Advanced One-Step Catalytic Synthesis of Benzofuran Intermediates for Commercial Scale-Up
Advanced One-Step Catalytic Synthesis of Benzofuran Intermediates for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable synthetic routes for complex heterocyclic intermediates. A significant breakthrough in this domain is documented in patent CN111747919B, which details a novel direct preparation method for 5-hydroxy-2,2-dimethyl-2H-benzofuran-6-formaldehyde. This compound serves as a critical building block for synthesizing biologically active molecules, including rotenone analogues like deguelin and tephrosin, which have shown promise in cancer research. The patented technology addresses long-standing inefficiencies in traditional synthesis by introducing a streamlined, one-pot catalytic process that utilizes readily available starting materials. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-hydroxy-2,2-dimethyl-2H-benzofuran-6-formaldehyde has been plagued by significant logistical and chemical challenges. Conventional routes typically rely on hazardous or unstable precursors such as 3-chloro-3-methylbutyne or 3-methyl-2-butyraldehyde. These reagents are not only expensive but also require stringent low-temperature storage conditions to prevent oxidation and degradation, complicating supply chain management. Furthermore, traditional methods often involve a multi-step sequence where an intermediate compound, specifically 2-hydroxy-4-(1,1-dimethyl-propargyloxy)-benzaldehyde, must be isolated before undergoing further rearrangement.
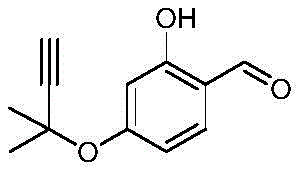
This necessity for intermediate isolation drastically increases processing time, solvent consumption, and waste generation. The operational complexity of separating and purifying intermediates creates bottlenecks in production throughput, making these legacy methods ill-suited for the demands of modern commercial scale-up. Additionally, the reliance on chlorinated alkynes introduces safety concerns regarding toxicity and corrosion, further elevating the operational costs for manufacturers aiming to produce reliable pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111747919B offers a transformative solution by enabling a direct, one-step synthesis. The core innovation lies in the use of 2-methyl-3-butyne-2-ol as a propargylating agent, reacting directly with 4-hydroxy-salicylaldehyde. This reagent is significantly cheaper, more stable at room temperature, and easier to source globally compared to its predecessors. The reaction proceeds via a tandem sequence of propargylation and cyclization, effectively bypassing the need for intermediate isolation.
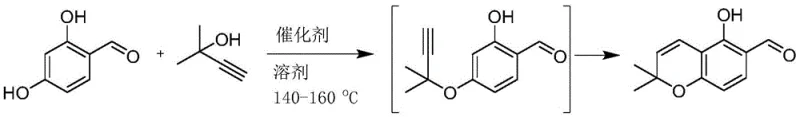
As illustrated in the reaction scheme, the process converts the starting materials directly into the target benzofuran aldehyde under reflux conditions. This telescoping of reactions not only simplifies the workflow but also enhances the overall atom economy. By eliminating the isolation step, the new approach reduces solvent usage and labor hours, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The ability to achieve high conversion rates in a single vessel makes this route exceptionally attractive for facilities looking to optimize their production lines for complex heterocycles.
Mechanistic Insights into Phosphine Oxide-Catalyzed Cyclization
The success of this direct synthesis hinges on the precise selection of the catalyst system. The patent specifies the use of a specialized phosphine oxide catalyst, characterized by a general structure where aryl groups (Ar1, Ar2) are attached to the phosphorus center, and the R group is selected from phenyl or substituted phenyl moieties. This specific electronic and steric environment is crucial for activating the hydroxyl group of the salicylaldehyde and facilitating the nucleophilic attack on the alkyne.
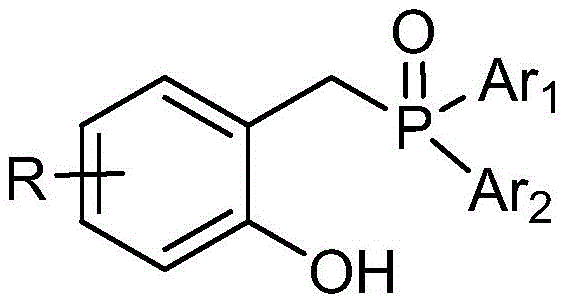
Mechanistically, the catalyst likely promotes the initial O-propargylation of the phenol, followed by an intramolecular cyclization to form the furan ring. The presence of the catalyst lowers the activation energy for these steps, allowing the reaction to proceed efficiently at temperatures between 140-160°C. The use of a Dean-Stark trap to continuously remove water drives the equilibrium forward, ensuring high yields without the need for excessive reagent loading. Understanding this catalytic cycle is vital for R&D teams, as it highlights the importance of catalyst purity and the specific substitution patterns on the phosphorus atom for achieving optimal selectivity and minimizing byproduct formation.
Furthermore, the choice of solvent plays a non-trivial role in the mechanism. High-boiling aromatic solvents like xylene, chlorobenzene, or anisole are preferred because they can sustain the necessary reflux temperatures while solubilizing both the polar phenolic substrate and the organic catalyst. The compatibility of these solvents with the phosphine oxide catalyst ensures a homogeneous reaction environment, which is essential for consistent batch-to-batch reproducibility. This level of mechanistic control allows for the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal impurity profiles, satisfying the rigorous quality standards of global regulatory bodies.
How to Synthesize 5-Hydroxy-2,2-Dimethyl-2H-Benzofuran-6-Formaldehyde Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins with the dissolution of the substrates and catalyst in the chosen solvent under an inert atmosphere. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring that technical teams can replicate the results effectively in a pilot or production setting.
- Dissolve 4-hydroxy-salicylaldehyde, 2-methyl-3-butyne-2-ol, and the phosphine oxide catalyst in an organic solvent such as xylene under nitrogen protection.
- Heat the mixture to reflux at 140-160°C using a Dean-Stark trap to remove water until the reaction is complete.
- Cool the reaction, wash the organic phase with aqueous sodium hydroxide and brine, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented route offers tangible strategic benefits beyond mere chemical efficiency. The shift from unstable, specialty reagents to commodity chemicals fundamentally alters the risk profile of the supply chain. By utilizing 2-methyl-3-butyne-2-ol, manufacturers can secure raw materials from a broader range of suppliers, reducing dependency on single-source vendors and mitigating the risk of supply disruptions.
- Cost Reduction in Manufacturing: The economic implications of this process are profound. By eliminating the intermediate isolation step, the facility saves significantly on solvent procurement, waste disposal, and energy consumption associated with multiple concentration and purification cycles. Furthermore, the replacement of expensive, low-temperature stored reagents with stable, ambient-temperature materials reduces cold-chain logistics costs. The simplified workflow also translates to lower labor costs, as fewer unit operations are required to reach the final product, thereby enhancing the overall profit margin for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Stability is a key factor in supply chain resilience. The new raw materials do not require specialized refrigerated transport or storage, simplifying inventory management and reducing the likelihood of spoilage during transit. This robustness ensures a continuous flow of materials to the production line, minimizing downtime. For global buyers, this means more predictable lead times and a more reliable pharmaceutical intermediate supplier capable of meeting tight delivery schedules without the complications of handling hazardous or unstable chemicals.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the one-pot nature of the reaction reduces the total volume of chemical waste generated. Fewer workup steps mean less aqueous waste and organic solvent residue to treat, aligning with increasingly strict environmental regulations. The process operates at atmospheric pressure with standard heating, avoiding the need for high-pressure reactors or cryogenic equipment. This simplicity facilitates easier scale-up from kilogram to tonne quantities, allowing manufacturers to rapidly respond to market demand while maintaining a smaller environmental footprint.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the process specifics and its applicability. These insights are drawn directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what to expect during technology transfer.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The new method utilizes cheap, stable raw materials like 2-methyl-3-butyne-2-ol instead of expensive, unstable reagents like 3-chloro-3-methylbutyne. It also eliminates the need for intermediate separation, reducing operational complexity.
Q: What specific catalyst is required for this transformation?
A: The process employs a specialized phosphine oxide catalyst where the aryl groups can be varied. This catalyst facilitates the direct cyclization and propargylation in a single pot.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common solvents like xylene, standard reflux conditions (140-160°C), and the elimination of sensitive low-temperature storage requirements make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Hydroxy-2,2-Dimethyl-2H-Benzofuran-6-Formaldehyde Supplier
The technological advancements detailed in CN111747919B underscore the potential for more efficient production of vital benzofuran derivatives. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative pathways for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-hydroxy-2,2-dimethyl-2H-benzofuran-6-formaldehyde meets the highest international standards.
We invite partners in the pharmaceutical and agrochemical sectors to collaborate with us to optimize their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
