Revolutionizing Nucleoside Cracking: A Scalable Route to High-Purity Tetraacetyl Ribofuranose and Acetylated Purines
The pharmaceutical industry's relentless demand for high-quality antiviral and antineoplastic intermediates has long been constrained by the inefficiencies of traditional nucleoside modification techniques. Patent CN101701027B introduces a transformative catalytic cracking method that addresses these bottlenecks by enabling the simultaneous production of 1,2,3,5-O-tetraacetyl-β-D-ribofuranose and valuable acetylated purine byproducts. This technology represents a significant leap forward for any reliable pharmaceutical intermediates supplier seeking to optimize their manufacturing portfolio for drugs like ribavirin and stavudine. By leveraging specific strong acid catalysts at controlled temperatures, the process achieves exceptional conversion rates while minimizing the formation of degradation products that often compromise downstream synthesis. The strategic integration of this methodology allows manufacturers to secure a consistent supply of critical building blocks essential for the global antiviral market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of tetraacetyl ribose has relied on methodologies that are fraught with economic and technical inefficiencies, creating substantial barriers for cost reduction in API manufacturing. Early approaches, such as the Beranek method, utilized glacial acetic acid as a catalyst but suffered from dismal yields of approximately 47%, rendering them commercially unviable for large-scale operations. Subsequent improvements involving tosic acid increased yields to around 84%, yet the purification processes remained cumbersome and prone to generating colored impurities that required extensive remediation. Furthermore, methods employing solid superacids or ion-exchange resins, while achieving higher theoretical yields, introduced severe logistical challenges regarding catalyst recovery and regeneration, often leading to prohibitive production costs and inconsistent batch quality. These legacy techniques frequently result in product discoloration and variable purity profiles, necessitating additional refining steps that erode profit margins and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative strategy outlined in the patent data utilizes soluble strong acid catalysts, such as trifluoroacetic acid or trifluoromethanesulfonic acid, to facilitate a highly efficient cleavage and acetylation cascade. This approach fundamentally alters the reaction landscape by operating at moderate temperatures between 50°C and 140°C, significantly reducing energy consumption compared to harsher thermal protocols. The use of acetic anhydride serves a dual purpose as both the solvent and the acetylating agent, driving the equilibrium towards the desired tetraacetyl ribose and acetylated purine products with remarkable selectivity. Unlike resin-based systems, the homogeneous nature of the liquid acid catalyst ensures uniform reaction kinetics throughout the mixture, eliminating mass transfer limitations and ensuring consistent product quality across large batches. This methodology not only simplifies the workflow by allowing for direct filtration of the purine byproduct but also enables the recovery of the ribose derivative through straightforward distillation and recrystallization, streamlining the entire production sequence.
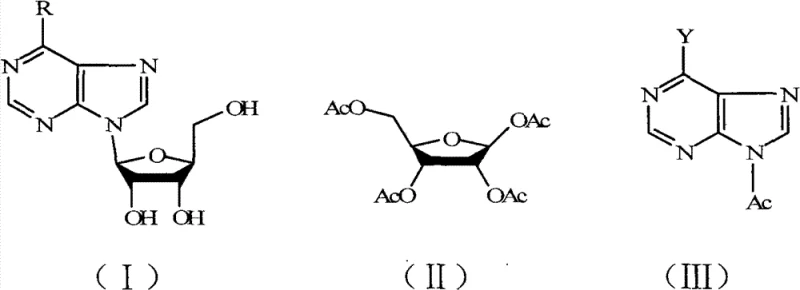
Mechanistic Insights into Acid-Catalyzed Glycosidic Bond Cleavage
The core of this technological advancement lies in the precise activation of the N-glycosidic bond within the nucleoside structure through potent proton donation from the fluorinated acid catalysts. When trifluoroacetic acid or trifluoromethanesulfonic acid is introduced to the reaction matrix, the high electronegativity of the fluorine atoms enhances the acidity, facilitating the rapid protonation of the purine base nitrogen. This protonation weakens the bond between the sugar moiety and the heterocyclic base, promoting the formation of a reactive oxocarbenium ion intermediate on the ribose ring. Simultaneously, the abundant acetic anhydride present in the system acts as a nucleophile trap, immediately acetylating the hydroxyl groups of the transient sugar species to stabilize them as the thermodynamically favored tetraacetyl derivative. This concerted mechanism prevents the reversion of the sugar to its free form or its degradation into polymeric tars, which is a common failure mode in less acidic environments. The result is a clean cleavage that preserves the stereochemical integrity of the beta-anomeric configuration, which is critical for the biological activity of downstream antiviral agents.
Impurity control is inherently managed through the solubility characteristics of the reaction components and the specific choice of recrystallization solvents post-reaction. The acetylated purine byproducts, such as acetyl xanthoglobulin or diacetyl vitamin B4, exhibit distinct solubility profiles that allow them to precipitate out of the reaction mixture upon cooling, effectively separating them from the ribose-containing filtrate. This physical separation minimizes cross-contamination and reduces the burden on subsequent purification columns. Furthermore, the use of catalysts that are fully soluble in the recrystallization media ensures that no residual solid acid remains trapped within the crystal lattice of the final product, thereby contributing to the reported purity levels of 99.5%. The careful optimization of the molar ratio between the nucleoside and acetic anhydride, typically maintained between 1:5 and 1:50, ensures that there is a sufficient excess of acetylating agent to drive the reaction to completion without generating excessive waste that would complicate solvent recovery systems.
How to Synthesize 1,2,3,5-O-Tetraacetyl-β-D-ribofuranose Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric balance of reagents and the precise control of thermal parameters to maximize yield and safety. The process begins with the suspension of the nucleoside starting material in acetic anhydride, followed by the slow addition of the catalytic acid under vigorous stirring to manage the exotherm. Maintaining the reaction temperature within the optimal window of 70°C to 120°C is crucial; temperatures that are too low may result in incomplete conversion, while excessive heat can promote charring and the formation of intractable tars. Once the reaction is deemed complete via TLC monitoring, the mixture is cooled to induce the crystallization of the purine byproduct, which is then removed by filtration. The remaining filtrate undergoes vacuum distillation to remove excess acetic anhydride and acetic acid, leaving behind a syrupy residue that is subsequently recrystallized from solvents such as ethyl acetate or isopropanol to yield the final white crystalline product. Detailed standardized synthetic steps see the guide below.
- Mix the nucleoside compound (inosine or adenosine) with acetic anhydride in a molar ratio of 1: 5 to 1:50 within a reaction vessel equipped with mechanical stirring.
- Add a catalytic amount (0.0001 to 0.1 molar ratio) of a strong acid catalyst such as trifluoroacetic acid or trifluoromethanesulfonic acid to the mixture.
- Heat the reaction mixture to 50-140°C until TLC indicates completion, then filter to separate the acetylated purine filter cake from the filtrate containing the ribose derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic cracking technology offers profound strategic benefits that extend far beyond simple yield improvements. The ability to utilize extremely low catalyst loadings, ranging from 0.0001 to 0.1 molar equivalents, translates directly into a drastic reduction in raw material costs and a significant simplification of waste treatment protocols. By eliminating the need for expensive solid superacids or complex resin regeneration cycles, manufacturers can achieve substantial cost savings in operational expenditures while simultaneously reducing the environmental footprint associated with hazardous waste disposal. The robustness of the process allows for the use of readily available industrial-grade acetic anhydride and common fluorinated acids, mitigating supply chain risks associated with specialized or scarce reagents. This reliability ensures that production schedules remain uninterrupted, providing a stable flow of critical intermediates to downstream API synthesis facilities without the volatility often seen with older, less predictable chemistries.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the minimal catalyst requirement, which removes the financial burden of purchasing and recovering high-value catalytic materials. Since the catalyst is used in trace amounts and is soluble, it does not require the capital-intensive filtration and washing equipment needed for heterogeneous catalysis, leading to lower CAPEX for reactor setups. Additionally, the high selectivity of the reaction minimizes the formation of side products, meaning that less raw material is wasted on unusable byproducts, effectively lowering the cost per kilogram of the final active pharmaceutical ingredient. The simplified workup procedure, which relies on basic filtration and distillation rather than complex chromatographic separations, further reduces labor costs and solvent consumption, creating a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic anhydride and trifluoroacetic acid ensures that the supply chain is resilient against market fluctuations that might affect niche reagents. Because the reaction conditions are moderate and do not require extreme pressures or cryogenic temperatures, the process can be easily scaled from pilot plants to multi-ton commercial reactors without significant re-engineering. This scalability guarantees that suppliers can meet sudden surges in demand for antiviral medications without facing the bottlenecks typical of more fragile synthetic routes. The consistent quality of the output, characterized by high purity and low impurity content, reduces the likelihood of batch rejections during quality control, ensuring a steady and dependable inventory for pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles by maximizing atom economy and minimizing waste generation. The absence of heavy metal catalysts or persistent organic pollutants simplifies the effluent treatment process, making it easier for facilities to comply with increasingly stringent environmental regulations. The ability to recycle the mother liquor from the recrystallization step further enhances the sustainability profile of the process, reducing the overall volume of organic solvents discharged. This compliance advantage not only protects the manufacturer from regulatory fines but also enhances the brand reputation among eco-conscious partners, positioning the company as a leader in sustainable pharmaceutical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this nucleoside cracking technology, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for R&D teams planning to integrate this process into their existing production lines or for quality assurance personnel establishing acceptance criteria for incoming intermediates. The answers reflect the specific performance metrics observed across multiple embodiments, highlighting the versatility of the catalyst system and the robustness of the purification strategy.
Q: What are the primary advantages of using trifluoroacetic acid over solid superacids for nucleoside cracking?
A: Unlike solid superacids which present significant challenges in industrial recovery and reuse, liquid acid catalysts like trifluoroacetic acid are homogeneous, allowing for easier handling and elimination of complex filtration steps associated with solid catalysts, thereby reducing operational complexity.
Q: How does this method improve the purity of 1,2,3,5-O-tetraacetyl-β-D-ribofuranose compared to traditional acetylation?
A: The patented method achieves purity levels up to 99.5% by utilizing specific recrystallization solvents like ethyl acetate or isopropanol after distillation, effectively removing colored impurities and side products that typically plague older methods like the Beranek protocol.
Q: Can this catalytic cracking process be adapted for both inosine and adenosine substrates?
A: Yes, the process is highly versatile and successfully converts both inosine into acetyl xanthoglobulin and adenosine into diacetyl vitamin B4, demonstrating broad substrate tolerance while maintaining high yields above 90% for the ribose component in both cases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,5-O-Tetraacetyl-β-D-ribofuranose Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent literature are faithfully reproduced in our state-of-the-art facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of tetraacetyl ribose meets the exacting standards required for antiviral drug synthesis. Our infrastructure is designed to handle the specific safety and handling requirements of fluorinated acid catalysts, guaranteeing a safe and efficient production environment that protects both our workforce and the integrity of your supply chain.
We invite you to collaborate with us to leverage this advanced catalytic cracking technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your bottom line while securing the high-quality intermediates your formulations depend on.
