Advanced Synthetic Route for High-Purity Sofosbuvir Impurity Standards
Advanced Synthetic Route for High-Purity Sofosbuvir Impurity Standards
The pharmaceutical industry's relentless pursuit of safety and efficacy mandates the availability of ultra-high purity reference standards for every Active Pharmaceutical Ingredient (API). Patent CN111848665B discloses a sophisticated synthetic methodology specifically designed for the preparation of Sofosbuvir impurities, which serve as critical control substances in the quality assurance of this vital antiviral medication. This technology addresses the complex challenge of regioselective functionalization on a highly sensitive lactone scaffold, utilizing a strategic sequence of orthogonal protection and deprotection steps. By implementing this novel approach, manufacturers can secure a reliable supply of characterized impurities that are essential for validating analytical methods and ensuring batch-to-batch consistency in drug manufacturing. The method described herein represents a significant advancement over traditional non-selective synthesis routes, offering a pathway to obtain target molecules with exceptional structural fidelity and minimal byproduct formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating nucleoside analog impurities often suffer from a lack of regioselectivity, particularly when dealing with polyhydroxylated intermediates. In conventional processes, attempting to differentiate between primary and secondary hydroxyl groups on a furanose or lactone ring frequently results in statistical mixtures of mono-protected, di-protected, and unprotected species. This lack of control necessitates extensive and costly downstream purification efforts, such as repeated recrystallizations or preparative HPLC, which drastically reduce overall process efficiency and yield. Furthermore, harsh reaction conditions employed in older methodologies can lead to epimerization or degradation of the sensitive lactone ring, introducing unknown impurities that complicate the validation of the final reference standard. These inefficiencies not only inflate the cost of goods but also pose significant risks to supply chain stability due to the difficulty in reproducing exact impurity profiles consistently across different production batches.
The Novel Approach
The methodology outlined in patent CN111848665B overcomes these historical hurdles through a meticulously designed orthogonal protection strategy that leverages the distinct chemical reactivity of silyl and acyl protecting groups. By initially installing a tert-butyldimethylsilyl (TBS) group on the terminal hydroxyl, followed by a second TBS group on the ring hydroxyl, the process creates a fully protected intermediate that can be manipulated with precision. The true innovation lies in the subsequent selective deprotection steps, where specific conditions are applied to remove the terminal silyl group while leaving the ring protection intact, or vice versa. This level of control allows for the sequential installation of a benzoyl group at a specific position, ultimately yielding the target impurity structure with high regiochemical purity. This approach effectively eliminates the formation of difficult-to-separate isomers, streamlining the purification workflow and ensuring that the final product meets the stringent purity specifications required for pharmaceutical reference materials.
Mechanistic Insights into Orthogonal Protection and Deprotection
The core of this synthetic success relies on the differential stability of the protecting groups under specific chemical environments. The initial silylation steps utilize tert-butyldimethylsilyl chloride (TBSCl) in the presence of imidazole and pyridine, which act as both base and nucleophilic catalyst to facilitate the formation of the silyl ether bonds. The choice of TBS is strategic; it is robust enough to withstand subsequent reaction conditions yet labile enough to be removed selectively. For instance, the selective removal of the terminal TBS group is achieved using hydrochloric acid in tetrahydrofuran at low temperatures. This acidic hydrolysis exploits the steric and electronic differences between the primary and secondary silyl ethers, allowing the primary terminal group to be cleaved preferentially while the more hindered ring silyl ether remains intact. This mechanistic nuance is critical for exposing the specific hydroxyl group required for the next transformation without disturbing the rest of the molecular architecture.
Following the selective deprotection, the exposed terminal hydroxyl is subjected to benzoylation using benzoyl chloride and a catalytic amount of DMAP (4-dimethylaminopyridine). The introduction of the benzoyl group serves as a permanent marker for that position, as it is stable to the fluoride conditions used in the final step. The culmination of the synthesis involves the global removal of the remaining ring TBS group using tetrabutylammonium fluoride (TBAF). Fluoride ions have a high affinity for silicon, driving the cleavage of the Si-O bond to regenerate the free hydroxyl group on the ring. This final deprotection step reveals the target Sofosbuvir impurity structure, completing the transformation from a fully protected precursor to the desired analytical standard. The entire sequence demonstrates a masterful command of organic synthesis principles to achieve a result that is difficult to obtain through direct functionalization.
How to Synthesize Sofosbuvir Impurity Efficiently
The synthesis of this high-value pharmaceutical intermediate requires precise control over reaction stoichiometry, temperature, and workup procedures to maximize yield and purity. The process begins with the dissolution of the starting lactone compound in a mixture of dichloromethane and pyridine, followed by the careful addition of silylating agents under ice-bath conditions to manage exothermicity. Subsequent steps involve rigorous aqueous workups using saturated sodium bicarbonate and brine to remove acidic byproducts and excess reagents, ensuring the integrity of the acid-sensitive lactone ring. Purification is primarily achieved through silica gel column chromatography using gradient elution systems of petroleum ether and ethyl acetate, which effectively separate the desired intermediates from unreacted starting materials and side products. For a detailed, step-by-step guide including exact molar ratios, solvent volumes, and specific isolation techniques, please refer to the standardized protocol below.
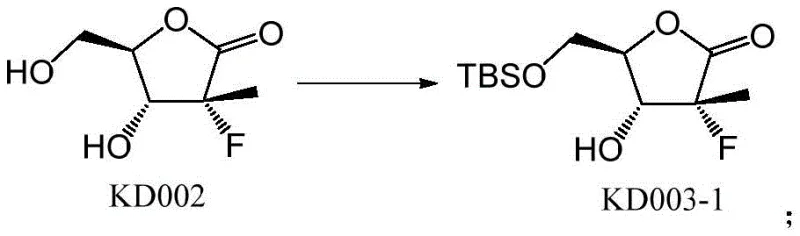
- Protect the terminal hydroxyl group of the starting lactone compound with a tert-butyldimethylsilyl (TBS) group using TBSCl and imidazole.
- Protect the cyclic hydroxyl group with a second TBS group to form a di-silylated intermediate.
- Selectively remove the terminal TBS group using acidic conditions (HCl in THF) to regenerate the terminal hydroxyl.
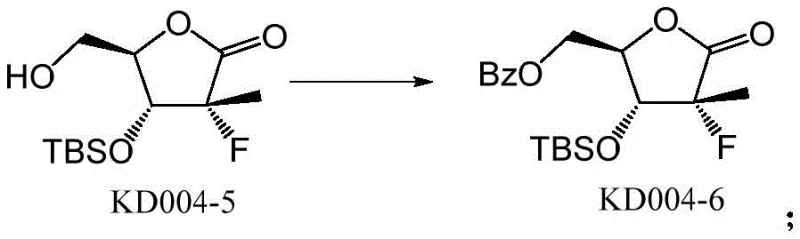
- Protect the regenerated terminal hydroxyl with a benzoyl (Bz) group using benzoyl chloride.
- Remove the remaining cyclic TBS group using tetrabutylammonium fluoride (TBAF) to yield the final high-purity impurity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthetic route offers substantial advantages for procurement managers and supply chain directors focused on cost optimization and reliability. The ability to synthesize complex impurity standards with high regioselectivity translates directly into reduced manufacturing costs by minimizing the loss of valuable starting materials to unwanted isomers. Traditional methods that produce mixtures require expensive and time-consuming separation technologies, whereas this orthogonal strategy delivers the target molecule with inherent purity, significantly simplifying the downstream processing requirements. This efficiency gain allows for a more predictable production schedule and reduces the dependency on specialized chromatographic equipment that can become a bottleneck in large-scale operations. Consequently, the overall cost of producing these critical reference materials is lowered, making high-quality quality control more accessible for generic drug manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex separation processes for isomeric byproducts leads to a drastic simplification of the production workflow. By avoiding the need for repetitive recrystallization or preparative HPLC to remove structurally similar impurities, the process consumes fewer solvents and stationary phases, which are significant cost drivers in fine chemical synthesis. Furthermore, the high yield observed in the key coupling and deprotection steps ensures that the consumption of expensive reagents like TBSCl and TBAF is optimized, directly contributing to a lower cost per gram of the final product. This economic efficiency is vital for maintaining competitive pricing in the highly regulated market of pharmaceutical reference standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dichloromethane, pyridine, imidazole, and benzoyl chloride ensures that the supply chain is robust and resistant to disruptions. Unlike processes that depend on exotic catalysts or custom-synthesized reagents with long lead times, this method utilizes widely available industrial solvents and reagents that can be sourced from multiple vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Additionally, the scalability of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard ice baths, facilitates easy technology transfer from laboratory to pilot plant, ensuring a continuous and reliable flow of materials to meet market demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as liquid-liquid extraction and column chromatography that are well-understood in industrial settings. The waste streams generated are primarily aqueous salts and organic solvents that can be managed through established recovery and treatment protocols, aligning with modern environmental compliance standards. The high atom economy of the protection-deprotection sequence minimizes the generation of hazardous waste compared to less selective routes that produce large quantities of unusable byproducts. This environmental stewardship not only reduces disposal costs but also enhances the sustainability profile of the manufacturing operation, a key consideration for multinational corporations aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Sofosbuvir impurities synthesized via this advanced methodology. Understanding these details is crucial for R&D teams integrating these standards into their analytical workflows and for procurement specialists evaluating supplier capabilities. The answers provided are derived directly from the technical specifications and experimental data disclosed in the underlying patent literature, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: Why is orthogonal protection critical for synthesizing Sofosbuvir impurities?
A: Orthogonal protection allows for the selective modification of specific hydroxyl groups on the lactone ring without affecting others. This precision is essential for generating specific structural isomers required as reference standards, ensuring that the impurity profile matches regulatory requirements for antiviral drug quality control.
Q: What purity levels can be achieved with this synthetic method?
A: The patented method utilizes rigorous purification steps, including column chromatography and selective crystallization, to achieve purity levels exceeding 99.5%. This high degree of purity is critical for analytical reference standards used in HPLC and GC assays to detect trace impurities in the final API.
Q: Is this synthesis scalable for commercial supply of reference materials?
A: Yes, the process employs robust reagents like TBSCl, benzoyl chloride, and TBAF which are readily available on an industrial scale. The reaction conditions, such as ice-bath cooling and ambient temperature stirring, are easily transferable from laboratory to pilot and commercial production scales, ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sofosbuvir Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the quality of the reference standards used to test them. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch of Sofosbuvir impurity we supply. Our commitment to excellence means that you receive materials that are fully characterized and ready for immediate use in your regulatory filings and quality control assays, providing you with peace of mind and confidence in your data.
We invite you to contact our technical procurement team to discuss your specific requirements for Sofosbuvir impurities and other pharmaceutical intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how our optimized synthetic routes can reduce your overall procurement expenses. We encourage you to reach out today to request specific COA data and route feasibility assessments tailored to your project needs, allowing us to demonstrate how our technical expertise can support your supply chain goals and accelerate your time to market.
