Advanced Biphasic Hydrolysis Technology for Commercial Sapropterin Manufacturing
Introduction to Next-Generation Sapropterin Synthesis
The pharmaceutical industry constantly seeks robust manufacturing processes that balance high purity with operational efficiency, particularly for complex molecules like tetrahydrobiopterin. Patent CN101959891A introduces a transformative approach to synthesizing sapropterin, also known as (6R)-tetrahydrobiopterin or BH4, by fundamentally reengineering the hydrolysis and hydrogenation stages. This technology addresses the critical bottlenecks of traditional synthesis routes, specifically the time-consuming isolation of solid intermediates which has historically constrained production throughput. By shifting to a biphasic liquid processing strategy, the method achieves a dramatic reduction in cycle time while simultaneously enhancing the stereochemical purity of the final active pharmaceutical ingredient. For R&D directors and supply chain leaders, this represents a pivotal opportunity to optimize the manufacturing of this essential cofactor used in treating phenylketonuria and other metabolic disorders.
The core innovation lies in the strategic manipulation of solubility properties during the hydrolysis of diacetylbiopterin. Instead of precipitating the intermediate biopterin as a solid, which requires extensive filtration and drying protocols, the new method retains the product in an aqueous phase while partitioning organic impurities into a separate immiscible organic layer. This liquid-liquid extraction principle not only accelerates the workflow but also inherently purifies the stream before the critical hydrogenation step. The patent data indicates that this modification can reduce the processing time for the hydrolysis and isolation sequence from an average of 265 hours down to merely 24 hours, a logistical breakthrough that significantly impacts facility utilization rates and overall cost structures for large-scale API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for sapropterin, such as those described in earlier patents like US 4,713,454, rely heavily on the isolation of L-biopterin as a solid intermediate. This approach presents severe inefficiencies when scaled to industrial levels, primarily due to the physical characteristics of the precipitated biopterin. The material tends to form fine granular particles that are notoriously difficult to filter, leading to prolonged processing times and potential product loss during the washing and drying phases. In a commercial setting aiming for metric ton production, the isolation step alone can consume approximately one-third of the total manufacturing timeline, creating a significant bottleneck that limits the number of batches a facility can produce annually. Furthermore, the extended exposure of the intermediate to acidic conditions during precipitation can lead to degradation, such as the elimination of hydroxyl groups from the side chain, thereby reducing the overall yield and complicating downstream purification efforts.
The Novel Approach
In stark contrast, the novel methodology detailed in the patent utilizes a biphasic reaction system comprising water and a water-immiscible organic solvent, such as n-butanol. By conducting the hydrolysis of diacetylbiopterin under alkaline conditions within this two-phase mixture, the desired biopterin product remains dissolved in the aqueous layer while the majority of organic by-products and unreacted starting materials partition into the organic phase. This eliminates the need for solid-liquid separation entirely at this stage, allowing for a seamless transition to the subsequent hydrogenation step. The process not only accelerates the timeline drastically but also improves the average yield of biopterin from roughly 60% in conventional methods to over 80% in this new system. This fluid processing capability enables the simultaneous preparation of multiple batches of biopterin solution, offering unparalleled flexibility and throughput for commercial manufacturing operations.
Mechanistic Insights into Biphasic Hydrolysis and Stereoselective Hydrogenation
The chemical efficacy of this process is rooted in the precise control of phase partitioning and catalytic stereoselectivity. During the hydrolysis phase, the pH is meticulously maintained between 11.5 and 12.2 using either inorganic bases like sodium hydroxide or organic amines such as diethylamine. This alkaline environment ensures rapid cleavage of the acetate esters while preventing the acid-catalyzed degradation pathways that plague older methods. The presence of the organic solvent is crucial; it acts as a scavenger for hydrophobic impurities, effectively cleaning the reaction stream in situ. As the reaction proceeds, the aqueous phase becomes enriched with high-purity biopterin, free from the dark organic contaminants that typically co-precipitate in single-phase systems. This inherent purification mechanism reduces the burden on downstream chromatography or recrystallization steps, directly contributing to the higher overall purity profiles observed in the final product.
Following hydrolysis, the aqueous stream undergoes stereoselective hydrogenation to establish the critical (6R) configuration. This step employs a platinum-based catalyst, such as platinum black or platinum oxide, under elevated hydrogen pressures ranging from 14 to 50 bar. The use of higher pressures and temperatures compared to historical methods significantly enhances the reaction kinetics, completing the hydrogenation in less than 23 hours rather than the 74 hours required by prior art. The basic conditions maintained during hydrogenation further promote the desired diastereoselectivity, achieving 6R:6S ratios exceeding 10:1. The mechanistic advantage here is twofold: the catalyst surface facilitates the specific addition of hydrogen to the pyrazine ring, while the alkaline medium stabilizes the transition state favoring the biologically active erythro-isomer. This combination of kinetic acceleration and thermodynamic control ensures that the manufacturing process delivers a product with the stringent stereochemical specifications required for pharmaceutical applications.
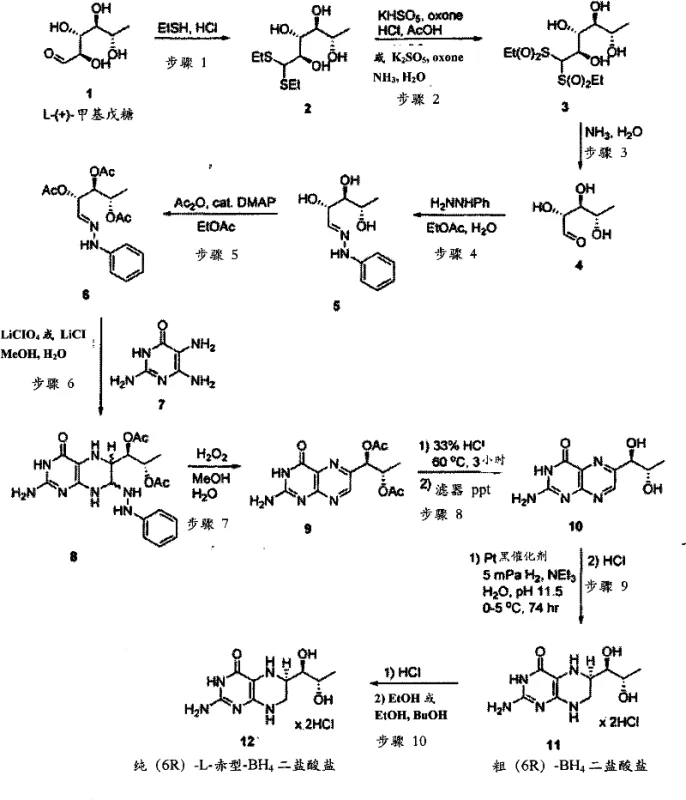
Furthermore, the management of ionic species plays a vital role in the final purification quality. Traditional methods often accumulate inorganic salts, such as sodium phosphates, which can interfere with the final crystallization of the dihydrochloride salt. The patented process offers a sophisticated solution by allowing the substitution of inorganic bases with volatile organic amines. When organic amines are used, the resulting salts are either soluble in the mother liquor or can be easily removed during the concentration steps, avoiding the contamination of the crystal lattice. Alternatively, if inorganic bases are employed, the process incorporates an ion exchange step to scrub residual cations from the solution prior to acidification. This attention to ionic purity ensures that the final crystallization yields a polymorphically stable product with minimal ash content, meeting the rigorous quality standards demanded by global regulatory bodies for API intermediates.
How to Synthesize Tetrahydrobiopterin Efficiently
Implementing this advanced synthesis route requires careful attention to phase separation dynamics and catalyst activation protocols to maximize yield and safety. The process begins with the suspension of diacetylbiopterin in a mixture of water and n-butanol, followed by the controlled addition of base to initiate hydrolysis. Once the conversion is complete, the phases are allowed to settle, and the aqueous layer is carefully decanted or separated for the next stage. The detailed standardized operating procedures for maintaining pH stability, managing exotherms during base addition, and optimizing catalyst loading are critical for reproducible results. For technical teams looking to adopt this methodology, understanding the interplay between solvent ratios and agitation speeds is essential to ensure efficient mass transfer between the phases. The following guide outlines the fundamental operational framework derived from the patent specifications to assist in process validation and scale-up activities.
- Hydrolyze diacetylbiopterin in a biphasic mixture of water and organic solvent under alkaline conditions to form biopterin.
- Separate the aqueous phase containing biopterin from the organic phase containing impurities without isolating the solid intermediate.
- Hydrogenate the aqueous biopterin solution using a platinum catalyst under controlled pH and pressure to yield tetrahydrobiopterin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biphasic synthesis technology translates into tangible strategic benefits beyond mere chemical yield. The most significant advantage is the drastic reduction in manufacturing cycle time, which directly enhances asset turnover and production capacity without the need for capital-intensive facility expansion. By eliminating the filtration and drying of the solid biopterin intermediate, the process removes a major bottleneck that previously dictated the pace of production. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, reducing the risk of stockouts for this critical therapeutic agent. Additionally, the simplified workflow reduces labor requirements and utility consumption associated with extended drying cycles and solid handling equipment, leading to a leaner and more cost-effective operational model.
- Cost Reduction in Manufacturing: The elimination of the solid isolation step significantly lowers operational expenditures by removing the need for extensive filtration, washing, and drying infrastructure. This reduction in unit operations decreases energy consumption and minimizes product loss associated with mechanical handling of fine powders. Furthermore, the higher overall yield of the process means that less raw material is required to produce the same amount of final API, directly improving the cost of goods sold. The ability to use organic amines which can be recovered or volatilized also reduces the waste disposal costs associated with inorganic salt by-products, contributing to a more sustainable and economically viable production profile.
- Enhanced Supply Chain Reliability: Shortening the production cycle from weeks to days dramatically improves the reliability of supply for downstream formulation partners. The reduced lead time allows for more frequent batch releases and tighter inventory control, mitigating the risks associated with long manufacturing queues. The robustness of the biphasic system, which tolerates variations in starting material purity better than precipitation methods, ensures consistent output quality even when sourcing raw materials from different suppliers. This resilience is crucial for maintaining continuous supply chains in the pharmaceutical sector, where interruptions can have severe consequences for patient access to life-saving medications.
- Scalability and Environmental Compliance: The liquid-phase nature of the key intermediate steps makes this process inherently easier to scale from pilot plant to multi-ton commercial production. Liquid handling is generally more amenable to automation and continuous processing technologies than solid filtration, facilitating smoother technology transfer. From an environmental perspective, the reduction in solvent usage for washing solids and the potential to minimize inorganic salt waste align with green chemistry principles. The process design supports compliance with increasingly stringent environmental regulations regarding effluent discharge, making it a future-proof choice for manufacturers aiming to reduce their ecological footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biphasic synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into existing manufacturing suites. The focus is on practical considerations such as catalyst recovery, solvent selection, and purity specifications that are critical for GMP compliance.
Q: How does the biphasic hydrolysis method improve production efficiency compared to conventional solid isolation?
A: Conventional methods require filtering and drying solid biopterin, taking up to 11 days. The biphasic method keeps biopterin in the aqueous phase, reducing this step to approximately 24 hours by eliminating solid handling.
Q: What catalyst is used to ensure high stereoselectivity in the hydrogenation step?
A: The process utilizes a platinum catalyst, such as platinum black or platinum oxide, under basic conditions and elevated hydrogen pressure to achieve a high 6R:6S diastereomeric ratio.
Q: How are inorganic salts managed to ensure high final product purity?
A: The method allows for the use of organic amines instead of inorganic bases to minimize salt formation, or employs ion exchange resins to remove residual salts before final crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrobiopterin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates like tetrahydrobiopterin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced patents like CN101959891A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international standards for API intermediates. Our commitment to process excellence means we can deliver high-purity sapropterin with consistent stereochemical profiles, supporting your drug development and commercial manufacturing needs with unwavering reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this biphasic technology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to enhance the efficiency and sustainability of your tetrahydrobiopterin supply chain, ensuring a steady flow of high-quality materials for your pharmaceutical formulations.
