Optimizing Fondaparinux Sodium Intermediate Production for Commercial Scale-Up
Optimizing Fondaparinux Sodium Intermediate Production for Commercial Scale-Up
The global demand for effective anticoagulants continues to drive innovation in the synthesis of complex carbohydrates, specifically regarding fondaparinux sodium. As a synthetic pentasaccharide inhibitor of Factor Xa, fondaparinux represents a critical therapeutic agent for preventing venous thrombosis. However, its intricate molecular architecture poses significant challenges for industrial manufacturing. Patent CN108690100B addresses these challenges by introducing a refined preparation method for key intermediates, specifically focusing on the synthesis and purification of the azido-sugar compound designated as Formula 3. This patent outlines a strategic approach to overcoming the historical bottlenecks associated with the polarity and physical state of these intermediates, offering a pathway to higher purity and improved process efficiency.
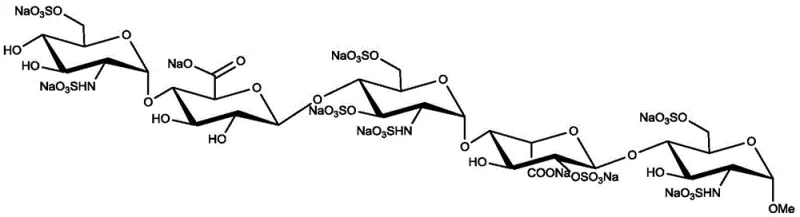
The significance of this technological advancement cannot be overstated for stakeholders in the pharmaceutical intermediates sector. The ability to produce high-quality precursors reliably is the cornerstone of a robust supply chain for life-saving medications. By optimizing the synthesis of the D5 skeleton and its precursors, this methodology directly impacts the feasibility of large-scale production, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards while maintaining economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the landscape of prior art, including references such as US4818816 and US8987430, the synthesis of fondaparinux sodium intermediates has historically been plagued by inefficiencies. A primary obstacle lies in the physical properties of the intermediate compound of Formula 3. In traditional synthetic routes, this compound typically manifests as a highly polar, oily substance. This physical state renders standard purification techniques, such as recrystallization, largely ineffective. Consequently, manufacturers are forced to rely on labor-intensive and solvent-heavy chromatographic separations to remove inorganic salts and organic byproducts. These limitations not only depress the overall yield of the synthesis but also introduce variability that complicates quality control and scale-up efforts.
The Novel Approach
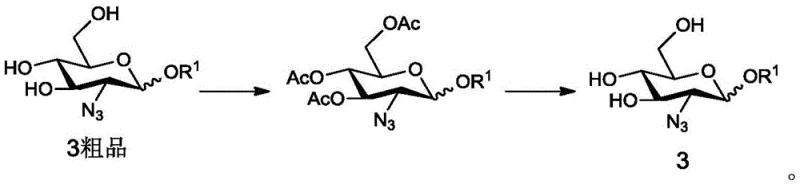
Patent CN108690100B introduces a transformative solution to this purification dilemma through the strategic use of temporary protecting groups. The core innovation involves converting the crude, oily Formula 3 into a protected derivative, specifically the acetylated compound known as Formula 5. Unlike its precursor, Formula 5 possesses physical properties that allow it to be easily purified, often through crystallization. This intermediate acts as a 'chemical filter,' trapping impurities or allowing them to be washed away effectively. Once the high-purity Formula 5 is isolated, it undergoes a straightforward deacetylation step to regenerate the target Formula 3 with exceptional purity. This detour, while adding a step, drastically simplifies the overall purification logic and enhances the final product quality.
Mechanistic Insights into Azide Introduction and Protecting Group Strategy
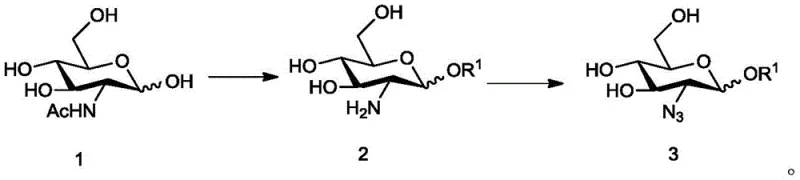
The chemical elegance of this process lies in the precise manipulation of functional groups on the glucosamine scaffold. The synthesis begins with the conversion of acetylglucosamine into a methyl glycoside, followed by the critical introduction of the azido group at the C-2 position. This diazotransfer reaction is pivotal, as the azide functionality is essential for the subsequent formation of the sulfonamido groups found in the final fondaparinux structure. The patent details the use of reagents such as imidazole-1-sulfonyl azide hydrochloride in the presence of copper sulfate and potassium carbonate to achieve this transformation efficiently. The reaction conditions are optimized to ensure high conversion rates while minimizing side reactions that could lead to difficult-to-remove impurities.
Furthermore, the mechanistic rationale for the acetylation-deacetylation cycle is rooted in solubility modulation. The hydroxyl groups on the sugar ring are highly hydrophilic, contributing to the oily nature of the free sugar. By masking these hydroxyls with acetyl groups (using acetic anhydride), the molecule becomes more lipophilic and structurally rigid, facilitating crystal lattice formation. This phase change from oil to solid is the key to the purification success. The deacetylation step, typically performed under basic conditions using sodium methoxide in methanol, is mild and selective, ensuring that the sensitive glycosidic bonds and the azido group remain intact while restoring the necessary hydroxyl functionality for downstream coupling reactions.
How to Synthesize Fondaparinux Sodium Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters and workup procedures to maximize the benefits of the purification strategy. The process is designed to be robust, utilizing commercially available reagents and standard unit operations familiar to process chemists. By adhering to the specific sequence of methanolysis, azidation, protective acetylation, and final deprotection, manufacturers can achieve purity levels that were previously difficult to attain without extensive chromatography. For a detailed breakdown of the standardized operational procedures, please refer to the technical guide below.
- Perform methanolysis on acetylglucosamine under acidic conditions followed by neutralization to obtain the free amine intermediate.
- Execute a diazotransfer reaction using imidazole-1-sulfonyl azide hydrochloride to introduce the critical azido functional group.
- Purify the crude azido-sugar by converting it into a crystallizable acetylated derivative (Formula 5), followed by deacetylation to yield high-purity Formula 3.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the methodology described in Patent CN108690100B offers tangible strategic benefits beyond mere chemical curiosity. The shift from chromatographic purification to crystallization-based purification fundamentally alters the cost structure and risk profile of the manufacturing process. This transition addresses several critical pain points in the supply of high-purity pharmaceutical intermediates, ensuring a more stable and cost-effective flow of materials for downstream drug production.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the elimination or drastic reduction of column chromatography steps. Chromatography is notoriously expensive due to the high consumption of silica gel, solvents, and energy, as well as the low throughput it offers. By enabling purification through crystallization of the acetylated intermediate, the process significantly lowers variable costs. Furthermore, the improved yield resulting from better impurity removal means less starting material is wasted, directly enhancing the cost-efficiency of the entire synthesis campaign.
- Enhanced Supply Chain Reliability: The robustness of a crystallization-based process translates directly to supply chain stability. Crystalline solids are generally easier to handle, store, and transport than oils, reducing the risk of degradation during logistics. Additionally, the ability to consistently produce intermediates with purity exceeding 95% reduces the likelihood of batch failures in subsequent synthesis steps. This reliability is crucial for maintaining continuous production schedules for the final API, mitigating the risk of shortages in the market for anticoagulant therapies.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior. Reducing solvent usage associated with chromatography aligns with green chemistry principles and lowers the burden on waste treatment facilities. The process is inherently more scalable because crystallization is a standard unit operation in chemical engineering that scales linearly, whereas chromatography often faces non-linear challenges at large volumes. This makes the technology ideal for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production can meet global demand without compromising environmental standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for partners evaluating its integration into their supply chains. The following questions address common inquiries regarding the purity, scalability, and specific chemical transformations involved in this patented process. These answers are derived directly from the experimental data and technical disclosures within the patent documentation.
Q: Why is the purification of Formula 3 considered a bottleneck in traditional fondaparinux synthesis?
A: Traditional methods often result in Formula 3 as a highly polar, oily substance that is extremely difficult to purify via recrystallization. This necessitates complex chromatographic separations which reduce overall yield and increase production costs significantly.
Q: How does the acetylation strategy in Patent CN108690100B improve product quality?
A: By temporarily protecting the hydroxyl groups with acetyl groups to form Formula 5, the intermediate becomes solid and crystallizable. This allows for effective removal of organic and inorganic impurities before the final deacetylation step, ensuring purity levels exceeding 95%.
Q: What are the scalability advantages of this new preparation method?
A: The process replaces difficult-to-scale chromatographic purifications with robust crystallization steps. This simplifies the workflow, reduces solvent consumption, and enhances the reproducibility required for industrial mass production of anticoagulant intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fondaparinux Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and manufacture of life-saving drugs like fondaparinux sodium. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and adhere to stringent purity specifications to ensure that every batch of intermediate we supply meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that we can navigate the complexities of carbohydrate chemistry with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to review specific COA data and route feasibility assessments for the compounds described in CN108690100B, we are ready to provide the data-driven insights necessary to drive your projects forward. Contact us today to secure a reliable supply of high-purity intermediates for your anticoagulant manufacturing programs.
