Advanced Asymmetric Synthesis of (R)-2-Methyl-4-Nitro-1-Butanol for Commercial API Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing chiral building blocks, and patent CN103483201A presents a significant breakthrough in the production of (R)-2-methyl-4-nitro-1-butanol. This compound serves as a critical chiral intermediate in the synthesis of various biologically active molecules, yet its historical production has been plagued by inefficiencies inherent to racemic synthesis. The disclosed method introduces a sophisticated organocatalytic strategy that bypasses the traditional need for resolving racemic mixtures, thereby fundamentally altering the economic and operational landscape for manufacturers. By leveraging a chiral pyrrolidine derivative derived from abundant (S)-proline, the process achieves direct stereoselective construction of the carbon backbone. This technical advancement is not merely an academic exercise but represents a viable industrial solution that addresses the growing demand for high-purity chiral intermediates while adhering to stricter environmental and cost constraints. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating potential supply chain optimizations and licensing opportunities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-2-methyl-4-nitro-1-butanol and similar chiral nitro-alcohols relied heavily on non-stereoselective reactions followed by resolution. In these conventional pathways, chemical reactions typically generate a 50:50 mixture of enantiomers, known as a racemate. To isolate the desired (R)-isomer, manufacturers must employ resolution techniques such as chiral chromatography or diastereomeric salt formation. These methods are inherently inefficient because they theoretically discard at least half of the produced material—the unwanted (S)-isomer—unless a dynamic kinetic resolution is employed, which adds further complexity and cost. Furthermore, resolution processes often require stoichiometric amounts of expensive chiral resolving agents and generate substantial chemical waste, creating a heavy burden on waste treatment facilities. The cumulative effect is a process with low atom economy, high production costs, and extended lead times, making it increasingly uncompetitive in the modern fine chemical market where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the methodology outlined in CN103483201A employs a direct asymmetric synthesis strategy utilizing a chiral organocatalyst. The core innovation lies in the use of a silyl-protected (S)-alpha,alpha-diphenylprolinol derivative, which acts as a highly effective stereocontrolling agent. This catalyst facilitates the enantioselective reaction between acetaldehyde and nitroethylene, constructing the chiral center with high fidelity in a single synthetic operation. By avoiding the formation of the racemate entirely, the process theoretically doubles the yield relative to the starting materials compared to resolution-based methods. The patent details a streamlined sequence where the chiral information from inexpensive (S)-proline is transferred efficiently to the final product. This approach not only simplifies the purification workflow by eliminating resolution steps but also significantly reduces the consumption of raw materials and solvents. For a reliable API intermediate supplier, adopting such a route translates directly into enhanced process robustness and a smaller environmental footprint, aligning perfectly with green chemistry principles.
Mechanistic Insights into Organocatalytic Asymmetric Nitro-Aldol Reaction
The heart of this synthetic innovation is the organocatalytic cycle driven by the diphenylprolinol silyl ether. The mechanism typically involves the formation of an enamine intermediate between the catalyst and the aldehyde substrate (acetaldehyde). This enamine species is nucleophilic and attacks the electron-deficient nitroethylene in a stereocontrolled manner, dictated by the bulky diphenyl groups on the catalyst which shield one face of the reactive intermediate. The presence of 3-nitrobenzoic acid in the reaction mixture, as specified in the patent embodiments, likely serves as a proton shuttle or co-catalyst to accelerate the turnover rate and improve enantioselectivity. Following the carbon-carbon bond formation, hydrolysis releases the chiral nitro-aldehyde product and regenerates the free amine catalyst. This catalytic cycle is remarkably efficient, allowing for the use of substoichiometric amounts of the chiral auxiliary relative to the substrates in optimized conditions, although the patent describes preparing the catalyst in situ or as a distinct step. Understanding this mechanism is vital for process chemists aiming to scale the reaction, as factors like water content and temperature can critically influence the stability of the enamine and the subsequent stereochemical outcome.
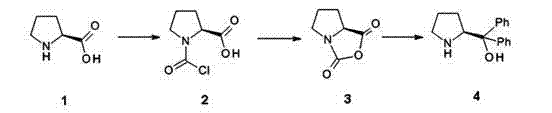
Furthermore, the preparation of the chiral catalyst itself is a critical unit operation that determines the overall success of the synthesis. As illustrated in the reaction scheme, the transformation begins with (S)-proline, which is activated using phosgene to form an N-carboxyanhydride intermediate. This reactive species is then subjected to a Grignard reaction with phenylmagnesium chloride to install the two phenyl groups, yielding the bulky (S)-alpha,alpha-diphenylprolinol. The subsequent protection of the hydroxyl group with trimethylsilyl trifluoromethanesulfonate (TMSOTF) generates the active silyl ether catalyst. Each of these steps requires precise control; for instance, the Grignard addition must be maintained at low temperatures (0 to -20°C) to prevent side reactions and ensure high optical purity. The patent emphasizes that the quality of this catalyst directly correlates with the enantiomeric excess of the final (R)-2-methyl-4-nitro-1-butanol. Impurities in the catalyst, such as unreacted proline or mono-phenylated byproducts, could act as poisons or lead to racemic background reactions, underscoring the need for rigorous quality control during the catalyst manufacturing phase.
How to Synthesize (R)-2-Methyl-4-Nitro-1-Butanol Efficiently
Executing this synthesis on a commercial scale requires a disciplined approach to reaction engineering and parameter control. The patent provides specific embodiments that serve as a robust starting point for process development, detailing molar ratios, solvent choices, and temperature profiles that have been empirically validated. The procedure generally involves the initial preparation of the chiral scaffold, followed by the key coupling reaction in a non-polar solvent like toluene to maximize stereoselectivity. Operators must pay close attention to the addition rates of reagents, particularly the nitroethylene and the reducing agent, to manage exotherms and maintain the integrity of the chiral centers. While the laboratory examples provide a clear roadmap, scaling up necessitates careful heat transfer management and potentially modified workup procedures to handle larger volumes of slurry and solvent. The detailed standardized synthesis steps below outline the critical operational parameters required to reproduce the high yields and purity reported in the intellectual property.
- Prepare the chiral catalyst (S)-alpha,alpha-diphenylprolinol by reacting (S)-proline with phosgene to form an N-carboxyanhydride, followed by Grignard reaction with phenylmagnesium chloride.
- Protect the hydroxyl group of the catalyst using trimethylsilyl trifluoromethanesulfonate (TMSOTF) and triethylamine to form the active silyl ether species.
- React the protected catalyst with acetaldehyde and nitroethylene in the presence of 3-nitrobenzoic acid at 0-5°C, followed by sodium borohydride reduction to yield the target chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN103483201A offers compelling strategic advantages beyond mere technical elegance. The most significant benefit is the drastic simplification of the manufacturing workflow. By eliminating the resolution step, the number of unit operations is reduced, which directly correlates to lower capital expenditure on equipment and reduced labor costs per kilogram of product. This streamlining also shortens the overall cycle time from raw material intake to finished goods, enhancing the agility of the supply chain to respond to market fluctuations. Moreover, the reliance on commodity chemicals like proline, acetaldehyde, and sodium borohydride ensures a stable and diversified supply base, mitigating the risks associated with sourcing exotic or single-source reagents. This resilience is crucial for maintaining continuity of supply for downstream API manufacturers who cannot afford interruptions.
- Cost Reduction in Manufacturing: The economic impact of switching to this asymmetric route is profound, primarily driven by the elimination of the 50% yield loss associated with racemic resolution. In traditional processing, half of the synthesized material is effectively waste unless complex recycling loops are established, which are often costly and inefficient. By synthesizing the desired enantiomer directly, the theoretical yield relative to the carbon source is doubled. Additionally, the removal of resolution agents and the associated solvents for crystallization or chromatography leads to substantial savings in raw material costs. The process also reduces waste disposal costs, as the volume of mother liquors containing the unwanted isomer is eliminated. These factors combine to create a significantly lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the use of robust, well-understood chemistry that does not depend on fragile biological enzymes or scarce transition metals. The reagents specified, such as (S)-proline and nitroethylene, are produced by multiple vendors globally, ensuring that supply disruptions are unlikely to halt production. The synthetic route is also less sensitive to minor variations in raw material quality compared to biocatalytic processes, offering greater operational flexibility. This robustness allows for the maintenance of safety stocks and consistent production schedules, which is vital for long-term contracts with pharmaceutical clients. Furthermore, the simplified process flow reduces the likelihood of batch failures due to operational complexity, ensuring a steady flow of high-purity product to customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this organocatalytic method aligns well with modern regulatory standards. The avoidance of heavy metal catalysts removes the need for expensive and technically challenging metal scavenging steps, which are often bottlenecks in API manufacturing. The solvents used, primarily toluene and THF, are easily recoverable and recyclable through standard distillation infrastructure, minimizing volatile organic compound (VOC) emissions. The high atom economy of the direct synthesis means less chemical waste is generated per unit of product, simplifying wastewater treatment and solid waste disposal. This environmental profile facilitates easier permitting for new production lines and supports the sustainability goals of multinational corporations, making the supplier a more attractive partner for green supply chain initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. They are derived from a detailed analysis of the patent specifications and practical considerations for industrial application. Understanding these aspects helps stakeholders assess the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the specific constraints and opportunities identified within the intellectual property documentation.
Q: What is the primary advantage of the method in CN103483201A over traditional routes?
A: The primary advantage is the direct asymmetric synthesis of the (R)-enantiomer, which completely eliminates the need for racemic resolution. Traditional methods produce a racemate requiring separation, inherently limiting maximum yield to 50% and increasing waste. This novel route achieves high enantioselectivity directly.
Q: What are the critical reaction conditions for the key coupling step?
A: The key coupling reaction between the silyl-protected catalyst, acetaldehyde, and nitroethylene requires strict temperature control between 0°C and 5°C. The presence of 3-nitrobenzoic acid as an additive is crucial for facilitating the reaction efficiency and maintaining stereocontrol.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials like (S)-proline and avoids expensive transition metal catalysts. The use of standard reagents such as sodium borohydride for reduction and common solvents like toluene and THF makes the process highly scalable and compliant with standard industrial safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-Methyl-4-Nitro-1-Butanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric synthesis technology detailed in CN103483201A for the production of high-value chiral intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We understand that consistency is key in the pharmaceutical supply chain; therefore, our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch. Whether you require custom synthesis services or a steady supply of this critical nitro-alcohol building block, our infrastructure is designed to meet the demanding requirements of global regulatory bodies.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthetic route for your specific applications. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a sustainable and competitive supply of high-purity (R)-2-methyl-4-nitro-1-butanol for your next generation of therapeutic agents.
