Scalable Cordycepin Production: A Technical Breakthrough in Nucleoside Analog Synthesis
Scalable Cordycepin Production: A Technical Breakthrough in Nucleoside Analog Synthesis
The pharmaceutical industry's demand for high-purity nucleoside analogs continues to surge, driven by the critical role these compounds play in antiviral and oncology therapeutics. Among these, Cordycepin (3'-deoxyadenosine) stands out as a potent bioactive molecule with significant potential in immunomodulation and metabolic regulation. However, historical manufacturing bottlenecks have often hindered its widespread commercial adoption. A pivotal advancement in this domain is detailed in patent CN108676048B, which outlines a revolutionary preparation method that fundamentally reimagines the synthetic landscape. By shifting away from hazardous organotin reagents and complex protecting group strategies, this technology offers a pathway to a reliable cordycepin supplier status through enhanced efficiency and safety. This report provides a deep technical dissection of this methodology, evaluating its mechanistic robustness and its profound implications for cost reduction in nucleoside manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cordycepin has been plagued by significant technical and economic hurdles that compromise scalability. The most prevalent legacy route involves the use of adenosine as a starting material, where the 2'- and 5'-hydroxyl groups are protected using tert-butyldimethylsilyl chloride. This is followed by a deoxygenation step utilizing phenyl thiocarbonate and tri-n-butyltin hydride. As illustrated in the reaction scheme below, this pathway is fraught with inefficiencies.
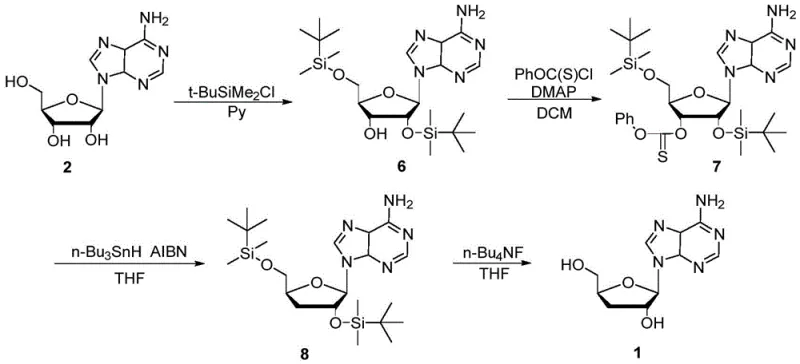
The reliance on organotin chemistry introduces severe environmental and purification challenges, as removing trace tin residues to meet pharmaceutical standards is notoriously difficult and costly. Furthermore, the strict requirement for anhydrous conditions throughout the silylation and subsequent steps increases operational complexity and energy consumption. Alternative routes involving orthoacetate cyclization often suffer from poor regioselectivity, generating substantial amounts of 2',3'-dideoxyadenosine byproducts, which drastically reduces the overall yield and complicates downstream isolation. These factors collectively render traditional methods suboptimal for the commercial scale-up of complex nucleoside analogs.
The Novel Approach
In stark contrast, the methodology disclosed in CN108676048B presents a streamlined, three-stage strategy that bypasses these historical impediments. The core innovation lies in the direct acylation of adenosine followed by a selective halogenation and a final catalytic reduction. This approach eliminates the need for bulky silyl protecting groups and toxic tin hydrides entirely. Instead, it employs common acid anhydrides or acyl chlorides to protect the amino and 5'-hydroxyl functionalities, creating a stable intermediate ready for transformation.
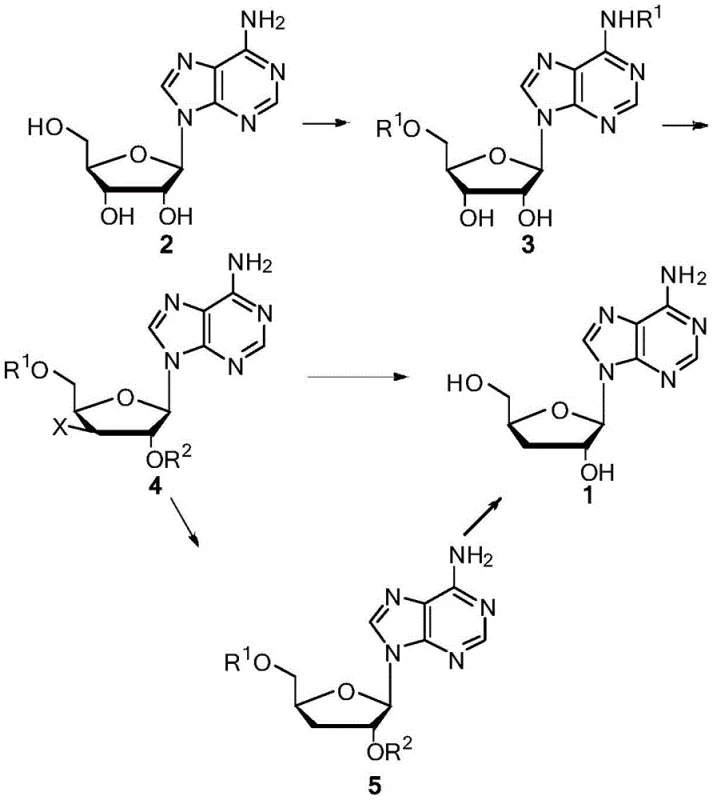
The subsequent conversion to the 3'-halo intermediate allows for a clean reduction step using a formate system. This catalytic transfer hydrogenation (CTH) operates under mild conditions, avoiding the high-pressure equipment required for gaseous hydrogenation. The result is a process that not only improves the impurity profile by minimizing side reactions but also drastically simplifies the workup procedure. By utilizing readily available starting materials and avoiding exotic reagents, this novel approach establishes a new benchmark for cost reduction in nucleoside manufacturing, making high-purity cordycepin accessible for broader therapeutic applications.
Mechanistic Insights into Catalytic Transfer Hydrogenation and Regioselective Acylation
The success of this synthetic route hinges on two critical chemical transformations: the regioselective acylation of the ribose moiety and the subsequent catalytic transfer hydrogenation. In the initial step, the differential reactivity of the hydroxyl groups on the ribose ring is exploited. Under controlled conditions with acetic anhydride in pyridine, the primary 5'-hydroxyl and the exocyclic amino group are preferentially acylated. This selectivity is crucial as it leaves the secondary 3'-hydroxyl group available for the subsequent activation step. The formation of the 3'-bromo intermediate via reaction with acetyl bromide proceeds through an SN2-like mechanism where the acylated environment stabilizes the transition state, ensuring that the halogenation occurs specifically at the 3'-position without affecting the sensitive glycosidic bond.
The reduction phase represents the mechanistic heart of this innovation, utilizing a palladium-catalyzed transfer hydrogenation system. Unlike traditional hydrogenation which relies on molecular hydrogen gas, this method uses formic acid or formate salts as the hydrogen donor. The mechanism involves the adsorption of the formate species onto the palladium surface, where it undergoes decarboxylation to generate a metal-hydride species in situ. This active hydride then transfers to the 3'-carbon of the nucleoside intermediate, displacing the halogen leaving group. This pathway is kinetically favorable and thermodynamically driven by the evolution of carbon dioxide. Crucially, this mechanism avoids the over-reduction of the purine base, a common side reaction in high-pressure hydrogenation, thereby preserving the structural integrity of the adenine ring and ensuring a superior impurity profile for the final high-purity cordycepin product.
How to Synthesize Cordycepin Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the dissolution of adenosine in a polar aprotic solvent such as pyridine or DMF, followed by the controlled addition of the acylating agent at low temperatures to manage exotherms. Once the protected intermediate is isolated, it is subjected to halogenation using acyl halides at elevated temperatures, typically between 40°C and 90°C, to drive the substitution to completion. The final reduction step is performed in alcoholic solvents using a heterogeneous palladium catalyst and a formate source. For a comprehensive, step-by-step operational guide including specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized protocol below.
- Protect the amino and 5'-hydroxyl groups of adenosine using acetic anhydride or acyl chloride in pyridine to form the protected intermediate.
- React the protected intermediate with an acyl halide (e.g., acetyl bromide) at elevated temperatures to introduce the leaving group at the 3'-position.
- Perform catalytic transfer hydrogenation using a formate system and Pd/C catalyst, followed by deacylation to yield high-purity cordycepin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers transformative benefits that extend far beyond simple yield improvements. The elimination of specialized, high-cost reagents such as tert-butyldimethylsilyl chloride and tri-n-butyltin hydride directly impacts the bill of materials, leading to substantial cost savings. Furthermore, the removal of toxic tin residues from the process stream significantly reduces waste disposal costs and environmental compliance burdens. This shift allows manufacturers to operate with a leaner, more agile supply chain that is less vulnerable to the price volatility of exotic chemical reagents.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling due to the replacement of expensive stoichiometric reagents with catalytic systems. By utilizing catalytic transfer hydrogenation, the need for high-pressure autoclaves and the associated safety infrastructure is removed, lowering capital expenditure requirements. Additionally, the use of commodity chemicals like acetic anhydride and formic acid ensures that raw material costs remain stable and predictable. The simplified purification process, which avoids complex chromatography, further reduces solvent consumption and processing time, driving down the overall cost of goods sold significantly.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for pharmaceutical intermediates, and this method secures the supply chain by relying on adenosine, a widely available fermentation product. Unlike routes dependent on custom-synthesized alkynes or specialized silylating agents which may have single-source suppliers, the reagents for this process are commoditized and available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions means that production can be maintained consistently without frequent batch failures due to moisture sensitivity.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process is inherently safer and more sustainable. The absence of high-pressure hydrogen gas eliminates explosion hazards, while the avoidance of organotin compounds prevents the generation of persistent organic pollutants. This green chemistry profile facilitates easier regulatory approval and permits for manufacturing facilities. The process is designed for linear scalability, meaning that the kinetics and heat transfer characteristics observed in the lab translate effectively to multi-ton reactors, ensuring that commercial scale-up of complex nucleoside analogs can be achieved without unforeseen engineering bottlenecks.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common concerns regarding the practical implementation and quality attributes of the cordycepin produced via this method. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities and limitations in a real-world manufacturing context.
Q: How does this new method improve upon traditional silyl-protection routes?
A: Traditional routes rely on expensive tert-butyldimethylsilyl chloride and toxic tributyltin hydride under strict anhydrous conditions. The new method utilizes inexpensive acylation reagents and catalytic transfer hydrogenation, eliminating toxic tin waste and simplifying purification.
Q: What are the safety advantages of using catalytic transfer hydrogenation?
A: Unlike high-pressure hydrogenation which requires specialized equipment and poses explosion risks, catalytic transfer hydrogenation uses formic acid or formates as hydrogen donors at atmospheric pressure and mild temperatures (20-60°C), significantly enhancing operational safety.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available adenosine as a starting material and avoids complex chromatographic purifications. The mild reaction conditions and robust catalyst systems make it highly amenable to multi-kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cordycepin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality. Our team of process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by CN108676048B are fully realized in our manufacturing suites. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of cordycepin meets the highest international pharmacopoeia standards. Our commitment to quality assurance ensures that our clients receive a product with a consistent impurity profile, essential for downstream drug formulation.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can secure your supply of high-purity cordycepin while driving down your overall production costs.
