Advanced Green Synthesis of Naproxen Intermediates via Resin-Mediated Heck Coupling for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for the production of critical non-steroidal anti-inflammatory drug (NSAID) precursors. Patent CN101161617A introduces a transformative methodology for synthesizing 2-(6'-methoxy-2'-naphthyl) allyl alcohol, a pivotal intermediate in the manufacturing of Naproxen. This innovation addresses long-standing challenges in process chemistry by leveraging a palladium-catalyzed Heck reaction mediated by a macroporous weak base styrene type anion-exchange resin. Unlike conventional methods that rely on hazardous oxidants or difficult-to-remove homogeneous bases, this technique operates in aprotic polar solvents at moderate temperatures ranging from 50°C to 150°C. The strategic integration of a solid-phase base not only facilitates the reaction mechanism but also allows for the seamless recovery and regeneration of the catalytic support, marking a significant leap forward in green chemistry applications for high-volume API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(6'-methoxy-2'-naphthyl) vinyl carbinol has been plagued by inefficient and environmentally detrimental processes. Early methodologies, such as those described in U.S. Pat 5286902, initiated from 2-methoxy-6-isopropyl naphthalene, requiring high-temperature tubular reactors for dehydrogenation followed by oxidation with tin anhydride. This multi-step sequence suffers from severe operational complexity, the use of highly toxic tin reagents, and the inherent difficulty in sourcing the specific isopropyl naphthalene starting material. Furthermore, traditional Heck coupling strategies typically necessitate the use of stoichiometric amounts of organic amines or inorganic bases alongside expensive phosphine ligands to maintain catalytic activity. These homogeneous additives create substantial downstream processing burdens, as they are difficult to separate from the product stream, leading to increased waste generation and higher purification costs that negatively impact the overall economic viability of large-scale manufacturing operations.
The Novel Approach
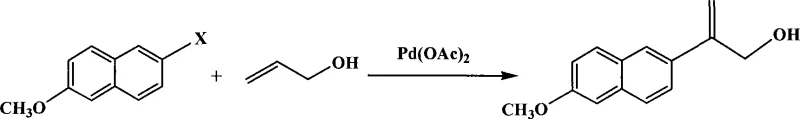
The methodology disclosed in the patent data presents a streamlined alternative that fundamentally reengineers the reaction environment to enhance efficiency and sustainability. By employing 2-halo-6-methoxynaphthalene and allyl alcohol as direct starting materials, the process bypasses the need for complex precursor synthesis. The core innovation lies in the substitution of soluble bases with a macroporous weak base styrene type anion-exchange resin, specifically types such as Styrene-DVB D301. This solid support acts as a proton scavenger during the catalytic cycle while remaining physically distinct from the reaction solution, enabling simple filtration for removal. Additionally, the system operates effectively without phosphine ligands, utilizing only palladium acetate as the catalyst source. This ligand-free condition significantly reduces raw material costs and eliminates the risk of phosphorus contamination in the final product, ensuring a cleaner profile suitable for stringent pharmaceutical quality standards.
Mechanistic Insights into Resin-Mediated Palladium Catalysis
The catalytic cycle driving this transformation adheres to the fundamental principles of the Heck reaction but is uniquely modified by the heterogeneous nature of the base. The cycle initiates with the oxidative addition of the aryl halide (where X is chlorine, bromine, or iodine) to the active palladium(0) species generated in situ from palladium acetate. Following this, the allyl alcohol coordinates and inserts into the palladium-carbon bond, forming a new carbon-carbon linkage at the 2-position of the naphthalene ring. The critical role of the macroporous weak base resin becomes apparent during the beta-hydride elimination step, where the resin neutralizes the hydrogen halide byproduct formed, driving the equilibrium forward and regenerating the active palladium catalyst. This heterogeneous base interaction prevents the accumulation of acidic species that could otherwise deactivate the catalyst or promote side reactions, thereby maintaining high turnover numbers throughout the reaction duration of 1 to 50 hours.
Impurity control in this system is inherently superior due to the absence of soluble amine bases and phosphine ligands which often lead to complex byproduct profiles. In traditional homogeneous systems, residual amines can react with electrophilic intermediates or coordinate strongly with the metal, requiring aggressive acid washes or chromatographic steps for removal. Here, the solid resin remains intact and is removed via simple filtration prior to workup, leaving a reaction mixture primarily containing the product, solvent, and trace palladium. The use of aprotic polar solvents like dimethylformamide, dimethyl sulfoxide, or 1-methyl-2-pyrrolidone ensures excellent solubility of the organic substrates while stabilizing the ionic transition states. This combination of factors results in a simplified purification workflow where the crude product can be isolated via extraction and concentration, significantly reducing the potential for impurity carryover into the final API intermediate.
How to Synthesize 2-(6'-Methoxy-2'-naphthyl) Allyl Alcohol Efficiently
The execution of this synthesis requires precise control over stoichiometry and thermal conditions to maximize yield while minimizing palladium loading. The patent outlines a robust protocol where the molar ratio of the aryl halide to allyl alcohol is maintained between 1:1 and 1:5, with a preferred ratio of 1:2 to ensure complete conversion of the more valuable halide substrate. The catalyst loading is remarkably low, ranging from 0.005 to 0.01 equivalents of palladium acetate, demonstrating the high efficiency of the ligand-free system. Detailed standard operating procedures regarding temperature ramping, agitation speeds, and specific workup techniques including toluene extraction and column chromatography are essential for replicating the reported yields of up to 90%. For a comprehensive guide on the exact step-by-step laboratory and pilot plant procedures, please refer to the standardized synthesis instructions provided below.
- Combine 2-halo-6-methoxynaphthalene and allyl alcohol in an aprotic polar solvent such as dimethylformamide or dimethyl sulfoxide.
- Add palladium acetate catalyst and macroporous weak base styrene type anion-exchange resin to the reaction mixture under stirring.
- Heat the mixture between 50°C and 150°C for 1 to 50 hours, then separate the resin, extract the product, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-mediated Heck reaction offers profound strategic benefits beyond mere technical feasibility. The elimination of expensive phosphine ligands and the reduction in palladium loading directly translate to a significant reduction in raw material expenditure per kilogram of produced intermediate. Furthermore, the ability to recover and regenerate the anion-exchange resin multiple times creates a circular economy within the manufacturing process, drastically lowering the volume of solid chemical waste requiring disposal. This reduction in waste handling not only cuts operational costs but also simplifies regulatory compliance regarding environmental discharge, making the supply chain more resilient against tightening global environmental regulations.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of high-cost auxiliary reagents. Traditional Heck couplings often require specialized, air-sensitive ligands that command premium pricing and necessitate inert atmosphere handling, increasing both material and infrastructure costs. By utilizing a ligand-free palladium system supported by a reusable polymeric resin, the process eliminates these cost centers entirely. Additionally, the simplified workup procedure reduces the consumption of solvents and energy associated with extensive purification steps, leading to substantial overall cost savings in the manufacturing of complex pharmaceutical intermediates without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity-grade starting materials and catalysts. The aryl halides and allyl alcohol are widely available bulk chemicals, reducing the risk of supply bottlenecks associated with custom-synthesized precursors. Moreover, the robustness of the catalyst system, which tolerates a range of temperatures and solvent conditions, ensures consistent production output even with minor variations in feedstock quality. The regenerable nature of the resin base further secures the supply chain by reducing dependency on single-use consumables, allowing manufacturers to maintain steady production schedules with lower inventory requirements for auxiliary chemicals.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the heterogeneous nature of the reaction components. The solid resin can be easily managed in large-scale reactors using standard filtration equipment, avoiding the emulsions and separation difficulties often encountered with liquid-liquid extractions of amine salts. From an environmental perspective, the avoidance of toxic tin oxidants and volatile organic amines aligns with green chemistry principles, reducing the facility's environmental footprint. This compliance advantage minimizes the risk of production shutdowns due to environmental violations and positions the manufacturer as a responsible partner for multinational pharmaceutical clients prioritizing sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits and chemical mechanisms for potential partners. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: What are the primary advantages of using anion-exchange resin over traditional organic amines in this Heck reaction?
A: The use of macroporous weak base styrene type anion-exchange resin eliminates the need for soluble organic amines or inorganic bases, which are often difficult to recover and generate significant wastewater pollution. This solid-phase base can be easily separated by filtration and regenerated for reuse, drastically simplifying the downstream purification process and reducing environmental compliance costs associated with amine disposal.
Q: Does this novel catalytic system require expensive phosphine ligands?
A: No, one of the most significant technical breakthroughs of this method described in patent CN101161617A is the ability to operate effectively without phosphine ligands. Traditional Heck reactions often rely on costly and air-sensitive phosphine ligands to stabilize the palladium catalyst; removing these ligands not only lowers the raw material cost but also prevents phosphorus contamination in the final pharmaceutical intermediate, thereby enhancing product purity and simplifying metal removal steps.
Q: How does this method compare to the traditional dehydrogenation-tin oxidation route for Naproxen precursors?
A: The traditional route involving dehydrogenation of isopropyl naphthalene followed by tin anhydride oxidation is characterized by severe reaction conditions, toxic reagents, and low yields. In contrast, this direct Heck coupling approach utilizes readily available halo-methoxynaphthalenes and allyl alcohol under milder conditions, achieving significantly higher yields (up to 90% in optimized examples) while avoiding the use of highly toxic tin oxidants, making it far more suitable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(6'-Methoxy-2'-naphthyl) Allyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable pathways for producing high-value pharmaceutical intermediates like 2-(6'-methoxy-2'-naphthyl) allyl alcohol. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative resin-mediated Heck reaction described in CN101161617A can be seamlessly translated from benchtop success to industrial reality. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for downstream API synthesis, providing our partners with a secure and high-quality supply foundation.
We invite global pharmaceutical and fine chemical enterprises to collaborate with us to leverage this advanced technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your production costs and environmental impact while securing a reliable source of this essential Naproxen precursor.
