Advanced Metal-Free Difluoromethylthiolation: Scaling High-Purity Intermediates for Global Pharma
The introduction of fluorine-containing functional groups into organic molecules is a cornerstone strategy in modern medicinal chemistry, particularly for enhancing metabolic stability and lipophilicity. Patent CN109503436B, published in March 2021, presents a significant breakthrough in this domain by disclosing a novel class of difluoromethylthiolation reagents and their efficient application in synthesizing heterocyclic compounds. The difluoromethylthio group (-SCF2H) possesses unique physicochemical properties, bridging the gap between the highly lipophilic trifluoromethylthio group and the hydrogen-bonding capability of the difluoromethyl group. As illustrated in the structural diversity of bioactive molecules below, this moiety is increasingly prevalent in high-value pharmaceutical candidates.
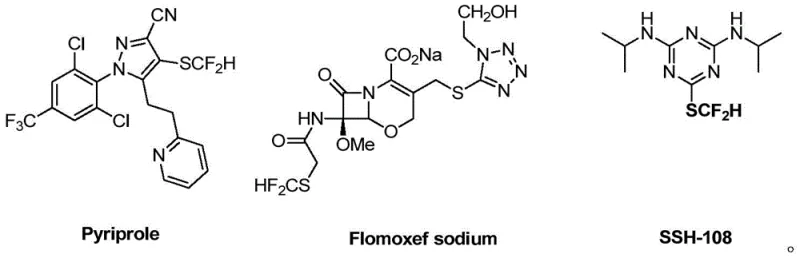
This patent specifically details the preparation of difluoromethylsulfinic acid methyl ester and ethyl ester, which serve as stable, easy-to-handle precursors for the direct introduction of the -SCF2H motif. For R&D directors and procurement specialists seeking a reliable difluoromethylthiolation reagent supplier, understanding the mechanistic advantages and supply chain implications of this technology is critical for optimizing the synthesis of next-generation agrochemical and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the installation of the difluoromethylthio group has been fraught with synthetic challenges that hinder commercial scalability. Early methodologies relied heavily on indirect alkylation strategies or the use of highly specialized, often unstable reagents. As depicted in the comparative analysis of existing reagents, traditional approaches frequently utilize complex organometallic species, such as silver-based complexes (e.g., [(SIPr)Ag(SCF2H)]) or multi-step synthesized silyl reagents.
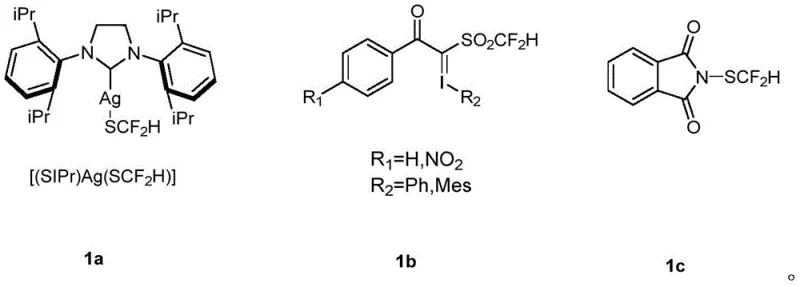
These conventional reagents suffer from significant drawbacks including high toxicity, thermal instability, and prohibitive costs associated with precious metal catalysts and ligands. Furthermore, the preparation of these reagents often requires more than four synthetic steps, leading to low overall atom economy and substantial waste generation. For a procurement manager, these factors translate into volatile supply chains and inflated raw material costs, making the consistent production of high-purity pharmaceutical intermediates economically unfeasible on a large scale.
The Novel Approach
In stark contrast, the technology disclosed in CN109503436B offers a paradigm shift by utilizing simple difluoromethylsulfinic acid esters (Formula I and II) as the sulfur source. This novel approach bypasses the need for transition metal catalysis entirely, relying instead on a chemically elegant reduction system composed of the sulfinate ester and an inexpensive alkylchlorosilane. The structural simplicity of these new reagents allows for their preparation in a single step from readily available difluoromethylsulfinyl chloride and common alcohols like methanol or ethanol.
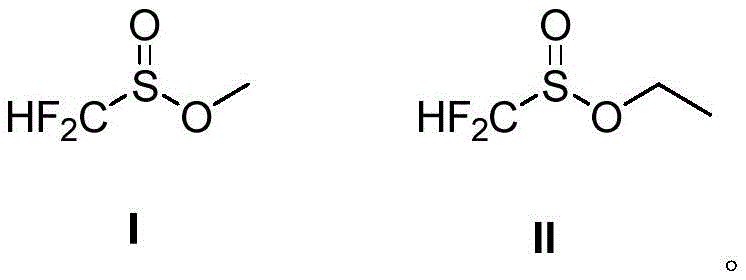
This streamlined synthesis not only drastically reduces the number of unit operations but also eliminates the environmental burden associated with heavy metal waste. By shifting from complex, multi-step reagent synthesis to a direct, one-pot esterification, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing while simultaneously improving the safety profile of the production facility. The robustness of these esters ensures long shelf-life and ease of transport, addressing key logistical pain points for global supply chains.
Mechanistic Insights into Metal-Free Difluoromethylthiolation
The core innovation of this patent lies in the generation of an active difluoromethylthio species in situ through a reductive activation mechanism. When the difluoromethyl sulfinate ester is mixed with an alkylchlorosilane (such as trimethylchlorosilane or tert-butyldimethylchlorosilane) in an organic solvent, a reactive intermediate containing the SCF2H+ equivalent is generated. This electrophilic species then attacks the electron-rich positions of heterocyclic substrates like indoles and pyrroles. The absence of external reducing agents or metal catalysts simplifies the reaction coordinate, minimizing the formation of side products that typically arise from metal-ligand dissociation or over-reduction.
From an impurity control perspective, this metal-free mechanism is exceptionally clean. Traditional metal-catalyzed cross-couplings often leave trace amounts of catalyst residues (e.g., Ag, Pd, Cu) that are difficult to remove and strictly regulated in final API products. By eliminating these metals from the reaction design, the downstream purification process is significantly simplified, often requiring only standard distillation and chromatography. This inherent purity advantage is crucial for meeting the stringent quality standards required for commercial scale-up of complex polymer additives and fine chemicals, ensuring that the final product specifications are consistently met without expensive scavenging steps.
How to Synthesize Difluoromethylthio Substituted Heterocycles Efficiently
The patent outlines a robust and scalable protocol for converting simple heterocycles into valuable difluoromethylthio derivatives. The process begins with the in-situ or pre-prepared formation of the sulfinate ester, followed by its reaction with the substrate in the presence of a silane promoter. This methodology is designed to be operationally simple, requiring standard glassware and common laboratory solvents, making it ideal for rapid process development and tech transfer.
- Preparation of Reagent: React difluoromethylsulfinyl chloride with methanol or ethanol at 60-80°C to generate the corresponding sulfinate ester.
- Reaction Setup: Combine the sulfinate ester solution, heterocyclic substrate (indole/pyrrole), and alkylchlorosilane (e.g., TMSCl) in an organic solvent like toluene.
- Execution & Purification: Stir the mixture at 20-80°C, then isolate the product via distillation and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this technology represents a strategic opportunity to optimize both cost structures and operational reliability. The shift away from precious metal catalysts and unstable reagents directly impacts the bottom line by reducing raw material expenditure and waste disposal costs. Furthermore, the mild reaction conditions enhance process safety, lowering insurance and compliance overheads associated with handling hazardous materials.
- Cost Reduction in Manufacturing: The elimination of expensive silver salts and complex ligands results in a dramatic decrease in reagent costs. Since the new sulfinate esters are prepared from commodity chemicals (methanol, ethanol, sulfinyl chloride), the cost of goods sold (COGS) is significantly lowered. Additionally, the simplified workup procedure reduces solvent consumption and labor hours, contributing to substantial overall process efficiency gains without compromising yield.
- Enhanced Supply Chain Reliability: Relying on exotic, custom-synthesized reagents often introduces single points of failure in the supply chain. By utilizing stable, shelf-stable esters that can be produced in bulk from widely available starting materials, manufacturers can secure a more resilient supply base. This stability ensures consistent availability of critical intermediates, reducing lead times and mitigating the risk of production stoppages due to reagent shortages.
- Scalability and Environmental Compliance: The metal-free nature of this reaction aligns perfectly with green chemistry principles and increasingly strict environmental regulations. The absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden for discharge permits. This facilitates easier commercial scale-up from pilot plant to multi-ton production, allowing companies to respond rapidly to market demand while maintaining a sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethylthiolation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear roadmap for potential adopters.
Q: What are the primary advantages of this new difluoromethylthiolation method over traditional silver-based reagents?
A: Unlike traditional methods requiring expensive silver complexes or unstable silyl reagents, this patent utilizes simple sulfinate esters and alkylchlorosilanes. This eliminates the need for costly transition metal catalysts and simplifies the purification process by avoiding heavy metal residues.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process operates under mild conditions (20-80°C) without the need for inert gas protection or cryogenic temperatures. The use of common solvents like toluene and stable liquid reagents makes it highly adaptable for kilogram-to-ton scale production.
Q: What types of substrates show the best reactivity with this reagent system?
A: The system demonstrates excellent compatibility with electron-rich heterocycles such as indoles and pyrroles. While electron-withdrawing groups may slightly reduce yields, the method remains effective across a broad range of substituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylthio Reagent Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free fluorination technologies in accelerating drug discovery and process optimization. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic protocols like CN109503436B are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our high-purity pharmaceutical intermediates meets the exacting standards of the global marketplace.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your synthesis route. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you secure a competitive advantage in the development of fluorine-containing therapeutics.
