize High-Purity Asymmetric Ligand Efficiently
The preparation of this advanced ligand follows a logical and scalable sequence that avoids the pitfalls of traditional chiral synthesis. The process begins with the stereoselective epoxidation of a cyclohexene derivative, establishing the foundational chirality. This is followed by a Mitsunobu etherification to attach the functionalized phenol, and finally, a phosphination-oxidation sequence to install the key phosphine oxide group. Each step is optimized for high yield and minimal purification requirements, making it suitable for industrial implementation. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Perform stereoselective epoxidation of cyclohexene derivatives using 3-chloroperbenzoic acid in a phosphate buffer system to establish the core chiral epoxide framework.
- Execute a Mitsunobu reaction between the chiral epoxide alcohol and substituted phenols using DIAD and triphenylphosphine to install the electron-deficient aromatic moiety.
- Conclude with nucleophilic ring-opening using lithium diphenylphosphide followed by oxidation with hydrogen peroxide to yield the final phosphine oxide ligand with >98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel ligand technology translates into tangible strategic benefits beyond mere technical performance. The shift from sugar-based to petrochemical-based chiral building blocks fundamentally alters the cost structure of ligand production. By removing the dependency on agricultural commodities and eliminating multiple synthetic steps, the manufacturing process becomes significantly more robust and predictable. This stability is crucial for long-term supply agreements and helps mitigate the risks associated with raw material price volatility, ensuring a steady flow of critical materials for downstream API synthesis.
- Cost Reduction in Manufacturing: The streamlined synthesis of the novel ligand inherently reduces production costs by minimizing reagent usage and solvent waste. Unlike sugar-derived ligands that require expensive protecting groups and harsh deprotection conditions, this cyclohexyl-based route utilizes straightforward transformations with high atom efficiency. The elimination of chromatographic purifications in favor of crystallization further drives down processing expenses. Consequently, this leads to substantial cost savings in the overall production of chiral intermediates, allowing pharmaceutical companies to optimize their margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing chiral ligands from renewable but fluctuating sugar markets can introduce uncertainty into the supply chain. In contrast, the precursors for this new ligand are derived from stable petrochemical streams, ensuring consistent availability and quality. The simplified synthetic route also means shorter lead times for manufacturing batches, enabling suppliers to respond more rapidly to demand spikes. This reliability is essential for maintaining continuous API production schedules and avoiding costly delays caused by material shortages or quality deviations in the upstream supply chain.
- Scalability and Environmental Compliance: The robustness of the synthetic pathway facilitates easy scale-up from laboratory to commercial tonnage. The reactions involved are exothermic but manageable, and the workup procedures are designed to minimize aqueous waste streams. By avoiding the use of heavy transition metals in the ligand synthesis itself (using rare earths only in catalytic amounts downstream), the process aligns better with environmental regulations regarding metal residues. This ease of scale-up ensures that the technology can meet the growing global demand for chiral drugs while adhering to increasingly stringent environmental, health, and safety (EHS) guidelines.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this catalytic system is vital for successful implementation. The following questions address common inquiries regarding the ligand's stability, compatibility, and application scope. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering a clear picture of what R&D teams can expect during process development and optimization phases.
Q: How does this novel ligand compare to traditional sugar-derived ligands in terms of synthesis cost?
A: Unlike sugar-derived ligands which require complex multi-step protection and deprotection sequences, this novel cyclohexyl-based ligand utilizes readily available petrochemical feedstocks, significantly reducing raw material costs and processing time.
Q: What specific metal catalysts are compatible with this ligand system?
A: The ligand is specifically designed to coordinate with rare earth metals, particularly Gadolinium (Gd) and Ytterbium (Yb), forming highly active catalytic species for asymmetric cyanation and ring-opening reactions.
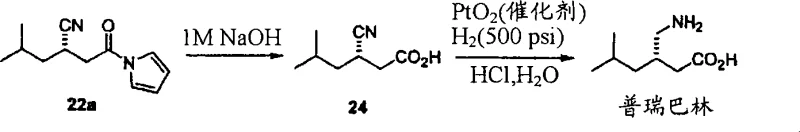
Q: Can this technology be applied to the synthesis of Pregabalin intermediates?
A: Yes, the patent explicitly demonstrates the utility of this catalyst system in the asymmetric cyano-Michael addition reaction, a key step in the efficient production of Pregabalin precursors with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Ligand Supplier
The potential of this novel asymmetric ligand technology to revolutionize the synthesis of chiral intermediates is immense, particularly for high-value targets like Pregabalin. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for GMP API synthesis.
We invite forward-thinking pharmaceutical partners to collaborate with us to leverage this cutting-edge catalytic technology. By integrating our Customized Cost-Saving Analysis into your development pipeline, we can identify specific opportunities to reduce your COGS and accelerate your time-to-market. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, quality, and mutual success.
