Advanced Chiral Synthesis of (S)-3-Boc-Aminomethyl Piperidine for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for chiral intermediates that drive the efficacy of cardiovascular therapeutics. Patent CN108409639B introduces a groundbreaking preparation and structure confirmation method for (S)-3-Boc-aminomethyl piperidine, a critical building block in this therapeutic class. Unlike conventional approaches that rely on inefficient resolution processes, this invention leverages a chiral pool strategy starting from inexpensive, readily available chiral raw materials. The technical breakthrough lies in the direct synthesis of the target chirality without the need for external resolving agents, which fundamentally alters the economic and operational landscape of production. By integrating benzylation, carbonyl reduction, amino protection, and catalytic hydrogenation into a streamlined sequence, the method ensures high optical purity exceeding 99% ee. This report analyzes the technical superiority of this route, offering R&D directors and procurement leaders a clear pathway to optimizing their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
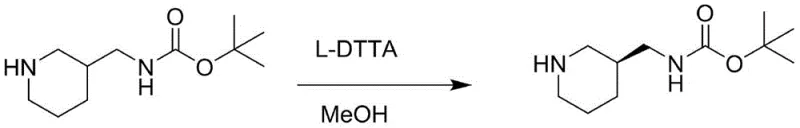
Historically, the production of (S)-3-Boc-aminomethyl piperidine has been plagued by the inefficiencies inherent in racemic resolution technologies. The prior art predominantly utilizes L-DTTA (L-(-)-di-p-methylbenzoyl tartaric acid) as a resolving agent to separate the desired S-enantiomer from the racemic mixture. This approach suffers from a theoretical maximum yield of 50%, but in practical industrial applications, yields often fall below 33% due to losses during crystallization and mother liquor processing. Furthermore, the reliance on stoichiometric amounts of expensive chiral acids significantly inflates the raw material costs, creating a heavy financial burden for large-scale manufacturing. The generation of substantial chemical waste, including the unwanted R-enantiomer and the resolving agent salts, poses severe environmental compliance challenges and increases the cost of three-waste treatment. These factors combined make the traditional resolution route economically unsustainable for modern, cost-sensitive API production.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent circumvents these bottlenecks by employing a chiral starting material that already possesses the desired stereochemistry. This strategic shift eliminates the need for any resolving agent, thereby removing the 50% yield ceiling associated with resolution. The synthetic route is designed to preserve the chiral integrity throughout the transformation, ensuring that the final product maintains high optical purity without the need for corrective purification steps. By avoiding the use of L-DTTA, the process drastically simplifies the workup procedure, reducing solvent consumption and waste generation. The economic implications are profound, as the cost of goods sold (COGS) is significantly lowered through both improved yield efficiency and reduced reagent costs. This approach represents a paradigm shift from separation-based chemistry to construction-based chemistry, offering a sustainable and commercially viable alternative for the production of complex piperidine derivatives.
Mechanistic Insights into Chiral Pool Synthesis and Catalytic Hydrogenation
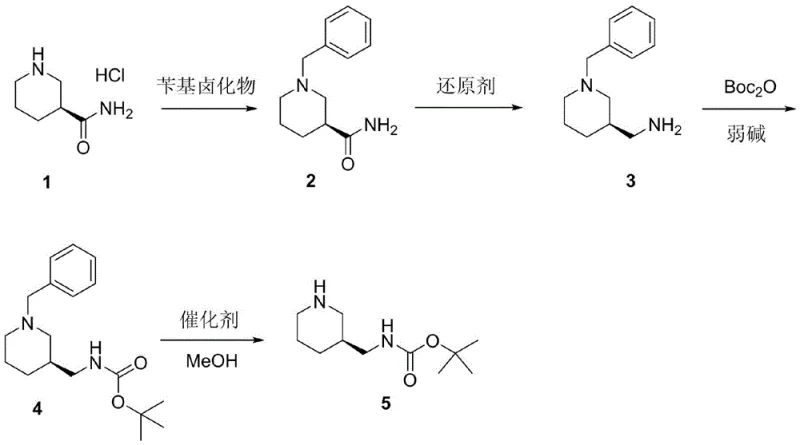
The core of this innovative process is a four-step synthetic sequence that meticulously constructs the target molecule while safeguarding its chiral center. The journey begins with the benzylation of the chiral starting Compound 1, where precise temperature control between 8-12°C during the addition of benzyl halide prevents racemization and side reactions. Following this, the carbonyl reduction step utilizes potent reducing agents such as lithium aluminum hydride or sodium borohydride variants in tetrahydrofuran, converting the amide functionality to the corresponding amine with high fidelity. The subsequent protection of the amino group using di-tert-butyldicarbonate (Boc2O) under weakly basic conditions ensures the stability of the intermediate for the final transformation. The culmination of the synthesis is a catalytic hydrogenation step using palladium on carbon or platinum on carbon in methanol, which cleanly removes the benzyl protecting group to reveal the free piperidine nitrogen. This final step is critical, as it must proceed under mild conditions (20-25°C) to avoid hydrogenolysis of the Boc group or epimerization of the chiral center.
Impurity control is embedded deeply within the mechanistic design of this route, addressing a primary concern for R&D directors focused on regulatory compliance. The use of high-purity chiral starting materials minimizes the introduction of diastereomeric impurities at the source. Throughout the synthesis, specific purification protocols, such as extraction with dichloromethane or methyl tert-butyl ether and crystallization from petroleum ether, are employed to remove inorganic salts and organic byproducts. The patent explicitly details a derivatization strategy where the final product is reacted with N-benzyloxycarbonyl-L-proline to form a crystalline derivative. This allows for rigorous structure confirmation via single-crystal X-ray diffraction, providing irrefutable evidence of the (S)-configuration. Such robust analytical validation ensures that the impurity profile remains within strict specifications, facilitating smoother regulatory filings and reducing the risk of batch rejection during quality control testing.
How to Synthesize (S)-3-Boc-Aminomethyl Piperidine Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the dissolution of the chiral starting material in DMF followed by the controlled addition of base and benzylating agent. Subsequent reduction and protection steps demand anhydrous conditions and precise stoichiometric ratios to prevent over-reaction or incomplete conversion. The final hydrogenation step is particularly sensitive to catalyst loading and pressure, requiring optimization to ensure complete deprotection without compromising the Boc group. For a detailed breakdown of the standardized operating procedures, including specific molar ratios, temperature profiles, and workup techniques derived from the patent examples, please refer to the technical guide below.
- Benzylation of chiral starting material Compound 1 using benzyl halide and potassium carbonate in DMF at controlled low temperatures.
- Reduction of the carbonyl group in Compound 2 using lithium aluminum hydride or sodium borohydride variants in tetrahydrofuran.
- Amino protection of Compound 3 using di-tert-butyldicarbonate (Boc2O) and weak base to form Compound 4.
- Final catalytic hydrogenation of Compound 4 using Pd/C or Pt/C in methanol to remove the benzyl group and yield the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers transformative benefits that extend beyond simple chemistry. The elimination of the resolution step fundamentally reshapes the cost structure of the intermediate, removing the need for expensive chiral acids and the associated processing costs. This leads to substantial cost savings in API manufacturing, allowing for more competitive pricing strategies in the final drug product. Moreover, the reliance on cheap and easily obtained chiral raw materials enhances supply chain resilience, reducing dependency on specialized reagents that may face availability fluctuations. The simplified process flow also translates to shorter production cycles, effectively reducing lead time for high-purity pharmaceutical intermediates and enabling faster response to market demand.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the removal of the resolution agent L-DTTA, which is not only costly but also results in the loss of half the material mass in traditional methods. By synthesizing the chiral center directly from the starting material, the process effectively doubles the theoretical yield relative to the input mass compared to resolution methods. This efficiency gain, combined with the use of common solvents like methanol and THF instead of specialized resolution media, drastically lowers the variable cost per kilogram. Additionally, the reduction in waste generation lowers the environmental compliance costs associated with waste disposal and treatment, further contributing to the overall economic advantage of the process.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing specialized chiral resolving agents. This new method mitigates that risk by utilizing commodity chemicals and widely available chiral pool starting materials. The robustness of the catalytic hydrogenation step, which uses standard heterogeneous catalysts like Pd/C, ensures that the process can be executed in diverse manufacturing facilities without requiring exotic equipment. This flexibility allows for multi-vendor sourcing strategies and reduces the risk of production stoppages due to raw material shortages. Consequently, partners can rely on a more stable and predictable supply of this critical cardiovascular intermediate, ensuring uninterrupted production of downstream APIs.
- Scalability and Environmental Compliance: From an operational perspective, the mild reaction conditions described in the patent, such as ambient temperature hydrogenation and atmospheric pressure operations for protection steps, make this route highly amenable to scale-up. The absence of cryogenic conditions or high-pressure hazards simplifies the engineering requirements for commercial reactors. Furthermore, the significant reduction in chemical waste aligns with green chemistry principles, minimizing the environmental footprint of the manufacturing process. This compliance with stringent environmental regulations facilitates smoother permitting processes and enhances the sustainability profile of the supply chain, which is increasingly important for global pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and experimental data presented in the patent documentation, providing clarity on yield expectations, purity standards, and scalability. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this new method improve yield compared to traditional resolution?
A: Traditional methods using L-DTTA resolution typically achieve yields below 33% due to the inherent 50% loss of the unwanted enantiomer. This novel chiral pool synthesis bypasses the resolution step entirely, directly utilizing cheap chiral raw materials to theoretically double the potential yield from the starting mass.
Q: What is the optical purity of the final product?
A: The patent data confirms that the synthesized (S)-3-Boc-aminomethyl piperidine achieves an optical purity (ee%) of greater than 99%, with a specific optical rotation value of -8.98, verified through single-crystal X-ray diffraction of its derivative.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (e.g., 20-25°C for hydrogenation) and common solvents like methanol and THF. The elimination of complex resolution agents and the use of robust catalytic hydrogenation make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Boc-Aminomethyl Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving cardiovascular medications. Our team of expert chemists has thoroughly analyzed the technology behind patent CN108409639B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chiral pool synthesis are realized in large-volume manufacturing. Our state-of-the-art facilities are designed to handle sensitive hydrogenation reactions and maintain stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this resolution-free method for your projects. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a reliable, cost-effective, and high-purity supply of (S)-3-Boc-aminomethyl piperidine, driving efficiency and innovation in your pharmaceutical manufacturing operations.
