Advanced Synthetic Route for Pyrrotinib Intermediate: Enhancing Efficiency and Scalability for Global Pharma
Advanced Synthetic Route for Pyrrotinib Intermediate: Enhancing Efficiency and Scalability for Global Pharma
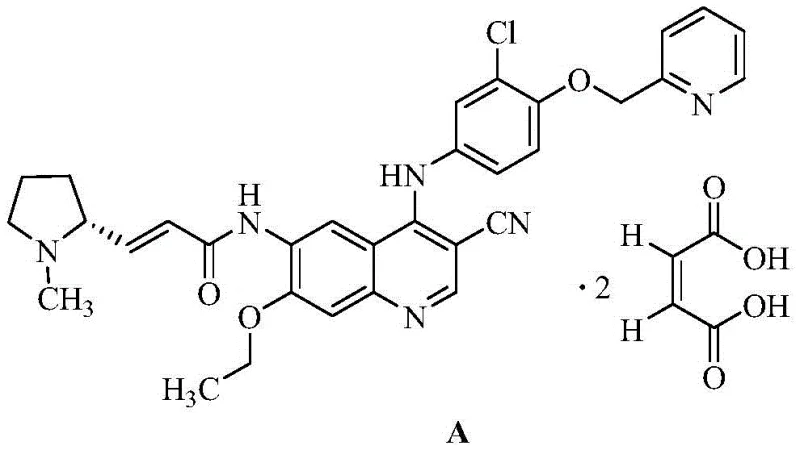
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for more efficient and cost-effective synthetic pathways for complex tyrosine kinase inhibitors. A pivotal development in this domain is documented in patent CN113336742B, which discloses a novel synthesis method for a key intermediate of Pyrrotinib maleate. Pyrrotinib, an oral, irreversible tyrosine kinase inhibitor targeting HER2, has established itself as a critical therapy for HER2-positive metastatic breast cancer. The efficiency of its production relies heavily on the availability of high-quality intermediates, specifically the quinoline derivative known as 6-amino-4-[[3-chloro-4-(2-pyridinylmethoxy)phenyl]amino]-7-ethoxy-3-quinolinecarbonitrile. This report analyzes the technical breakthroughs presented in the patent, offering strategic insights for R&D directors, procurement managers, and supply chain leaders seeking to optimize their API sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
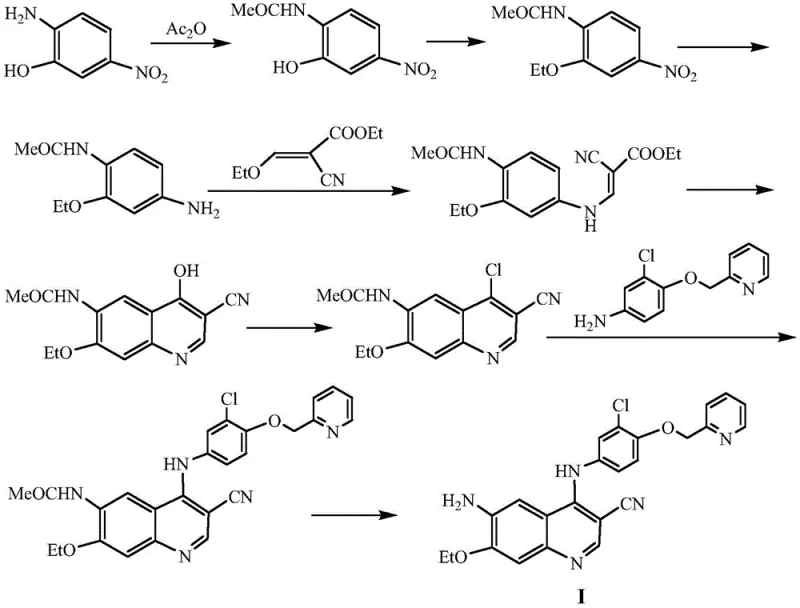
Prior to the innovations detailed in recent patents, the synthesis of the crucial quinoline intermediate for Pyrrotinib was fraught with significant operational and economic challenges. As illustrated in the legacy synthetic routes, the conventional process typically involves a cumbersome eight-step sequence starting from 5-nitro-2-aminophenol. This lengthy pathway not only accumulates impurities at every stage but also necessitates the use of hazardous and expensive reagents. For instance, traditional methods often rely on phosphorus oxychloride for chlorination, a reagent known to generate viscous byproducts that complicate purification and drastically reduce step yields to approximately 30%. Furthermore, the reduction steps in older protocols frequently require palladium on carbon (Pd/C) catalysts under high-pressure hydrogenation conditions, demanding specialized equipment and posing safety risks. The cumulative effect of these inefficiencies is a low overall yield, high production costs, and a supply chain vulnerable to bottlenecks, making it difficult for manufacturers to meet the growing global demand for this life-saving medication.
The Novel Approach
In stark contrast to the convoluted legacy processes, the methodology outlined in patent CN113336742B introduces a streamlined, three-step synthetic strategy that fundamentally reshapes the production landscape. This novel approach bypasses the need for high-pressure hydrogenation and hazardous chlorinating agents by leveraging a sophisticated sequence of amidation, Lewis acid-catalyzed cyclization, and hydrazine-mediated reduction. By condensing the synthetic timeline, the new route minimizes material loss and significantly enhances the purity profile of the final intermediate. The use of readily accessible starting materials, such as cyanoacetamide and substituted anilines, coupled with mild reaction conditions, allows for a drastic simplification of the operational workflow. This shift not only lowers the barrier to entry for manufacturing but also aligns with modern green chemistry principles by reducing waste generation and energy consumption. For pharmaceutical companies, adopting this route translates directly into a more robust and economically viable supply chain for Pyrrotinib intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Hydrazine Reduction
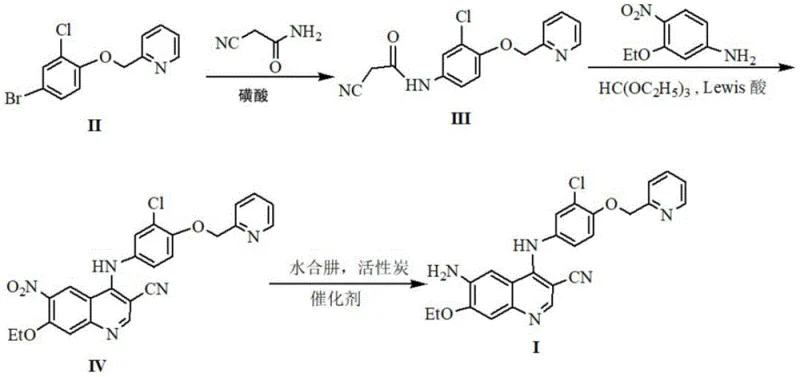
The core of this technological advancement lies in the precise orchestration of the cyclization and reduction steps. The formation of the quinoline core (Formula IV) is achieved through a condensation reaction between the amide intermediate (Formula III), 4-nitro-3-ethoxyaniline, and triethyl orthoformate, followed by a critical ring-closing step. This cyclization is facilitated by Lewis acids such as aluminum trichloride (AlCl3) or ferric trichloride (FeCl3) at temperatures ranging from 90°C to 140°C. The choice of Lewis acid is paramount; it activates the electrophilic centers necessary for ring closure while avoiding the side reactions typical of phosphorus-based chlorinating agents. Following the construction of the quinoline scaffold, the nitro group is selectively reduced to an amino group to yield the final intermediate (Formula I). Unlike catalytic hydrogenation, this process employs hydrazine hydrate in the presence of activated carbon and a dual-metal catalyst system (e.g., FeCl3·6H2O and AlCl3·6H2O). This specific catalytic combination ensures high selectivity and conversion rates at moderate temperatures (50-90°C), effectively preventing the over-reduction or degradation of sensitive functional groups within the complex molecule.
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The avoidance of phosphorus oxychloride eliminates the formation of phosphate ester byproducts and viscous tars that are notoriously difficult to remove. Similarly, the hydrazine reduction method circumvents the potential for dehalogenation—a common side reaction in Pd/C catalyzed hydrogenations where the chlorine atom on the phenyl ring might be inadvertently stripped. The patent data indicates that by optimizing the molar ratios of the catalysts and maintaining strict temperature controls during the exothermic hydrazine addition, manufacturers can achieve yields exceeding 80% in the final step. This level of control is essential for meeting the stringent purity specifications required for oncology APIs, ensuring that the final drug product is free from genotoxic impurities and heavy metal residues often associated with transition metal catalysts.
How to Synthesize Pyrrotinib Intermediate Efficiently
Implementing this novel synthetic route requires a clear understanding of the operational parameters defined in the patent to ensure reproducibility and safety. The process begins with the amidation of the bromo-substituted precursor with cyanoacetamide, followed by the crucial cyclization and reduction steps described above. Each stage demands precise stoichiometric control and thermal management to maximize yield and minimize byproduct formation. For R&D teams looking to transfer this technology to pilot or commercial scale, adhering to the specific solvent systems—such as isopropanol for amidation and diethylene glycol dimethyl ether for cyclization—is critical for solubility and reaction kinetics. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a foundational reference for process development.
- React compound II with cyanoacetamide using a sulfonic acid catalyst in isopropanol at 50-80°C to form the amide intermediate (Formula III).
- Condense Formula III with 4-nitro-3-ethoxyaniline and triethyl orthoformate, followed by ring closure using a Lewis acid (e.g., AlCl3) at 90-140°C to yield the nitro-quinoline (Formula IV).
- Reduce the nitro group of Formula IV to an amino group using hydrazine hydrate and activated carbon with a metal chloride catalyst system at 50-90°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN113336742B presents a compelling value proposition centered on cost efficiency and supply security. The primary driver of cost reduction in this manufacturing process is the elimination of expensive and scarce reagents. By replacing high-cost acrylates and noble metal catalysts like palladium with commodity chemicals such as cyanoacetamide and iron salts, the raw material cost base is significantly lowered. Furthermore, the reduction in synthetic steps from eight to three inherently reduces the consumption of solvents, energy, and labor hours per kilogram of output. This streamlined operation translates into substantial cost savings in API manufacturing, allowing pharmaceutical companies to improve their margins or pass savings on to healthcare systems without compromising quality.
- Cost Reduction in Manufacturing: The economic benefits of this route are multifaceted, extending beyond simple reagent costs. The removal of the high-pressure hydrogenation step eliminates the need for costly autoclaves and the associated safety infrastructure, representing a significant capital expenditure saving for manufacturing sites. Additionally, the higher overall yield resulting from fewer purification stages means less raw material is wasted, directly improving the cost of goods sold (COGS). The use of non-precious metal catalysts also simplifies the downstream processing, as there is no need for expensive and complex heavy metal scavenging procedures to meet regulatory limits, further driving down production expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of raw materials and the robustness of the manufacturing process. This novel route utilizes widely available starting materials that are not subject to the same supply constraints as specialized organometallic reagents. The simplified three-step process reduces the lead time for production batches, enabling manufacturers to respond more agilely to fluctuations in market demand. By mitigating the risks associated with complex, low-yield multi-step syntheses, suppliers can guarantee more consistent delivery schedules, ensuring that downstream drug formulation lines remain operational without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, but this route is designed with scalability in mind. The reaction conditions are mild and do not require extreme temperatures or pressures, making them easier to manage in large-scale reactors. From an environmental standpoint, the process generates less hazardous waste, particularly by avoiding phosphorus-containing byproducts and heavy metal sludge. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with waste disposal and environmental compliance, making it a sustainable choice for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Pyrrotinib intermediates. These answers are derived directly from the technical specifications and advantageous effects reported in the patent literature, providing clarity on the feasibility and benefits of this advanced manufacturing approach. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or strategic sourcing partnerships.
Q: Why is the new synthesis route superior to conventional methods for Pyrrotinib intermediates?
A: The new route described in CN113336742B reduces the synthesis from 8 steps to just 3 key transformations. It eliminates the need for expensive palladium catalysts, high-pressure hydrogenation equipment, and hazardous phosphorus oxychloride, resulting in a safer, more cost-effective, and higher-yielding process suitable for industrial scale-up.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization step utilizes a Lewis acid catalyst such as aluminum trichloride or ferric trichloride. The reaction is conducted in diethylene glycol dimethyl ether at elevated temperatures between 90°C and 140°C. This specific condition set ensures efficient ring closure to form the quinoline core without generating viscous byproducts common in traditional chlorination methods.
Q: How does this method improve supply chain reliability for oncology drug manufacturers?
A: By utilizing readily available raw materials like cyanoacetamide and avoiding complex, multi-step sequences with low overall yields, this method significantly shortens production lead times. The robust nature of the hydrazine reduction step also removes the dependency on specialized high-pressure hydrogenation facilities, ensuring more consistent and reliable batch-to-batch supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrotinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving oncology therapies. Our technical team has extensively analyzed the synthetic pathways described in CN113336742B and possesses the expertise to implement this optimized route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Pyrrotinib intermediate we deliver adheres to the highest international standards for pharmaceutical use.
We invite global pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage in the market through superior chemistry and unwavering supply chain commitment.
