Advanced Asymmetric Hydrogenation for Scalable Production of Critical Carbapenem Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral centers, particularly for beta-lactam antibiotics like carbapenems. Patent CN111499538A introduces a groundbreaking preparation method for (2S,3R)-2-amidomethyl-3-hydroxybutyrate, a pivotal intermediate in the synthesis of 4-AA, which serves as the core scaffold for potent antibiotics such as meropenem and biapenem. This innovation leverages a divalent ruthenium complex coordinated with a series of C3*-Tunephos C2-axisymmetric bidentate phosphine ligands to facilitate an asymmetric reduction reaction under a hydrogen atmosphere. The disclosed technology addresses critical bottlenecks in existing synthetic routes by offering a catalytic system that combines high atom economy with exceptional green chemistry principles. By enabling the efficient transformation of ketone precursors into chiral hydroxy esters with unparalleled selectivity, this method provides a reliable pharmaceutical intermediate supplier pathway that meets the rigorous demands of modern drug manufacturing.
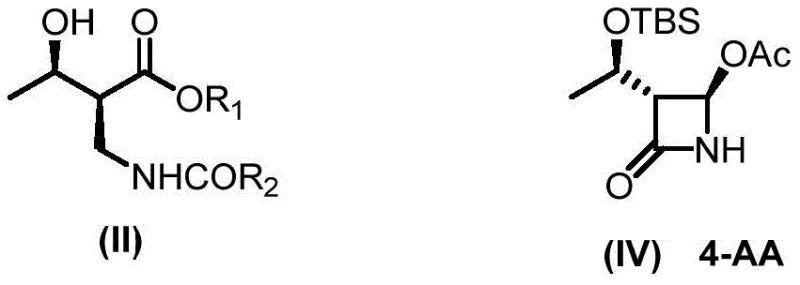
The synthesis of 4-AA has historically been plagued by structural complexity, involving three chiral centers and eight possible stereoisomers, which complicates isolation and purification. The present invention focuses on the asymmetric synthesis of the key intermediate designated as formula (II), specifically (2S,3R)-2-benzoylaminomethyl-3-hydroxybutyrate. By optimizing the catalytic environment and ligand architecture, the process achieves single selectivity and high production efficiency. This advancement is crucial for ensuring the supply continuity of high-purity pharmaceutical intermediates required for the global production of life-saving antibiotics, positioning this technology as a cornerstone for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
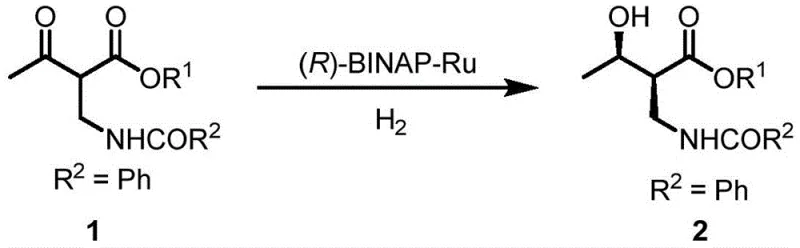
Historically, the industrial synthesis of this critical intermediate has relied on routes established by major chemical entities, such as the Takasago process, which utilizes ethyl acetoacetate as a starting material. The core step in this conventional approach involves the asymmetric reduction of 2-benzoylamino-3-carbonyl butyrate using a chiral (R)-BINAP-Ru catalyst. While this method can achieve stereoselectivity greater than 97% ee, it suffers from significant operational drawbacks that hinder its economic viability on a massive scale. Specifically, the requirement for a specially prepared catalyst containing triethylamine (NEt3) adds layers of complexity to the catalyst preparation and increases the overall production cost substantially. Furthermore, conventional methods often struggle to maintain high selectivity and conversion efficiency when the reaction is scaled up, leading to inconsistent batch quality and increased waste generation.
Additionally, existing literature and patents, such as CN 1931831A and CN103373934A, have attempted improvements but have failed to fully resolve the issues of catalyst loading and residual impurities. For bulk drug substance production, obtaining a near-single enantiomeric product with high ee values while minimizing catalyst content is paramount. The inability of traditional systems to consistently meet these dual requirements limits the industrial application of 4-AA synthesis. The reliance on expensive, additive-dependent catalysts not only inflates the cost of goods but also introduces potential contamination risks that require extensive downstream purification, thereby extending lead times and reducing overall process throughput.
The Novel Approach
In stark contrast to legacy technologies, the novel approach detailed in the patent employs a sophisticated catalytic system comprising a ruthenium catalyst and C3*-Tunephos series ligands. This system facilitates the direct and highly selective conversion of raw material (I) into the target compound (II) without the need for problematic amine additives. The reaction proceeds under hydrogen atmosphere with remarkable efficiency, demonstrating insensitivity to substituent changes on the substrate. Experimental data indicates that this method can consistently synthesize the target compound with up to 99% ee and 99% de, surpassing the selectivity benchmarks of previous generations. The simplicity of the catalyst preparation, combined with its robust performance, represents a paradigm shift in how complex chiral intermediates are manufactured commercially.
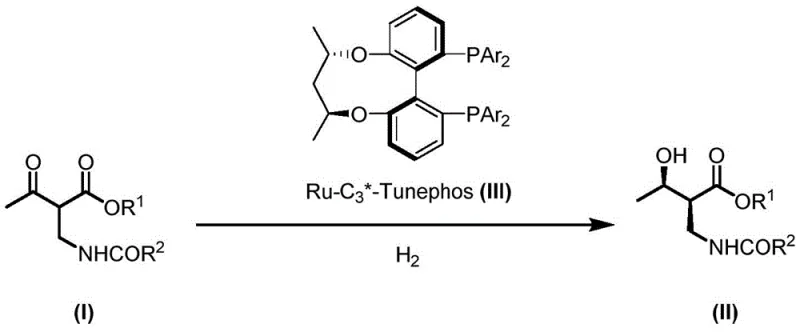
Furthermore, the novel catalytic system exhibits a turnover number (TON) as high as 5000, which signifies a drastic reduction in the amount of precious metal required per unit of product. This efficiency translates directly into lower raw material costs and reduced environmental burden associated with heavy metal waste. The process operates effectively across a range of solvents, including halogenated alkanes and alcohols, providing flexibility in process optimization. By eliminating the need for specialized catalyst formulations and achieving near-quantitative conversion, this approach offers a streamlined, cost-effective solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring a stable supply chain for critical antibiotic production.
Mechanistic Insights into Ru-C3*-Tunephos Catalyzed Asymmetric Hydrogenation
The success of this asymmetric hydrogenation lies in the precise molecular architecture of the C3*-Tunephos ligands, which are C2-axisymmetric bidentate phosphines. These ligands, characterized by their specific aryl substitutions (such as p-tolyl, 3,5-dimethylphenyl, or 3,5-di-tert-butylphenyl groups), create a highly defined chiral pocket around the ruthenium center. This steric environment dictates the approach of the hydrogen molecule and the substrate, ensuring that hydride transfer occurs exclusively to one face of the prochiral ketone. The rigidity of the ligand backbone prevents conformational flexibility that could lead to non-selective binding, thereby locking the transition state into a configuration that favors the formation of the (2S,3R) stereoisomer. This mechanistic precision is what allows the system to maintain high enantioselectivity even under varying reaction conditions.

Impurity control is another critical aspect governed by this mechanism. In traditional reductions, the presence of multiple stereoisomers necessitates complex crystallization or chromatographic steps to isolate the desired enantiomer. However, the high diastereoselectivity (up to 99% de) achieved by the Ru-C3*-Tunephos system ensures that the formation of unwanted diastereomers is suppressed at the source. This "prevention rather than cure" approach to impurity management significantly simplifies the workup procedure. The reaction mixture typically requires only a simple filtration through silica gel to remove the catalyst, followed by solvent evaporation, to yield the product in high purity. This reduction in processing steps not only lowers operational costs but also minimizes product loss, enhancing the overall yield and sustainability of the manufacturing process.
How to Synthesize (2S,3R)-2-benzoylaminomethyl-3-hydroxybutyrate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a production setting. The process begins with the in situ generation of the active catalytic species, followed by the hydrogenation reaction in a pressurized vessel. The detailed标准化 synthesis steps involve precise control over temperature, pressure, and solvent composition to maximize the turnover frequency and stereoselectivity. Operators must adhere to strict inert atmosphere conditions during catalyst preparation to prevent oxidation of the phosphine ligands, which could deactivate the catalyst. The subsequent hydrogenation step leverages the unique reactivity of the Ru-ligand complex to drive the reduction to completion with minimal byproduct formation.
- Preparation of the active ruthenium complex by mixing the ruthenium dimer and C3*-Tunephos ligand in a halogenated alkane and alcohol solvent mixture under argon protection, followed by heating and solvent removal.
- Charging the autoclave with the substrate, the pre-formed catalytic complex, and the reaction solvent, followed by purging with hydrogen gas to establish an inert and reactive atmosphere.
- Conducting the asymmetric hydrogenation reaction at elevated temperature and pressure (e.g., 80°C, 80 atm) for approximately 24 hours, followed by filtration to remove metal residues and isolation of the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalytic technology offers transformative benefits that extend beyond mere technical performance. The primary advantage lies in the substantial cost savings driven by the high efficiency of the catalyst system. With turnover numbers reaching 5000, the consumption of expensive ruthenium and chiral ligands is minimized, leading to a significant reduction in the variable cost per kilogram of the intermediate. Moreover, the elimination of auxiliary additives like triethylamine simplifies the bill of materials and reduces the logistical burden of sourcing and handling multiple specialized reagents. This streamlining of the supply chain enhances reliability and reduces the risk of production delays caused by material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the drastic simplification of the catalyst system and the high atom economy of the reaction. By removing the need for costly amine additives and reducing the catalyst loading through high TON values, manufacturers can achieve a leaner cost structure. The simplified workup procedure, which avoids complex purification steps, further reduces utility consumption and labor costs associated with downstream processing. These factors combine to deliver a highly competitive cost profile for the production of high-purity pharmaceutical intermediates, allowing for better margin management in a price-sensitive market.
- Enhanced Supply Chain Reliability: The robustness of the Ru-C3*-Tunephos system ensures consistent output quality regardless of minor fluctuations in raw material quality or reaction conditions. This reliability is crucial for maintaining uninterrupted supply lines to API manufacturers. The insensitivity of the catalyst to substrate substituents means that the same protocol can be adapted for various analogues without extensive re-optimization, providing flexibility in product portfolios. Furthermore, the use of common solvents like dichloromethane and methanol ensures that solvent availability is not a bottleneck, securing the continuity of operations even during global supply disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with green chemistry initiatives. The high selectivity reduces the generation of hazardous waste streams associated with separating stereoisomers, lowering disposal costs and environmental impact. The process has been demonstrated to scale effectively, as evidenced by successful larger-batch experiments that maintained 100% conversion and 99% ee. This scalability assures stakeholders that the technology can meet commercial volume demands without compromising on quality or safety standards, facilitating a smoother path from pilot plant to full-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the Ru-C3*-Tunephos catalytic system. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure.
Q: What represents the key advantage of the Ru-C3*-Tunephos system over traditional BINAP-Ru catalysts?
A: The Ru-C3*-Tunephos system offers significantly higher turnover numbers (TON up to 5000) and eliminates the need for specialized amine additives like NEt3, simplifying the process and reducing catalyst costs while maintaining superior stereoselectivity (>99% ee).
Q: Is this asymmetric hydrogenation process suitable for large-scale industrial manufacturing?
A: Yes, the patent demonstrates successful scale-up capabilities with robust conversion rates and selectivity maintained even at larger batches, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this method impact the purity profile of the final carbapenem precursor?
A: By achieving diastereomeric excess (de) and enantiomeric excess (ee) values up to 99%, the method drastically reduces the formation of unwanted stereoisomers, thereby simplifying downstream purification and ensuring stringent purity specifications for API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-2-amidomethyl-3-hydroxybutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for critical antibiotic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify enantiomeric excess and chemical purity. By leveraging the advanced Ru-C3*-Tunephos technology, we can offer our partners a supply of (2S,3R)-2-amidomethyl-3-hydroxybutyrate that is both cost-effective and of the highest quality, supporting the global demand for next-generation carbapenem antibiotics.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in asymmetric catalysis can drive value and efficiency in your manufacturing operations.
