Scalable and Safe Production of Chiral Brivaracetam Intermediates via Novel Hydrolysis Route
Scalable and Safe Production of Chiral Brivaracetam Intermediates via Novel Hydrolysis Route
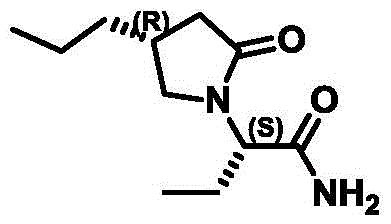
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value antiepileptic agents, with Brivaracetam standing out as a critical next-generation therapeutic. The synthesis of its key chiral building block, (R)-4-propyl-dihydrofuran-2-one, has historically been plagued by safety hazards and operational complexities. Patent CN111349007B introduces a transformative preparation method that fundamentally alters the risk profile of this synthesis. By replacing dangerous oxidative reagents with conventional hydrolysis and reduction protocols, this technology offers a pathway to high-purity intermediates that aligns with modern green chemistry principles. This report analyzes the technical merits of this innovation, providing R&D and procurement leaders with a clear understanding of its potential to optimize supply chains for pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral lactones like (R)-4-propyl-dihydrofuran-2-one has relied on routes that introduce significant safety liabilities at the industrial scale. As illustrated in prior art such as CN106008411A, traditional pathways often necessitate the use of hydrogen peroxide and borane dimethyl sulfide complexes. These reagents are thermally unstable and pose severe explosion risks, particularly when exothermic reactions are not perfectly controlled in large reactors. Furthermore, alternative routes involving Grignard reagents, as shown in earlier literature, frequently result in racemic mixtures that require costly and yield-reducing chiral separation steps. The reliance on such hazardous materials not only inflates insurance and compliance costs but also creates bottlenecks in production scheduling due to stringent safety protocols.
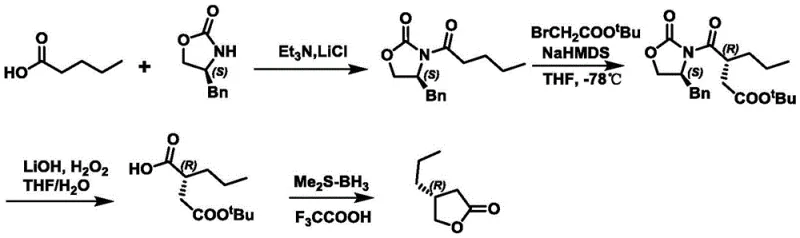
The Novel Approach
The methodology disclosed in CN111349007B circumvents these historical pitfalls by employing a mild hydrolysis followed by a selective reduction. Instead of aggressive oxidation, the process utilizes standard acidic or basic conditions to cleave the precursor ester, generating a carboxylic acid salt that is inherently easier to handle. This shift allows manufacturers to utilize industrially cheap and readily available reagents, effectively decoupling production costs from the volatility of specialty chemical markets. By eliminating the need for energetic reagents, the novel approach not only enhances operator safety but also simplifies the regulatory burden associated with storing and transporting hazardous materials, making it a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Base Mediated Hydrolysis and Reduction
The core innovation lies in the strategic manipulation of pH to drive both reaction progress and purification. In the first stage, the oxazolidinone derivative (Compound I) undergoes hydrolysis in the presence of a base such as lithium hydroxide or sodium hydroxide. This reaction converts the ester moiety into a water-soluble carboxylate salt (Compound II). Unlike traditional methods that might require organic extraction of the acid, this salt formation allows for a unique workup where organic impurities remain in the organic phase while the desired intermediate resides in the aqueous layer. This phase separation is driven purely by solubility differences dictated by ionization, removing the need for chromatographic purification or multiple recrystallizations that typically erode yield.
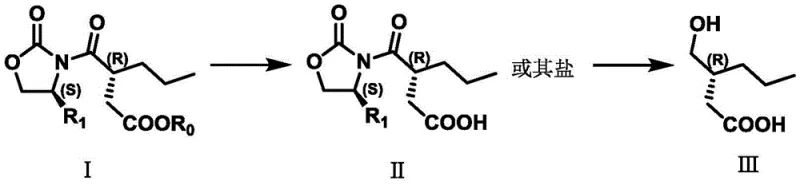
Following hydrolysis, the subsequent reduction step utilizes mild hydride donors like sodium borohydride or potassium borohydride. These reagents selectively reduce the ketone functionality without affecting the sensitive chiral centers or the oxazolidinone auxiliary. The reaction proceeds efficiently in mixed solvent systems containing tetrahydrofuran and water, ensuring homogeneous reaction conditions that facilitate heat transfer. The mechanism ensures that the stereochemical integrity established by the chiral auxiliary is preserved throughout the transformation, delivering the hydroxy-acid intermediate (Compound III) with high optical purity. This mechanistic elegance ensures that the final cyclization step proceeds with minimal formation of diastereomeric impurities, a critical factor for meeting the stringent quality standards of global regulatory agencies.
How to Synthesize (R)-4-Propyl-Dihydrofuran-2-One Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing the target lactone with high fidelity. The process begins with the preparation of the aqueous salt solution, followed by the addition of the reducing agent under controlled temperature conditions to manage exotherms. The final cyclization is achieved by acidifying the reaction mixture, which triggers the intramolecular esterification to close the lactone ring. For detailed operational parameters, stoichiometry, and specific temperature profiles required to replicate this high-yielding process, please refer to the standardized technical guide below.
- Perform hydrolysis on the oxazolidinone precursor (Compound I) using aqueous acid or base to generate the carboxylic acid salt (Compound II), utilizing pH control for purification instead of crystallization.
- Execute a reduction reaction on Compound II using safe borohydride reagents (e.g., NaBH4 or KBH4) in a mixed solvent system to yield the hydroxy-acid intermediate (Compound III).
- Complete the synthesis via acid-catalyzed cyclization of Compound III in an organic solvent to form the final lactone product (Compound IV), followed by standard aqueous workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route represents a significant opportunity to de-risk the supply of critical epilepsy medication ingredients. The elimination of explosive reagents removes a major category of operational risk, ensuring that production facilities can maintain continuous operation without the frequent safety shutdowns associated with handling peroxides. Furthermore, the simplification of the workup procedure—relying on phase separation rather than filtration and recrystallization—drastically reduces the cycle time per batch. This efficiency gain translates directly into increased plant capacity and the ability to respond more agilely to fluctuating market demand without requiring capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous specialty reagents with commodity chemicals like sodium hydroxide and sodium borohydride leads to substantial raw material cost savings. Additionally, the avoidance of complex purification steps such as recrystallization reduces solvent consumption and waste disposal costs. The overall process efficiency means that less manpower and utility time are required per kilogram of product, driving down the fully loaded cost of goods sold and improving margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: By utilizing reagents that are globally sourced and not subject to the strict transportation restrictions of hazardous explosives, the supply chain becomes more resilient. Manufacturers are less vulnerable to logistical disruptions caused by regulatory changes in the transport of dangerous goods. The robustness of the chemistry also ensures consistent batch-to-batch quality, reducing the risk of supply interruptions due to failed quality control tests and ensuring a steady flow of high-purity pharmaceutical intermediates to API production sites.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The aqueous workup generates less organic waste compared to solvent-intensive recrystallization processes, aligning with increasingly strict environmental regulations. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized explosion-proof equipment, facilitating rapid technology transfer from pilot plants to full-scale commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for feasibility assessments.
Q: How does this new method improve safety compared to prior art routes?
A: Unlike previous methods disclosed in CN106008411A which utilized hydrogen peroxide and borane dimethyl sulfide—reagents known for thermal instability and explosion risks—this patent employs standard hydrolysis and borohydride reduction. This substitution eliminates the handling of energetic materials, drastically lowering the safety profile risks associated with industrial scale-up.
Q: What are the purification advantages of this specific synthetic route?
A: The process leverages the acid-base properties of the intermediates to achieve purification. By adjusting the pH of the reaction system, impurities are separated into the aqueous or organic phase without the need for energy-intensive recrystallization or complex filtration steps, thereby simplifying the post-treatment workflow and improving overall throughput.
Q: Is this route suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial mass production. The use of commercially available, inexpensive reagents and the avoidance of hazardous conditions make it highly adaptable for multi-kilogram to ton-scale production environments required by global supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-Propyl-Dihydrofuran-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safe and efficient synthesis routes for high-value chiral intermediates. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (R)-4-propyl-dihydrofuran-2-one meets the exacting standards required for Brivaracetam synthesis, providing our partners with absolute confidence in their supply continuity.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this safer methodology. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production volume requirements, and let us help you secure a competitive advantage in the antiepileptic drug market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
