Advanced Photocatalytic Migration Strategy for Scalable Baclofen Production
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing, driven by the urgent need to reduce environmental footprints and operational costs. A groundbreaking development in this arena is detailed in Chinese Patent CN114685298A, which discloses a novel method for synthesizing baclofen, a critical GABA_B receptor agonist used widely in the treatment of spasticity. This patent introduces a sophisticated photocatalytic migration strategy that fundamentally重构 s the construction of the baclofen core skeleton. Unlike traditional routes that rely on harsh thermal conditions and toxic reagents, this innovation leverages visible-light-mediated photoredox catalysis to achieve a phenyl migration rearrangement. For R&D directors and procurement specialists seeking a reliable baclofen intermediate supplier, this technology represents a significant leap forward, offering a pathway to high-purity products with drastically simplified downstream processing and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of baclofen has been plagued by significant technical and safety challenges that hinder efficient cost reduction in pharmaceutical manufacturing. One prevalent legacy route involves a Knoevenagel-Doebner condensation between p-chlorobenzaldehyde and malonic acid, utilizing pyridine as both solvent and catalyst. This step is notoriously problematic due to the strong, unpleasant odor and inherent toxicity of pyridine, creating severe occupational health hazards and complicating waste management. Furthermore, subsequent steps often require cryogenic conditions, such as Michael additions performed at temperatures as low as -78°C, which impose a massive energy burden on the facility. The final stages typically necessitate high-pressure catalytic hydrogenation, demanding expensive, specialized reactor equipment and introducing substantial safety risks associated with handling hydrogen gas at scale. These cumulative factors result in a lengthy, step-economically poor process with a high total cost of ownership.
The Novel Approach
In stark contrast, the methodology outlined in CN114685298A offers a streamlined, green alternative that directly addresses these pain points. By employing a photocatalytic phenyl migration strategy, the inventors have successfully constructed the complex carbon framework of baclofen under remarkably mild conditions. The process initiates with a straightforward nucleophilic addition to form a key intermediate, followed by a visible-light-driven rearrangement. This approach completely eliminates the need for cryogenic cooling and high-pressure hydrogenation vessels. The reaction proceeds efficiently at near-ambient temperatures using white light irradiation, significantly lowering energy consumption. Moreover, the substitution of toxic solvents and reagents with safer alternatives aligns perfectly with modern green chemistry principles, making this route highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Photocatalytic Phenyl Migration
The core of this technological breakthrough lies in the precise application of organic photoredox catalysis to drive a radical rearrangement. The mechanism involves the excitation of an organic photocatalyst, specifically an naphthalimide derivative, by visible light to generate a potent excited state species. This excited catalyst engages in a single-electron transfer (SET) process with a pyridinium salt additive, generating a radical intermediate that triggers the migration of the phenyl group. This radical cascade allows for the formation of carbon-carbon bonds that are traditionally difficult to access without extreme conditions. The elegance of this mechanism is its ability to bypass high-energy transition states required in thermal reactions, thereby minimizing the formation of thermal degradation byproducts and ensuring a cleaner reaction profile.
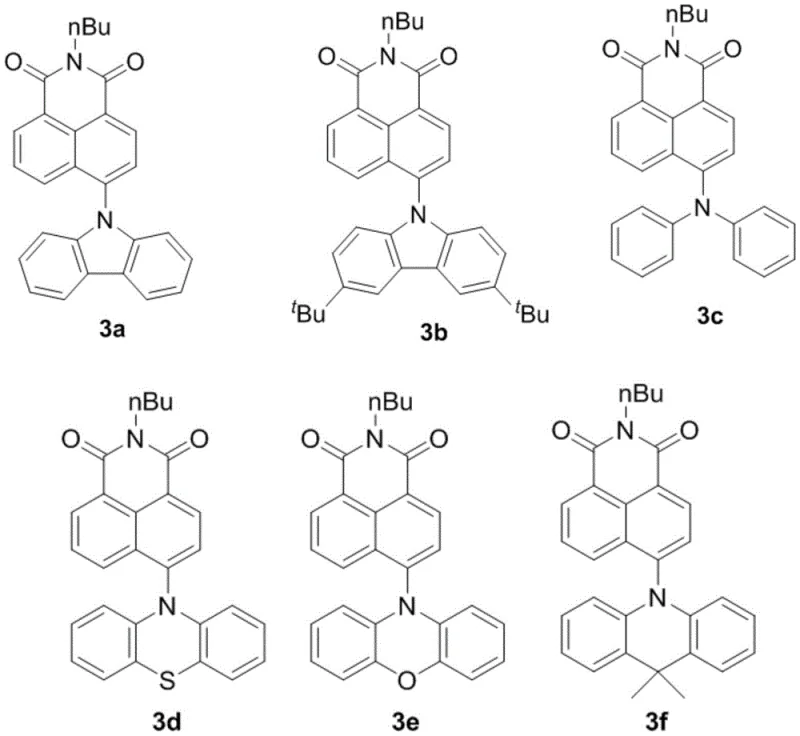
Crucially, the selection of the photocatalyst plays a pivotal role in the success of this transformation. As illustrated in the structural analysis, the patent highlights a series of naphthalimide-based catalysts, with N-n-butyl-4-carbazole-1,8-naphthalimide (designated as NI-Cz or 3a) emerging as the superior candidate. This specific molecular architecture optimizes the redox potentials necessary for the efficient generation of radical intermediates while maintaining stability under prolonged irradiation. From an impurity control perspective, the mildness of the photoredox cycle ensures that sensitive functional groups remain intact, reducing the complexity of the impurity spectrum. This results in a crude product that requires less rigorous purification, directly translating to higher overall yields and reduced solvent usage during isolation.
How to Synthesize Baclofen Efficiently
Implementing this photocatalytic route requires careful attention to reaction parameters to maximize the 88% yield reported in the preferred embodiments. The process begins with the preparation of the vinyl-substituted intermediate via Grignard addition at 0°C, followed by the critical photoredox step. Optimization studies within the patent indicate that the molar ratio of the intermediate, pyridinium salt, and photocatalyst is critical, with a range of 1:1~5:0.001~0.05 proving effective. The use of specific additives such as DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) and TMEDA further enhances the reaction efficiency by facilitating the radical generation cycle. For detailed operational protocols, the following guide outlines the standardized synthesis steps.
- Perform nucleophilic addition of vinyl Grignard reagent to methyl 4-chlorobenzoylformate at 0°C under inert atmosphere to generate Intermediate 1.
- Conduct the photocatalytic reaction using Intermediate 1, a pyridinium salt, and the organic photocatalyst NI-Cz (3a) under white light irradiation.
- Execute post-reaction processing including desolvation, reduction with hypophosphorous acid, and hydrolysis to isolate high-purity baclofen.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible strategic benefits beyond mere technical novelty. The shift from a multi-step, hazard-heavy process to a streamlined photochemical route fundamentally alters the cost structure and risk profile of baclofen production. By removing the dependency on specialized high-pressure infrastructure and cryogenic capabilities, manufacturers can utilize more versatile, general-purpose reactor trains. This flexibility enhances supply chain resilience, allowing for faster response times to market demand fluctuations. Furthermore, the elimination of toxic reagents like nitromethane and pyridine simplifies regulatory compliance and waste disposal, leading to substantial cost savings in environmental management.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of energy-intensive unit operations. Traditional routes requiring -78°C cooling and high-pressure hydrogenation incur significant utility and capital equipment costs. By operating under visible light and mild thermal conditions, the new method drastically reduces energy consumption. Additionally, the use of an organic photocatalyst (NI-Cz) avoids the expense associated with precious metal catalysts like Iridium or Ruthenium, and eliminates the need for costly metal scavenging steps to meet strict residual metal specifications in the final API.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, such as methyl 4-chlorobenzoylformate and vinyl Grignard reagents, are commercially available and stable, reducing the risk of supply bottlenecks. The robustness of the organic photocatalyst ensures consistent batch-to-batch performance, minimizing the risk of production delays caused by catalyst deactivation or variability. This reliability is crucial for maintaining continuous supply lines for essential medications like baclofen, ensuring that downstream formulation partners receive materials on schedule without interruption.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but recent advancements in flow chemistry and LED reactor design have made this highly feasible. The process described avoids the generation of hazardous waste streams associated with heavy metals and toxic solvents, aligning with increasingly stringent global environmental regulations. This "green" profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, a key factor for partnerships with major multinational pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility and advantages for potential manufacturing partners.
Q: What are the primary advantages of this photocatalytic route over traditional baclofen synthesis?
A: This method eliminates the need for hazardous reagents like nitromethane and toxic solvents like pyridine used in conventional Knoevenagel-Doebner reactions. It also avoids energy-intensive cryogenic conditions (-78°C) and high-pressure hydrogenation, significantly improving process safety and environmental compliance.
Q: Which photocatalyst demonstrates the highest efficiency for this transformation?
A: The patent identifies N-n-butyl-4-carbazole-1,8-naphthalimide (NI-Cz, structure 3a) as the optimal organic photocatalyst. Screening data indicates it outperforms traditional transition metal catalysts like Ruthenium and Iridium complexes, offering superior yields up to 88% while being metal-free.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It utilizes mild reaction conditions (white light, ambient to moderate temperatures) and avoids specialized high-pressure equipment. The use of robust organic photocatalysts and simplified workup procedures facilitates seamless transition from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baclofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic migration strategy detailed in CN114685298A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle both traditional synthetic routes and cutting-edge photochemical processes, ensuring that we can deliver baclofen and its precursors with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to monitor impurity profiles, guaranteeing that every batch meets the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in green chemistry can drive efficiency and reliability in your baclofen sourcing strategy.
