Scalable Chemical Synthesis of High-Purity Inulin-type Fructooligosaccharide Monomers for Drug Development
The pharmaceutical and functional food industries have long recognized the therapeutic potential of inulin-type fructooligosaccharides, yet accessing pure monomeric forms has remained a significant bottleneck. Patent CN115073540A introduces a groundbreaking chemical synthesis method that overcomes the limitations of traditional extraction and enzymatic hydrolysis. This innovation enables the precise construction of inulin-type fructooligosaccharide monomers, ranging from kestose to heptaose, through a highly stereoselective modular approach. By utilizing a sucrose derivative with a naked terminal hydroxyl group as a glycosyl acceptor and a specifically designed fructofuranose thioglycoside as a donor, the process achieves exceptional control over the glycosidic bond formation. This technological leap not only ensures the production of high-purity intermediates essential for rigorous pharmacological studies but also establishes a robust foundation for the commercial manufacturing of these valuable bioactive compounds.
The limitations of conventional methods for obtaining inulin-type fructooligosaccharides are profound and multifaceted, primarily stemming from their reliance on natural sources. Traditionally, these compounds are extracted from plants such as chicory or Morinda officinalis, followed by enzymatic or acid hydrolysis. This approach inherently yields a complex mixture of oligosaccharides with varying degrees of polymerization, making the isolation of specific monomers like kestose or nystose extremely labor-intensive and inefficient. Chromatographic separation is often required, which drastically reduces overall yield and increases production costs, rendering the final products unsuitable for large-scale pharmaceutical applications where consistency and purity are paramount. Furthermore, the structural heterogeneity of extracts complicates the elucidation of structure-activity relationships, hindering the development of targeted therapeutics.
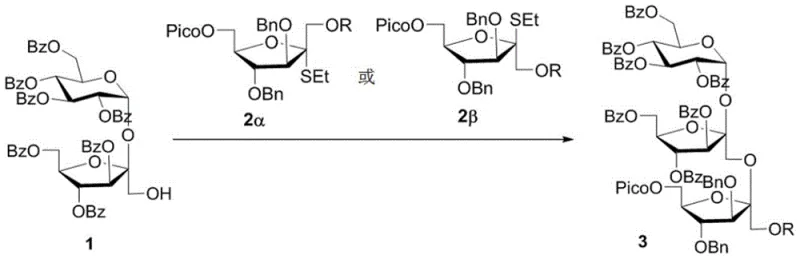
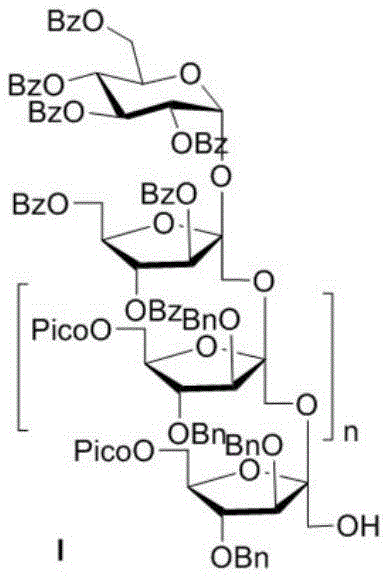
In stark contrast, the novel approach detailed in the patent utilizes a rational chemical synthesis strategy that offers unparalleled precision and flexibility. The core of this methodology lies in the iterative assembly of sugar units using a modular building block system. By employing a fructofuranose thioglycoside donor equipped with orthogonal protecting groups, specifically a 2-picolinoyl (Pico) group at the C6 position, the synthesis ensures the exclusive formation of the challenging β(2,1)-D-fructofuranoside glycosidic bond. This allows for the stepwise elongation of the sugar chain, where each cycle adds a single fructose unit with defined stereochemistry. The ability to control the polymerization degree chemically means that manufacturers can produce specific monomers on demand, eliminating the need for difficult separations and ensuring a consistent supply of high-quality material for drug development pipelines.
Mechanistic Insights into Hydrogen Bond-Mediated Stereoselective Glycosylation
The success of this synthesis hinges on the sophisticated mechanistic design of the glycosyl donor, which leverages a hydrogen bond-mediated intramolecular glycosyl transfer strategy. The presence of the 2-picolinoyl (Pico) ester at the C6-hydroxyl position of the fructofuranose donor plays a critical role in directing the stereochemical outcome of the reaction. During the activation of the thioglycoside, the carbonyl oxygen of the Pico group forms a transient hydrogen bond with the incoming hydroxyl group of the acceptor or stabilizes the oxocarbenium ion intermediate in a specific conformation. This interaction effectively shields the alpha-face of the sugar ring, forcing the nucleophilic attack to occur exclusively from the beta-face. Consequently, the reaction proceeds with remarkable stereoselectivity, achieving a beta-to-alpha ratio greater than 20:1, which is a significant improvement over previous methods that often struggled with anomeric mixtures.
From an impurity control perspective, this high level of stereocontrol is transformative for process chemistry. In traditional glycosylation reactions, the formation of alpha-anomers constitutes a major impurity that is structurally similar to the desired product, making purification difficult and costly. By virtually eliminating the formation of the alpha-anomer at the source, this method simplifies downstream processing significantly. The use of orthogonal protecting groups, such as benzoyl (Bz) and benzyl (Bn) groups alongside the Pico and silyl groups, further enhances purity profiles by allowing for selective deprotection steps that do not affect other parts of the molecule. This orthogonality ensures that side reactions are minimized, and the final product retains the integrity of the sensitive fructofuranose rings, which are prone to hydrolysis under harsh acidic or basic conditions.
How to Synthesize Inulin-type Fructooligosaccharide Monomers Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these complex oligosaccharides with high efficiency. The process begins with the preparation of the key fructofuranose thioglycoside donor, followed by its coupling with a sucrose-based acceptor. The reaction conditions are optimized to maintain the stability of the furanose ring while ensuring rapid activation of the donor. Subsequent steps involve the selective removal of the temporary protecting group at the new terminal position to regenerate the acceptor for the next coupling cycle. This iterative loop can be repeated multiple times to build oligosaccharides of varying lengths. The detailed standardized synthetic steps for executing this modular assembly are provided in the guide below.
- Perform stereoselective glycosylation using a sucrose derivative acceptor and a fructofuranose thioglycoside donor with a Pico protecting group.
- Selectively remove the terminal C1 hydroxyl protecting group to regenerate the glycosyl acceptor for the next cycle.
- Repeat the glycosylation and deprotection sequence to extend the sugar chain to the desired polymerization degree, followed by global deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from extraction-based sourcing to this chemical synthesis platform offers substantial strategic benefits. The primary advantage lies in the decoupling of production from agricultural variables. Unlike plant extraction, which is subject to seasonal fluctuations, crop yields, and geographical constraints, this chemical method relies on stable, commercially available starting materials. This shift ensures a consistent and reliable supply of critical intermediates, mitigating the risk of shortages that can disrupt pharmaceutical manufacturing schedules. Furthermore, the modular nature of the synthesis allows for flexible production planning, where inventory levels of specific monomers can be adjusted rapidly based on market demand without the long lead times associated with cultivating and processing biomass.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of cost-effective reagents and streamlined operations. The glycosylation promoter, DBDMH, is inexpensive compared to many precious metal catalysts used in other fine chemical syntheses, leading to direct savings in raw material costs. Additionally, the high stereoselectivity reduces the burden on purification processes, lowering solvent consumption and waste disposal costs. The ability to perform multiple steps without intermediate isolation, as demonstrated in the preparation of the thioglycoside donors, further compresses the manufacturing timeline and reduces labor expenses, resulting in a more competitive cost structure for the final active ingredients.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved through the use of robust and scalable chemical transformations. The reagents and solvents employed, such as dichloromethane, toluene, and common silyl chlorides, are commodity chemicals with established global supply networks. This reduces dependency on niche suppliers and minimizes the risk of logistics bottlenecks. Moreover, the synthetic route is designed to tolerate scale-up, with examples in the patent demonstrating reactions on the scale of tens of grams. This proven scalability provides confidence that the process can be transferred to multi-kilogram or ton-scale production facilities to meet commercial volume requirements without compromising quality or yield.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles. The elimination of heavy metal catalysts simplifies the removal of toxic residues, ensuring that the final pharmaceutical intermediates meet stringent safety specifications. The process generates less hazardous waste compared to traditional extraction methods that require large volumes of organic solvents for purification. Furthermore, the high atom economy of the glycosylation steps and the potential for solvent recycling contribute to a reduced environmental footprint. This compliance facilitates smoother regulatory approvals and supports corporate sustainability goals, making the supply chain more attractive to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of inulin-type fructooligosaccharide monomers. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these intermediates into your specific product development workflows.
Q: What is the key advantage of this chemical synthesis over natural extraction?
A: Chemical synthesis provides single-component monomers with defined structures and high purity, whereas natural extraction yields complex mixtures that are difficult to separate.
Q: How is stereocontrol achieved in the glycosylation reaction?
A: Stereocontrol is achieved through a hydrogen bond-mediated intramolecular glycosyl transfer strategy directed by a 2-picolinoyl (Pico) protecting group at the C6 position.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method uses common reagents and mild conditions, and the modular nature allows for efficient scale-up from grams to industrial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Inulin-type Fructooligosaccharide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies into commercial reality. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising synthesis method described in CN115073540A can be effectively implemented at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch of inulin-type fructooligosaccharide monomer meets the highest standards of quality and consistency demanded by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will accelerate your development process. Contact us today to discuss how we can secure your supply chain with high-purity, chemically synthesized fructooligosaccharide monomers.
