Advanced Solid-Base Heck Reaction for Scalable Production of Acetyl-Substituted Nitrogen Heterocycles
Introduction to Patent CN101177412B: A Paradigm Shift in Heterocyclic Functionalization
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the functionalization of electron-deficient nitrogen-containing heterocycles, a class of structures pivotal to modern drug discovery. Patent CN101177412B introduces a groundbreaking synthetic protocol that addresses long-standing challenges in introducing acetyl groups onto pyridine, pyrimidine, and quinoline scaffolds. Unlike traditional electrophilic aromatic substitution which often fails on electron-poor rings, this invention leverages a modified Heck reaction mechanism facilitated by a unique solid-base catalytic system. By replacing conventional soluble organic amines and inorganic bases with a macroporous weakly basic styrene-based anion-exchange resin, the process achieves high conversion rates while drastically simplifying the workup procedure. This technological advancement represents a significant leap forward for any organization aiming to establish itself as a reliable pharmaceutical intermediates supplier, offering a pathway to high-purity products with minimized environmental footprint.
The core innovation lies in the synergistic combination of palladium acetate as the catalyst and the specific polymeric resin as the acid scavenger. This dual-system approach eliminates the need for toxic and expensive organic phosphine ligands, which are typically required to stabilize palladium species in standard cross-coupling reactions. The absence of these ligands not only reduces the raw material cost but also removes a major source of metallic impurities that are notoriously difficult to purge from final active pharmaceutical ingredients. For R&D directors evaluating process scalability, this patent offers a compelling solution that balances reaction efficiency with downstream processing ease, ensuring that the transition from laboratory bench to commercial production is seamless and economically viable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of acetyl groups onto nitrogen-containing heterocyclic rings has been fraught with synthetic difficulties. Traditional Friedel-Crafts acylation is generally ineffective for pyridine and related heterocycles due to the deactivating nature of the ring nitrogen atom, which complexes with Lewis acid catalysts and inhibits the electrophilic attack. Alternative strategies involving lithiation followed by reaction with acetylating agents require cryogenic conditions and pose significant safety risks on a large scale. Furthermore, previous iterations of Heck coupling for this transformation relied heavily on homogeneous catalytic systems employing soluble organic amines like triethylamine or inorganic bases such as potassium carbonate. These conventional bases generate stoichiometric amounts of salt waste, complicating the isolation of the product and creating substantial wastewater treatment burdens. Additionally, the reliance on phosphine ligands in older Heck protocols introduced issues regarding ligand oxidation, catalyst deactivation, and the presence of phosphorus impurities in the final product, necessitating costly purification steps.
The Novel Approach
The methodology described in CN101177412B circumvents these obstacles through a cleverly designed heterogeneous system. The reaction utilizes vinyl ether compounds as the acetyl source, which undergo Heck coupling with halogenated nitrogen heterocycles in the presence of palladium acetate. The defining feature of this novel approach is the utilization of a macroporous weakly basic anion-exchange resin, specifically of the styrene-DVB type (such as D301), to serve as the solid base. This resin effectively neutralizes the hydrogen halide byproduct generated during the coupling without dissolving into the reaction medium. 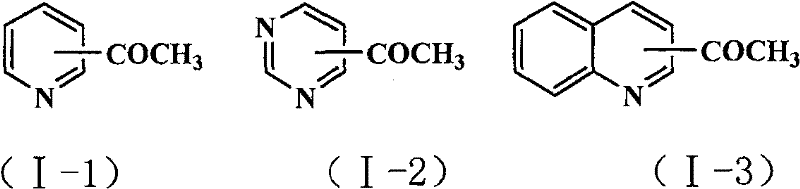 As illustrated in the reaction scheme, this allows for the direct formation of acetyl-substituted products like 2-acetylpyridine or 3-acetylquinoline with high selectivity. The solid nature of the base means it can be filtered off post-reaction, regenerating the catalyst system and allowing for potential reuse, which is a critical factor for cost reduction in pharmaceutical intermediates manufacturing. This approach transforms a complex, waste-generating process into a streamlined, green chemical synthesis suitable for modern industrial standards.
As illustrated in the reaction scheme, this allows for the direct formation of acetyl-substituted products like 2-acetylpyridine or 3-acetylquinoline with high selectivity. The solid nature of the base means it can be filtered off post-reaction, regenerating the catalyst system and allowing for potential reuse, which is a critical factor for cost reduction in pharmaceutical intermediates manufacturing. This approach transforms a complex, waste-generating process into a streamlined, green chemical synthesis suitable for modern industrial standards.
Mechanistic Insights into Solid-Base Catalyzed Heck Coupling
To fully appreciate the technical merit of this invention, one must understand the mechanistic nuances of the palladium cycle in the absence of phosphine ligands. Typically, palladium-catalyzed cross-couplings require electron-rich ligands to facilitate the oxidative addition of the aryl halide. However, in this specific system, the electron-deficient nature of the nitrogen heterocycle actually aids the oxidative addition step, reducing the dependency on auxiliary ligands. The vinyl ether coordinates to the palladium center, followed by migratory insertion and beta-hydride elimination to yield the enol ether intermediate. The crucial role of the macroporous resin comes into play during the regeneration of the active palladium species and the neutralization of the acid. The tertiary amine groups embedded within the polymer matrix of the resin act as proton sponges, trapping the HX byproduct. 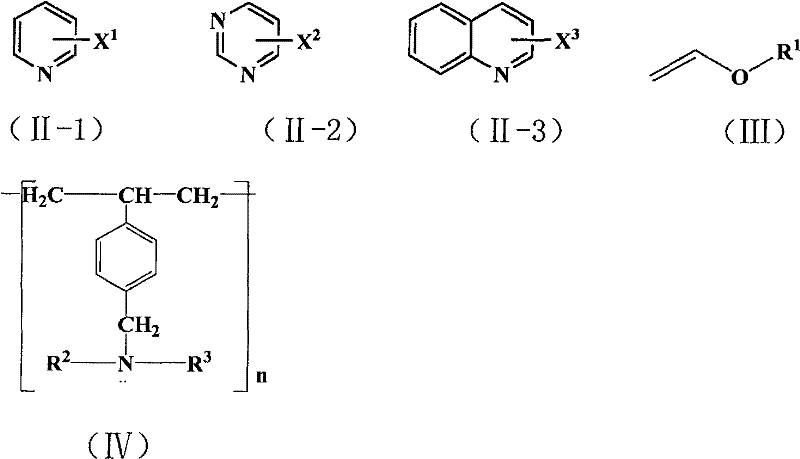 This interaction prevents the acidification of the reaction medium, which could otherwise protonate the heterocyclic nitrogen and deactivate the substrate towards further coupling. The structure of the resin, characterized by its styrene-divinylbenzene backbone and pendant amine functionality, provides a high surface area for this acid-base interaction while maintaining physical integrity under the reaction conditions of 60°C to 180°C.
This interaction prevents the acidification of the reaction medium, which could otherwise protonate the heterocyclic nitrogen and deactivate the substrate towards further coupling. The structure of the resin, characterized by its styrene-divinylbenzene backbone and pendant amine functionality, provides a high surface area for this acid-base interaction while maintaining physical integrity under the reaction conditions of 60°C to 180°C.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of phosphine ligands eliminates the formation of phosphine oxides, which are common contaminants that co-elute with polar heterocyclic products during chromatography. Furthermore, the use of a solid base minimizes the formation of emulsion during the aqueous workup, a frequent problem when using soluble amines that act as surfactants. The reaction proceeds through a clean hydrolysis of the enol ether intermediate upon acidification in the workup stage, converting the vinyl ether moiety directly into the acetyl group. This tandem Heck-hydrolysis sequence ensures that the final product is the desired ketone rather than a stable enol ether derivative, simplifying the synthetic route by combining bond formation and functional group transformation into a single operational pot. Such mechanistic elegance is vital for ensuring the high purity specifications required by stringent QC labs in the global supply chain.
How to Synthesize Acetyl-Substituted Nitrogen Heterocycles Efficiently
The practical implementation of this synthesis involves a straightforward protocol that is amenable to standard reactor setups found in most pilot and production plants. The process begins with the suspension of the halogenated heterocycle and vinyl ether in a polar aprotic solvent such as N-methylpyrrolidone or dimethyl sulfoxide. To this mixture, catalytic amounts of palladium acetate and a stoichiometric quantity of the macroporous resin are added. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for different substrates, are outlined in the guide below.
- Mix halogenated nitrogen heterocycle, vinyl ether, palladium acetate, and macroporous weakly basic styrene-based anion-exchange resin in an aprotic polar solvent.
- Stir and heat the mixture at 60°C to 180°C for 1 to 30 hours to facilitate the Heck coupling reaction.
- Acidify the reaction liquid to pH 1-3, separate the resin, extract with toluene, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational efficiencies and risk mitigation. The primary economic driver is the elimination of expensive and air-sensitive phosphine ligands, which often constitute a significant portion of the catalyst system cost in traditional Heck reactions. By utilizing simple palladium acetate and a commodity-grade ion-exchange resin, the raw material bill of materials is drastically simplified. Moreover, the recyclability of the resin base means that the consumption of base reagents is significantly reduced over multiple batches, leading to substantial cost savings in reagent procurement. The simplified workup procedure, which avoids complex extractions needed to remove soluble amine salts, reduces solvent consumption and labor hours, further driving down the cost of goods sold.
- Cost Reduction in Manufacturing: The removal of organic phosphine ligands and soluble amine bases creates a leaner manufacturing process. Without the need for specialized handling of air-sensitive ligands or the disposal of large volumes of amine salts, the overall production cost is optimized. The ability to recover and regenerate the ion-exchange resin adds a layer of circular economy to the process, ensuring that the cost per kilogram of the final intermediate decreases as batch cycles increase. This economic model supports competitive pricing strategies for high-purity pharmaceutical intermediates in a crowded global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available, off-the-shelf reagents such as palladium acetate and standard macroporous resins mitigates supply chain risks associated with specialty chemicals. Unlike custom-synthesized ligands that may have long lead times or single-source dependencies, the inputs for this process are widely sourced. This diversity in supply sources ensures continuity of supply even during market fluctuations. Additionally, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond rapidly to fluctuating demand from downstream API manufacturers without compromising quality.
- Scalability and Environmental Compliance: Scaling a heterogeneous catalytic process is inherently safer and more predictable than scaling homogeneous systems prone to exotherms and mixing issues. The solid resin acts as a buffer, and the reaction can be run at elevated temperatures without the volatility concerns of low-boiling amine bases. From an environmental standpoint, the reduction in waste salt generation and the avoidance of phosphorus-containing effluents align with increasingly strict environmental regulations. This compliance reduces the burden on wastewater treatment facilities and minimizes the risk of regulatory shutdowns, ensuring a stable and sustainable supply of critical chemical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-base Heck reaction technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for their own production pipelines. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the advantages of using macroporous resin over traditional amine bases?
A: The macroporous weakly basic styrene-based anion-exchange resin acts as a solid base that is easily separable and recyclable, unlike soluble organic amines which generate difficult-to-remove salt waste and complicate downstream purification.
Q: Does this method require expensive phosphine ligands?
A: No, the patented process operates effectively without organic phosphine ligands, utilizing palladium acetate directly, which significantly reduces raw material costs and eliminates phosphine oxide impurities.
Q: What types of nitrogen heterocycles are compatible with this synthesis?
A: The method is highly versatile and supports halogenated pyridines, pyrimidines, and quinolines, allowing for the introduction of acetyl groups at various positions on the heterocyclic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetyl-Substituted Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our technical team has extensively analyzed the potential of the solid-base Heck reaction described in CN101177412B and possesses the expertise to implement this technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is executed with precision. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of acetyl-substituted heterocycles meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes innovation, sustainability, and reliability, positioning your organization for success in the competitive landscape of drug development.
