Revolutionizing Patulin Detection with High-Purity Stable Isotope Labeled Standards
Revolutionizing Patulin Detection with High-Purity Stable Isotope Labeled Standards
The global demand for rigorous food safety testing has never been more critical, particularly concerning mycotoxins like patulin found in apple-based products. Patent CN113004300A introduces a groundbreaking total synthesis method for stable isotope labeled patulin, specifically patulin-13C7, which serves as an essential internal standard for Isotope Dilution Mass Spectrometry (IDMS). This technological breakthrough addresses the scarcity of high-quality reference materials, which have historically been dominated by foreign suppliers with opaque manufacturing processes. By utilizing glycerol-13C3 as a strategic starting material, this novel route ensures exceptional isotopic enrichment and chemical purity, meeting the stringent requirements of modern analytical laboratories. The ability to produce this complex molecule domestically represents a significant leap forward for supply chain security in the analytical reagent sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of patulin and its isotopologues relied heavily on methodologies described in older literature, such as Tetrahedron Letters, 1995, which presented significant operational hurdles for industrial scale-up. The conventional approach typically utilized a double-protection furan derivative that suffered from inherent structural instability, making the subsequent mountaineering reaction highly unreliable and prone to failure. Furthermore, the critical oxidation step converting hydroxyl groups to aldehydes required precise pH control at approximately 9, a condition that is notoriously difficult to maintain consistently in large-scale reactors. These technical bottlenecks not only resulted in low overall yields but also introduced variable impurity profiles that compromised the utility of the final product as a quantitative standard. The reliance on such fragile intermediates created a bottleneck for reliable procurement of high-purity analytical standards.
The Novel Approach
The methodology disclosed in patent CN113004300A fundamentally reengineers the synthetic pathway to bypass these historical limitations through a robust and linear sequence. Instead of unstable furan derivatives, the process initiates with the protection and oxidation of glycerol-13C3, creating a stable scaffold for subsequent carbon-carbon bond formations. A key innovation lies in the optimization of the hydroxyl oxidation step, which utilizes Dess-Martin periodinane under mild conditions, completely eliminating the need for difficult pH adjustments. This streamlined approach not only enhances the stability of key intermediates like compound 10 and compound 14 but also facilitates a smoother progression through the aldol condensation and final cyclization steps. The result is a reproducible process capable of delivering the target molecule with chemical purity and isotopic abundance both exceeding 98 percent.
Mechanistic Insights into Total Synthesis and Isotope Incorporation
The core of this synthetic strategy involves the precise assembly of three distinct 13C-labeled fragments to ensure uniform labeling across the patulin skeleton. The process begins with the conversion of glycerol-13C3 into a protected aldehyde equivalent, which is then subjected to a Wittig-type olefination using a phosphorus ylide derived from ethyl bromoacetate-13C2. This step is crucial for extending the carbon chain while maintaining the isotopic integrity of the backbone. Following this, the intermediate undergoes a series of protection and deprotection cycles, including silyl ether formation and pivaloyl esterification, to differentiate reactive sites for selective functionalization. The final ring closure is achieved through an intramolecular aldol condensation catalyzed by DBU, which constructs the characteristic lactone ring of the patulin structure. Each step is meticulously optimized to minimize isotopic dilution and maximize the recovery of the heavy isotope.
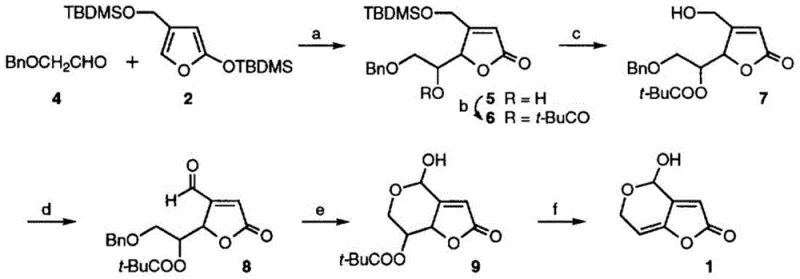
Impurity control is paramount in the production of analytical standards, and this patent employs several mechanisms to ensure a clean final profile. The use of column chromatography purification after nearly every major transformation step effectively removes side products and unreacted starting materials before they can propagate through the synthesis. Additionally, the selection of specific protecting groups, such as the tert-butyldimethylsilyl group and the pivaloyl group, provides orthogonal stability that prevents unwanted side reactions during the harsh conditions of the aldol condensation. The final debenzylation using boron trichloride is conducted at low temperatures (-80 to -75°C) to prevent degradation of the sensitive furan ring. This rigorous attention to reaction conditions and purification ensures that the final patulin-13C7 product meets the >98% purity specification required for accurate mass spectrometric quantification without interference from isotopic impurities.
How to Synthesize Patulin-13C7 Efficiently
The synthesis of patulin-13C7 is a complex multi-step organic transformation that requires precise control over stoichiometry, temperature, and reaction times to achieve the reported high yields. The process involves fifteen distinct operational steps, ranging from initial protection of glycerol to the final cyclization, each contributing to the overall efficiency of the route. Operators must adhere strictly to the specified molar ratios, such as the 1:2.0-2.2 ratio of glycerol to triphenylchloromethane in the first step, to ensure complete conversion. While the detailed reaction conditions are proprietary to the patent holder, the general workflow emphasizes the importance of anhydrous conditions and inert atmosphere protection, particularly during the lithiation and oxidation phases. For a complete breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Protection and Oxidation: React glycerol-13C3 with triphenylchloromethane and DMAP, followed by oxidation with pyridinium dichromate to form the protected intermediate.
- Ylide Reaction and Coupling: Generate the phosphorus ylide from ethyl bromoacetate-13C2 and couple it with the furan derivative under controlled thermal conditions.
- Cyclization and Deprotection: Perform aldol condensation using DBU and final deprotection with boron trichloride to yield the target 13C7-patulin with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to improved supply continuity and reduced risk of production delays. By eliminating the unstable intermediates that plagued previous methods, the new process ensures a more predictable production schedule, allowing suppliers to maintain consistent inventory levels of this critical reference material. Furthermore, the high yield and purity reported in the patent imply a significant reduction in raw material waste and downstream purification costs, which are key drivers in the final pricing of stable isotope standards. This efficiency gain enables manufacturers to offer more competitive pricing structures without compromising on the quality certifications required by regulated industries.
- Cost Reduction in Manufacturing: The elimination of complex pH control systems and the use of more robust intermediates significantly lowers the operational expenditure associated with production. By avoiding the failure rates associated with unstable double-protection furan derivatives, manufacturers can reduce the cost of goods sold through higher batch success rates. Additionally, the streamlined purification requirements reduce the consumption of expensive chromatography media and solvents, further driving down the unit cost. These cumulative efficiencies allow for a more sustainable economic model for producing high-value isotopic reagents.
- Enhanced Supply Chain Reliability: Sourcing stable isotope labeled compounds has historically been a challenge due to limited global suppliers and long lead times. This domestic synthesis capability breaks the monopoly of foreign providers, offering a secure and localized source for patulin-13C7. The robustness of the synthetic route means that production can be scaled up rapidly to meet surges in demand from food safety testing laboratories. This reliability is crucial for regulatory bodies and testing agencies that require uninterrupted access to certified reference materials to maintain accreditation and compliance.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents and reagents that are readily available in bulk quantities. The avoidance of hazardous pH adjustment steps and the optimization of reaction times contribute to a greener manufacturing footprint with reduced waste generation. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more resilient against tightening environmental regulations. Consequently, partners can secure a long-term supply of essential analytical tools that are both economically and environmentally sustainable.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of isotope labeled synthesis is essential for making informed procurement decisions. The following questions address common inquiries regarding the specifications, applications, and stability of the patulin-13C7 produced via this method. These answers are derived directly from the experimental data and claims presented in the underlying patent documentation, ensuring accuracy and relevance for technical stakeholders. For further clarification on custom synthesis capabilities or bulk ordering, our technical team is available to provide detailed consultations.
Q: What is the isotopic abundance of the synthesized patulin-13C7?
A: According to patent CN113004300A, the synthesized stable isotope labeled patulin achieves an isotopic abundance of greater than 98%, ensuring high accuracy for Isotope Dilution Mass Spectrometry (IDMS).
Q: Why is this new synthesis method superior to previous literature methods?
A: The new method overcomes the instability of double-protection furan derivatives found in prior art and simplifies the hydroxyl oxidation step, eliminating the need for difficult pH control around 9.
Q: What are the primary applications for this isotope standard?
A: This standard is primarily used for the accurate quantitative analysis of patulin contamination in rotten apples, apple juice, and other fruit products using Stable Isotope Dilution Mass Spectrometry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Patulin-13C7 Supplier
As the demand for precise food safety analytics grows, securing a partner with deep expertise in complex isotope synthesis is vital for your laboratory's success. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced synthetic methodologies like those described in patent CN113004300A to deliver uncompromising quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for R&D or routine QC. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that verify every batch for isotopic abundance and chemical integrity before release.
We invite you to collaborate with us to optimize your supply chain for analytical standards. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific consumption patterns and budget constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for patulin-13C7 and other specialized intermediates. Let us help you achieve greater accuracy and efficiency in your toxicology screening programs.
