Advanced Cyclization Technology for High-Purity Lornoxicam Intermediates and Commercial Scalability
Advanced Cyclization Technology for High-Purity Lornoxicam Intermediates and Commercial Scalability
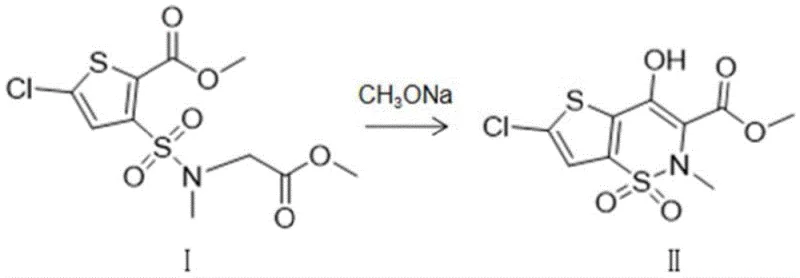
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic feasibility, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) like lornoxicam. A pivotal advancement in this domain is detailed in patent CN115894525A, which discloses a novel synthetic method for producing 6-chloro-4-hydroxy-2-methyl-2H-thieno[2,3-e]-1,2-thiazine-3-carboxylate-1,1-dioxide, a critical intermediate in lornoxicam manufacturing. This technology addresses long-standing challenges in cyclization efficiency by employing a specific dual-catalyst system comprising magnesium methoxide and sodium formate in a methanol solvent. Unlike traditional approaches that often suffer from incomplete conversion and complex impurity profiles, this innovation achieves exceptional reaction metrics, with reported yields surpassing 96% and product purity exceeding 99.9%. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-value anti-inflammatory agents through a process that is not only chemically superior but also inherently designed for safe, large-scale industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key thienothiazine dioxide scaffold has relied heavily on the use of sodium methoxide as a standalone base catalyst to drive the intramolecular cyclization of sulfonamide precursors. While conceptually straightforward, this conventional pathway is plagued by significant chemical inefficiencies that impact downstream processing and overall cost structures. The primary drawback lies in the aggressive nature of the reaction environment, which frequently induces the decomposition of the starting material into stubborn by-products, most notably 5-chloro-3-sulfinylthiophene-2-carboxylic acid derivatives. These impurities possess physicochemical properties remarkably similar to the target molecule, rendering their removal via standard crystallization or chromatography extremely difficult and costly. Furthermore, the conversion rates in these traditional processes are often suboptimal, leading to substantial losses of valuable raw materials and necessitating extensive recycling loops that increase energy consumption and extend production lead times, thereby creating bottlenecks for reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to the limitations of single-catalyst systems, the methodology outlined in CN115894525A introduces a synergistic catalytic regime that fundamentally alters the reaction landscape. By integrating magnesium methoxide with sodium formate, the process creates a buffered basic environment that promotes the desired cyclization while simultaneously suppressing deleterious side reactions. This dual-catalyst approach ensures that the starting material, 5-chloro-3-(N-methyl acetate-N-methylaminosulfonyl)-thiophene-2-formic acid methyl ester, undergoes rapid and complete transformation into the target cyclic structure. The result is a dramatic improvement in selectivity, effectively eliminating the formation of the problematic sulfinyl impurities that plague older methods. Consequently, the crude product obtained from this novel route is of such high quality that it requires only simple post-treatment steps, such as extraction and recrystallization, to achieve pharmaceutical-grade purity, streamlining the entire manufacturing workflow.
Mechanistic Insights into Magnesium Methoxide and Sodium Formate Catalyzed Cyclization
The success of this synthetic route can be attributed to the unique mechanistic roles played by the magnesium cation and the formate anion within the reaction matrix. Magnesium methoxide acts not merely as a base but potentially as a Lewis acid coordinator, stabilizing the transition state of the cyclization through interaction with the carbonyl and sulfonyl oxygen atoms of the substrate. This coordination lowers the activation energy required for the nucleophilic attack of the methyl group on the ester carbonyl, facilitating ring closure under milder thermal conditions. Simultaneously, the presence of sodium formate serves to moderate the basicity of the medium, preventing the harsh deprotonation events that typically lead to substrate degradation. This delicate balance allows the reaction to proceed efficiently at temperatures between 60°C and 65°C, a range that is sufficiently energetic to drive kinetics but low enough to prevent thermal decomposition, ensuring that the integrity of the sensitive thiophene ring system is maintained throughout the transformation.
From an impurity control perspective, the mechanism effectively shuts down the pathways leading to sulfinyl by-product generation. In conventional sodium methoxide reactions, the strong basicity can induce elimination or oxidation side reactions that generate sulfur-containing impurities which are notoriously difficult to separate. The buffered environment created by the formate salt mitigates these risks by maintaining a steady pH profile throughout the reaction duration of 6 to 7 hours. This stability is crucial for achieving the reported purity levels of greater than 99.9%, as observed in the chromatographic data where the target peak dominates the profile with minimal interference from adjacent impurity peaks. For process chemists, this implies a robust window of operation where minor fluctuations in temperature or addition rates do not catastrophically impact product quality, providing a safety margin that is essential for consistent commercial manufacturing.
How to Synthesize Lornoxicam Intermediate Efficiently
Implementing this advanced cyclization protocol requires precise adherence to the stoichiometric ratios and thermal profiles established in the patent examples to replicate the high yields and purity. The process begins with the preparation of a methanol solution containing the specific molar ratio of magnesium methoxide and sodium formate, typically ranging from 1.4 to 1.6 equivalents of the magnesium salt and 0.4 to 0.6 equivalents of the formate salt relative to the substrate. The sequential addition of reagents is critical to manage the exotherm and ensure homogeneous mixing before the substrate is introduced. Once the reaction mixture reaches the optimal temperature window of 60°C to 65°C, it is maintained for a defined period, usually around 6 to 7 hours, to guarantee full conversion as monitored by TLC or HPLC. Following the reaction, a controlled quench with hydrochloric acid at low temperatures precipitates the product or prepares it for extraction, followed by solvent removal and recrystallization to isolate the final high-purity solid. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by sequentially adding magnesium methoxide, sodium formate, and compound I into methanol solvent under controlled temperature conditions.
- Maintain the cyclization reaction at 60°C to 65°C for 6 to 7 hours to ensure complete conversion while minimizing thermal decomposition.
- Quench the reaction with hydrochloric acid at low temperature, followed by solvent removal, extraction with dichloromethane, and recrystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dual-catalyst synthesis method offers transformative benefits that extend far beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the downstream purification process. By virtually eliminating the formation of hard-to-remove sulfinyl impurities, manufacturers can bypass complex and expensive chromatographic separations or multiple recrystallization cycles that are often required in conventional routes. This reduction in processing steps translates directly into lower operational expenditures, reduced solvent consumption, and decreased waste generation, aligning perfectly with modern green chemistry initiatives and cost reduction strategies in pharmaceutical intermediates manufacturing. Furthermore, the use of common, inexpensive reagents like magnesium methoxide and sodium formate ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic or scarce catalysts.
- Cost Reduction in Manufacturing: The elimination of difficult purification stages significantly lowers the overall cost of goods sold (COGS) by reducing labor, energy, and solvent usage. Since the crude product purity is already exceptionally high, the need for extensive reprocessing is removed, allowing facilities to increase throughput without expanding physical infrastructure. This efficiency gain means that the same production assets can generate more saleable product in less time, effectively lowering the unit cost and improving margins for the final API manufacturer.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the moderate temperature range of 60°C to 65°C and the use of stable catalysts, minimizes the risk of batch failures due to thermal runaway or sensitivity issues. This reliability ensures consistent delivery schedules and reduces the likelihood of supply disruptions caused by out-of-specification batches. Additionally, the raw materials required are commodity chemicals with widespread availability, reducing dependency on single-source suppliers and enhancing the overall resilience of the supply network against geopolitical or logistical shocks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard reactor setups and common solvents like methanol which are easily recovered and recycled. The high selectivity of the reaction reduces the load on wastewater treatment systems by minimizing the discharge of complex organic by-products. This environmental advantage facilitates easier regulatory compliance and permitting for expansion, making it an ideal candidate for scaling up from pilot plant quantities to multi-ton commercial production volumes without encountering significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel lornoxicam intermediate synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear understanding of why this method outperforms traditional techniques. Understanding these nuances is vital for technical teams evaluating route feasibility and for procurement professionals negotiating supply contracts based on quality specifications. The answers below clarify the specific advantages of the magnesium-formate system and its impact on final product quality.
Q: Why is the magnesium methoxide and sodium formate system superior to traditional sodium methoxide?
A: The dual-catalyst system significantly suppresses the formation of 5-chloro-3-sulfinylthiophene-2-carboxylic acid by-products, resulting in yields exceeding 96% and purity greater than 99.9%, compared to lower yields and difficult purification in conventional methods.
Q: What are the critical process parameters for maintaining high selectivity?
A: Maintaining the reaction temperature strictly between 60°C and 65°C and controlling the molar ratio of magnesium methoxide to sodium formate to compound I (approx. 1.5:0.5:1) are essential for maximizing conversion and minimizing side reactions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, common solvents like methanol, and a simple work-up procedure involving extraction and recrystallization, making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lornoxicam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in patent CN115894525A can be reliably replicated on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of lornoxicam intermediate delivered meets the exacting standards required for global pharmaceutical registration. We understand the critical nature of API intermediates in the drug development timeline and are committed to providing a seamless, compliant, and efficient supply solution.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthetic technology for their lornoxicam projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-efficiency route for your specific volume requirements. We encourage you to contact us today to discuss your project needs,索取 specific COA data, and receive comprehensive route feasibility assessments that will empower your team to make informed decisions optimizing both cost and quality in your supply chain.
