Advanced Synthesis of Bosutinib 1,3-Propylene Diether Dimer Impurity for Quality Control
Introduction to Advanced Impurity Profiling for Bosutinib
The rigorous control of impurity profiles is a cornerstone of modern pharmaceutical development, particularly for potent kinase inhibitors like Bosutinib (SKI-606). As detailed in patent CN112552236A, the identification and synthesis of specific process-related impurities are critical for ensuring drug safety and efficacy. This patent introduces a novel, high-efficiency synthesis method for the Bosutinib 1,3-propylene diether dimer impurity, a potential byproduct that must be strictly monitored. The ability to synthesize this specific reference standard with high purity allows manufacturers to validate their analytical methods and ensure compliance with global regulatory standards.
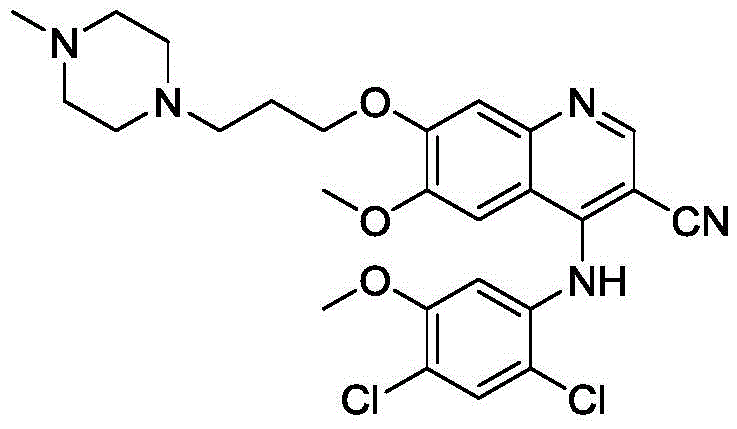
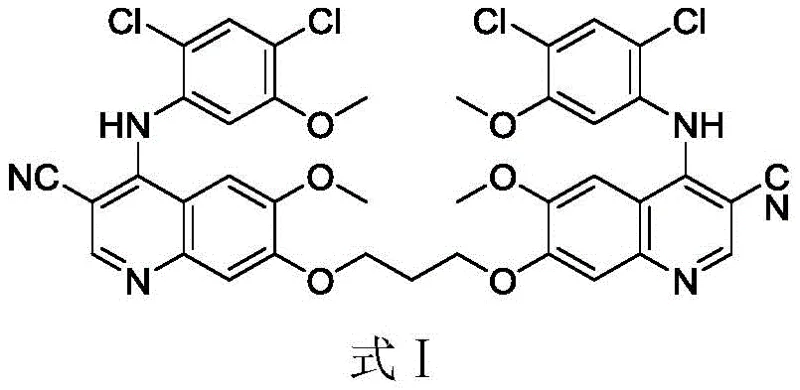
The target molecule, identified as Formula I in the patent documentation, represents a dimeric structure formed via a propylene linker, which can theoretically arise during the etherification steps of the main Bosutinib synthesis. Having access to an authentic sample of this impurity is essential for spiking studies and limit testing. The disclosed method addresses the industry need for reliable sources of such complex reference materials, utilizing a streamlined five-step sequence that prioritizes yield and purity over traditional, more convoluted approaches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining specific dimeric impurities for drugs like Bosutinib has been a significant challenge for quality control laboratories. In standard manufacturing processes, such impurities often appear only in trace amounts, making isolation and purification for characterization extremely difficult and cost-prohibitive. Conventional attempts to synthesize these structures often suffer from poor regioselectivity during the etherification stage, leading to complex mixtures of mono- and di-substituted products that require extensive chromatographic separation. Furthermore, older synthetic routes may rely on harsh conditions or unstable intermediates that degrade the final product, resulting in low overall yields and insufficient purity for use as certified reference standards.
The Novel Approach
The methodology presented in CN112552236A offers a decisive break from these limitations by employing a convergent synthesis strategy designed specifically for the target dimer. Instead of relying on random formation during API production, this route constructs the quinoline core first and then introduces the dimeric linker in a controlled final step. By utilizing 1,3-dibromopropane as a bridging agent in the presence of potassium carbonate, the process ensures the precise formation of the 1,3-propylene diether linkage. This targeted approach not only simplifies the purification workflow but also guarantees that the resulting material is structurally identical to the process impurity found in Bosutinib manufacturing, providing a robust tool for quality assurance.
Mechanistic Insights into Phosphorus Oxychloride-Mediated Cyclization
A critical juncture in this synthesis is the formation of the quinoline ring system, achieved through a Vilsmeier-Haack type cyclization using phosphorus oxychloride (POCl3). In step 4 of the process, the enamine intermediate (Formula VI) undergoes cyclization in sulfolane at elevated temperatures ranging from 100°C to 110°C. Sulfolane acts as a high-boiling, polar aprotic solvent that stabilizes the charged intermediates formed during the electrophilic attack of the imidoyl chloride species on the aromatic ring. This specific solvent choice is crucial for driving the reaction to completion without decomposing the sensitive cyano and chloro substituents present on the molecule.
Following the cyclization, the reaction mixture is carefully quenched with aqueous potassium hydroxide at low temperatures (0-5°C) to hydrolyze the imidoyl chloride to the corresponding ketone or aldehyde functionality, which then tautomerizes to the stable quinoline structure (Formula VII). The pH is subsequently adjusted to 5-6 to precipitate the product, a technique that leverages the acid-base properties of the quinoline nitrogen to achieve high recovery rates. This meticulous control over temperature and pH during the workup phase is instrumental in minimizing the formation of hydrolysis byproducts, thereby ensuring the high purity (>99%) reported in the patent examples.
How to Synthesize Bosutinib Dimer Impurity Efficiently
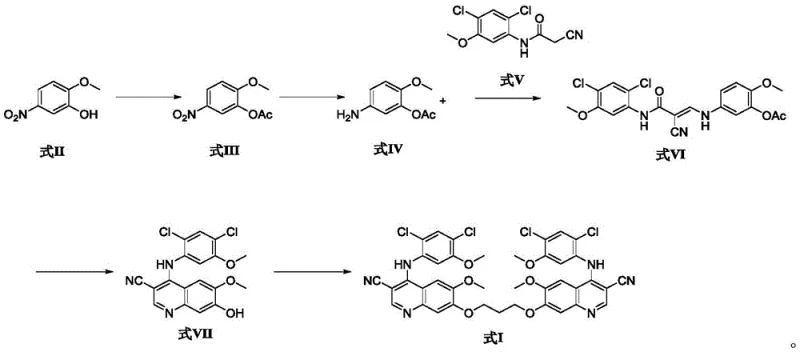
The synthesis of this complex pharmaceutical intermediate requires precise adherence to reaction stoichiometry and thermal conditions to maximize yield. The process begins with the protection of the phenolic hydroxyl group, followed by reduction and condensation to build the core scaffold. The final coupling step utilizes a Williamson ether synthesis mechanism to link two quinoline units. For a complete breakdown of the specific molar ratios, solvent volumes, and workup procedures required to replicate this high-purity synthesis, please refer to the standardized guide below.
- Esterification of 2-methoxy-4-nitrophenol with acetic anhydride using DMAP and triethylamine to form the acetylated intermediate.
- Catalytic reduction of the nitro group to an amine using Pd/C and ammonium formate in isopropanol/THF solvent system.
- Condensation with 2-cyano-N-(2,4-dichloro-5-methoxyphenyl)acetamide using triethyl orthoformate to form the enamine intermediate.
- Cyclization using phosphorus oxychloride in sulfolane at elevated temperatures followed by hydrolysis to form the quinoline core.
- Final coupling with 1,3-dibromopropane and potassium carbonate in DMF to generate the target dimer impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals such as acetic anhydride, triethylamine, and 1,3-dibromopropane significantly de-risks the supply chain, as these materials are globally available from multiple vendors. This reduces the dependency on single-source specialty reagents that often lead to production bottlenecks. Furthermore, the elimination of complex transition metal catalysis in the final coupling step simplifies the downstream processing, removing the need for expensive metal scavenging resins and reducing the environmental footprint associated with heavy metal waste disposal.
- Cost Reduction in Manufacturing: The process achieves substantial cost optimization by utilizing high-yielding reactions that minimize raw material waste. For instance, the reduction step using ammonium formate and Pd/C is highly efficient, avoiding the need for high-pressure hydrogenation equipment which entails higher capital expenditure and safety costs. Additionally, the use of simple precipitation and filtration for product isolation in multiple steps reduces the consumption of large volumes of chromatography solvents, directly lowering the operational expenditure associated with solvent recovery and waste treatment.
- Enhanced Supply Chain Reliability: By establishing a robust in-house or contract manufacturing route for this critical impurity standard, pharmaceutical companies can insulate themselves from external supply shocks. The synthetic pathway is linear and predictable, allowing for accurate forecasting of lead times. The stability of the intermediates, such as the acetylated nitro compound and the enamine, permits the stocking of key precursors, enabling a rapid response to increased quality control demands without waiting for long synthesis cycles to complete from scratch.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram batches with consistent results. The use of sulfolane, while requiring careful handling, allows for high concentration reactions which improves reactor throughput. Moreover, the aqueous workups described in the patent facilitate the separation of organic byproducts into waste streams that are easier to treat, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific Bosutinib impurity. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring that the information is grounded in verified chemical reality rather than theoretical speculation.
Q: What is the primary advantage of this synthesis method for Bosutinib impurities?
A: The primary advantage is the ability to produce the specific 1,3-propylene diether dimer impurity with extremely high purity (>99%) using simple, commercially available raw materials. This allows pharmaceutical manufacturers to establish accurate quality control standards and impurity profiles required by regulatory bodies like the FDA.
Q: How does the cyclization step ensure product quality?
A: The cyclization step utilizes phosphorus oxychloride in sulfolane at controlled temperatures (100-110°C). This specific solvent and temperature combination facilitates efficient ring closure to form the quinoline core while minimizing side reactions, ensuring the structural integrity required for a certified reference standard.
Q: Is this process suitable for large-scale production of reference standards?
A: Yes, the process is designed for scalability. It avoids exotic catalysts and uses standard unit operations such as filtration, solvent evaporation, and crystallization. The use of robust reagents like potassium carbonate and common solvents like DMF and THF makes it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bosutinib Impurity Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your drug substance relies on the quality of your reference standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for method validation or kilograms for stability studies, we can deliver. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Bosutinib 1,3-propylene diether dimer impurity meets the exacting standards required by global regulatory agencies.
We invite you to contact our technical procurement team to discuss your specific requirements. We are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments. Let us support your quality control objectives with reliable supply and technical excellence.
