Industrial Scale-Up of Scopoletin: A Novel Synthetic Route for High-Purity Pharmaceutical Intermediates
Industrial Scale-Up of Scopoletin: A Novel Synthetic Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for producing bioactive coumarins, specifically Scopoletin (6-methoxy-7-hydroxycoumarin). Patent CN101519393A introduces a transformative approach to synthesizing this valuable intermediate, moving away from unreliable natural extraction towards a precise, chemically defined total synthesis. This innovation addresses the critical bottlenecks of traditional sourcing, such as low recovery rates from plant materials and the high costs associated with botanical processing. By leveraging a two-step sequence involving selective demethylation and subsequent cyclization, this technology offers a reliable pathway for generating high-purity Scopoletin suitable for downstream drug development.
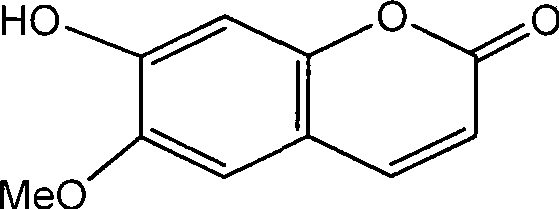
For R&D directors and procurement specialists, the shift from extraction to synthesis represents a pivotal opportunity to secure supply chains against agricultural volatility. The patent details a method that utilizes commodity chemicals like aluminum trichloride and ethyl triphenylphosphonoacetate, ensuring that raw material availability remains stable regardless of harvest seasons. This strategic pivot not only enhances the reproducibility of the active pharmaceutical ingredient (API) intermediate but also lays the groundwork for significant cost reduction in pharmaceutical intermediate manufacturing by eliminating the complex purification steps inherent to natural product isolation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Scopoletin has been plagued by inefficiencies inherent to both botanical extraction and early synthetic attempts. Traditional extraction from sources such as Swpolia japonica or Angelica dahurica is notoriously labor-intensive, yielding minimal product relative to the biomass processed, which drives up the unit cost prohibitively for large-scale applications. Furthermore, earlier synthetic routes, such as those reported by Crosby et al. in 1962, relied on difficult-to-prepare starting materials like 2,4-dihydroxyanisole and expensive reagents like 3,3-diethoxy ethyl propionate. These legacy methods often resulted in suboptimal yields and required cumbersome separation techniques that generated excessive chemical waste, rendering them economically unviable for modern industrial standards.
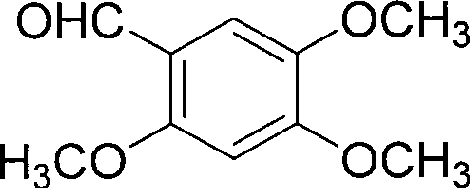
The Novel Approach
The methodology outlined in CN101519393A circumvents these historical hurdles by employing a streamlined, two-stage synthesis starting from the commercially accessible 2,4,5-trimethoxybenzaldehyde. This novel route replaces exotic reagents with standard industrial chemicals, utilizing aluminum trichloride for efficient demethylation and ethyl triphenylphosphonoacetate for ring closure. By optimizing reaction conditions, specifically temperature and solvent systems, the inventors have created a process that is not only chemically elegant but also operationally simple. This approach eliminates the need for specialized microwave equipment found in other recent attempts, thereby facilitating immediate adoption in standard chemical reactors found in global manufacturing facilities.
Mechanistic Insights into Lewis Acid Demethylation and Cyclization
The core of this synthetic strategy lies in the precise control of functional group transformations, beginning with the selective demethylation of the trimethoxy precursor. In this step, aluminum trichloride acts as a potent Lewis acid, coordinating with the methoxy groups to facilitate their cleavage under mild thermal conditions (30°C–50°C). The addition of TEBA (triethylbenzyl ammonium chloride) serves as a phase transfer catalyst, enhancing the interaction between the organic substrate and the inorganic salt in the dichloromethane medium. This careful orchestration ensures that only the specific methoxy groups at the 2 and 4 positions are removed, preserving the 5-methoxy group essential for the final Scopoletin structure, thus demonstrating exceptional regioselectivity.
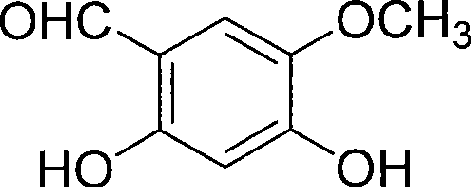
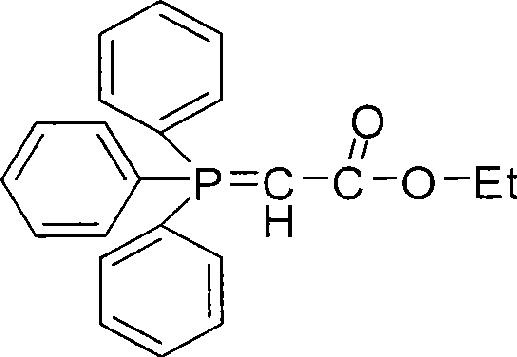
Following the formation of the dihydroxy intermediate, the process advances to a high-temperature Wittig-Horner type cyclization. The intermediate aldehyde reacts with ethyl triphenylphosphonoacetate in N,N-dimethylaniline, a high-boiling solvent capable of sustaining the rigorous 160°C–195°C temperatures required for ring closure. This thermal energy drives the condensation and subsequent lactonization to form the coumarin core. The mechanism inherently favors the formation of the thermodynamically stable aromatic system, minimizing the generation of open-chain byproducts. Rigorous purification protocols, including acid washes and recrystallization, are integrated to remove triphenylphosphine oxide and residual solvents, ensuring the final product meets stringent purity specifications.
How to Synthesize Scopoletin Efficiently
The synthesis of Scopoletin via this patented route is designed for operational clarity and safety, making it an ideal candidate for technology transfer to pilot and production plants. The process begins with the preparation of the key aldehyde intermediate, followed by the thermal cyclization step which constructs the heterocyclic ring system. Detailed standard operating procedures regarding reagent stoichiometry, temperature ramping, and work-up sequences are critical for maximizing yield and minimizing impurities. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Perform selective demethylation of 2,4,5-trimethoxybenzaldehyde using aluminum trichloride and TEBA in dry dichloromethane to yield 2,4-dihydroxy-5-methoxybenzaldehyde.
- Execute high-temperature cyclization of the intermediate aldehyde with ethyl triphenylphosphonoacetate in N,N-dimethylaniline under nitrogen protection.
- Purify the crude product through solvent extraction, column chromatography, and recrystallization to achieve high-purity Scopoletin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for supply chain stability and cost management. By decoupling production from agricultural cycles, manufacturers can guarantee continuous availability of Scopoletin, mitigating the risks of crop failure or seasonal price fluctuations that plague natural extract markets. The reliance on bulk commodity chemicals rather than specialized, low-volume reagents further insulates the production cost from market volatility. This stability allows procurement managers to negotiate long-term contracts with greater confidence, knowing that the underlying manufacturing process is robust, scalable, and less susceptible to external disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive natural extraction processes and the use of cost-effective reagents like aluminum trichloride significantly lower the overall cost of goods sold. By avoiding the need for complex microwave reactors and utilizing standard heating methods, capital expenditure for equipment is also minimized. The ability to recover and reuse high-boiling solvents like N,N-dimethylaniline further contributes to substantial cost savings over the lifecycle of the production campaign.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as 2,4,5-trimethoxybenzaldehyde and phosphonate esters are widely available from global chemical suppliers. This abundance ensures that production schedules are not delayed by material shortages. Furthermore, the synthetic nature of the process allows for rapid scale-up from kilogram to multi-ton quantities without the lead times associated with cultivating and harvesting botanical sources, ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily managed in large-scale stirred tank reactors. The work-up procedures, which involve standard extraction and crystallization techniques, are well-understood by plant operators, reducing the risk of batch failures. Additionally, the structured waste management protocols, including the neutralization of acidic byproducts and solvent recovery, align with modern environmental compliance standards, reducing the burden of hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Scopoletin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality attributes of the process for potential partners and licensees.
Q: How does this synthetic route compare to natural extraction in terms of yield?
A: Unlike natural extraction from plants like Swpolia japonica which suffers from low yields and seasonal variability, this synthetic method offers a consistent, scalable production pathway with significantly improved efficiency and purity control.
Q: What are the key advantages of using N,N-dimethylaniline as the solvent?
A: N,N-dimethylaniline serves as a high-boiling point solvent that facilitates the rigorous thermal conditions required for the cyclization step, ensuring complete reaction conversion while allowing for solvent recovery and reuse to minimize waste.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly designs the process for industrialization by utilizing readily available raw materials like aluminum trichloride and avoiding complex microwave equipment, making it highly adaptable for multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scopoletin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Scopoletin meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and reliability for your organization.
