Advanced One-Step Synthesis of Tetraisopropyl Hydrazine for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing complex nitrogen-containing heterocycles and hydrazine derivatives, which serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN109232301B, which details a novel preparation method for tetraisopropyl hydrazine. This compound is not merely a theoretical curiosity but a fundamental material for studying separable free radical cations, making its reliable production essential for advanced organic synthesis research. The patent introduces a streamlined one-step alkylation strategy that fundamentally alters the economic and operational landscape of producing this specialized intermediate. By shifting away from multi-step sequences involving hazardous or prohibitively expensive oxidants, this technology offers a robust alternative for reliable pharma intermediate supplier networks aiming to optimize their catalog offerings. The core innovation lies in the direct conversion of triisopropylhydrazine to the tetra-substituted product using a lithium diisopropylamide (LDA) and copper(I) iodide system, bypassing the need for the notoriously costly nitrosohexafluorophosphoric acid.
The Limitations of Conventional Methods vs. The Novel Approach
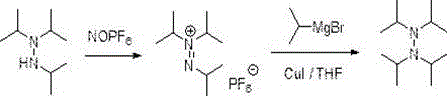
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of tetraisopropyl hydrazine was governed by a rigid and inefficient two-step protocol reported in the Journal of the American Chemical Society (1995). This legacy method necessitates the initial reaction of triisopropylhydrazine with nitrosohexafluorophosphoric acid (NOPF6) in acetonitrile to generate a triisopropylazonium hexafluorophosphate intermediate. This first step is fraught with logistical challenges, primarily due to the reliance on NOPF6, an imported reagent that commands an exorbitant market price of approximately 3500 RMB per 25 grams. Furthermore, the subsequent step requires the addition of an isopropyl Grignard reagent in the presence of cuprous iodide to complete the alkylation. The cumulative effect of these two distinct reaction stages results in a disappointing overall yield of only 23%, creating significant material loss and waste generation. For procurement managers, the dependency on a single-source imported oxidant represents a severe supply chain vulnerability, while the low yield inflates the cost per kilogram of the final API intermediate to unsustainable levels.
The Novel Approach
In stark contrast to the cumbersome legacy process, the methodology disclosed in CN109232301B achieves the transformation in a single synthetic operation, effectively collapsing the reaction coordinate and eliminating the isolation of unstable intermediates. This novel approach utilizes a strong base, LDA, generated in situ, to deprotonate the triisopropylhydrazine substrate, followed immediately by a copper-catalyzed cross-coupling with 2-bromopropane. By removing the requirement for the expensive NOPF6 oxidant, the process drastically reduces the raw material expenditure, with the patent noting that the total reagent cost for producing 5 grams of product drops from roughly 6000 RMB to under 300 RMB. Moreover, the yield sees a tangible improvement, rising from the historical 23% to a more commercially viable 32%. This shift not only enhances cost reduction in pharma intermediate manufacturing but also simplifies the operational workflow, reducing the time and labor required for intermediate workups and purifications, thereby increasing overall throughput capacity for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into LDA-Mediated Copper-Catalyzed Alkylation
The success of this one-step synthesis hinges on the precise control of organometallic species and the stabilization of reactive intermediates through the use of specific ligands. The reaction initiates at cryogenic temperatures (-78°C) where LDA, formed from diisopropylamine and n-butyllithium, acts as a potent non-nucleophilic base to abstract a proton from the nitrogen center of triisopropylhydrazine. The inclusion of TMEDA (tetramethylethylenediamine) is critical here; it serves as a stabilizing ligand that complexes with the lithium cation, enhancing the solubility and reactivity of the resulting lithiated hydrazine species. Without this stabilization, the highly reactive anion might decompose or undergo unwanted side reactions before the electrophile is introduced. Once the lithiated intermediate is formed, the system is warmed to room temperature to ensure complete deprotonation before being cooled again for the addition of the copper catalyst and alkyl halide.
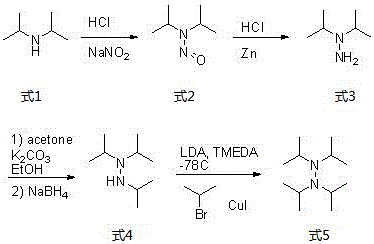
The subsequent alkylation step proceeds via a copper-mediated mechanism, likely involving the transmetallation of the lithiated hydrazine to a copper species, followed by oxidative addition of the 2-bromopropane and reductive elimination to form the new N-C bond. This mechanistic pathway avoids the formation of the azo-cation intermediate required in the old method, thereby sidestepping the associated stability issues and low yields. Impurity control is inherently improved because the reaction avoids the harsh oxidative conditions of the NOPF6 step, which often leads to over-oxidation or decomposition products. The use of TLC monitoring with a PE:EA (3:1) solvent system allows for precise determination of reaction completion, ensuring that the valuable starting material is fully consumed without prolonged exposure to conditions that might degrade the product. This level of mechanistic understanding is vital for R&D directors aiming to replicate the process for high-purity OLED material or pharmaceutical precursors where impurity profiles are strictly regulated.
How to Synthesize Tetraisopropyl Hydrazine Efficiently
The practical execution of this synthesis requires strict adherence to temperature protocols and stoichiometric ratios to maximize the 32% yield potential. The process begins with the generation of LDA in diethyl ether, followed by the sequential addition of the stabilizer and substrate. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to ensure reproducibility in a pilot or production setting.
- Prepare LDA in diethyl ether at -78°C using diisopropylamine and n-butyllithium, then add TMEDA stabilizer and triisopropylhydrazine.
- Warm the mixture to room temperature for deprotonation, then cool back to -78°C before adding CuI catalyst and 2-bromopropane.
- Stir at room temperature until completion, followed by aqueous workup, extraction with petroleum ether, and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented methodology translates directly into enhanced operational resilience and significant financial benefits. The primary advantage is the complete elimination of nitrosohexafluorophosphoric acid (NOPF6) from the bill of materials. As noted in the patent data, this reagent is not only expensive but also imported, creating a single point of failure in the supply chain that can lead to production stoppages. By replacing this with readily available commodity chemicals like diisopropylamine, n-butyllithium, and 2-bromopropane, manufacturers can secure a more stable and predictable supply of raw materials. This shift mitigates the risk of price volatility associated with specialty imported reagents and ensures continuous production capability, which is essential for meeting the demanding delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this process change is profound, driven by the substitution of a high-cost oxidant with inexpensive bulk chemicals. The patent explicitly highlights that the reagent cost for a small-scale batch drops from approximately 6000 RMB to under 300 RMB, representing a massive reduction in variable costs. This efficiency is achieved not just through cheaper inputs but also through the consolidation of two reaction steps into one, which reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours. Consequently, the overall cost of goods sold (COGS) for tetraisopropyl hydrazine is significantly lowered, allowing suppliers to offer more competitive pricing structures while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Dependence on imported specialty reagents like NOPF6 introduces long lead times and customs-related uncertainties that can disrupt production planning. The new method relies on a supply chain of common organic solvents and reagents that are widely produced and stocked by multiple vendors globally. This diversification of the supply base drastically reduces the lead time for raw material acquisition and minimizes the risk of stockouts. For supply chain heads, this means greater agility in responding to sudden increases in demand and the ability to maintain safety stock levels without tying up excessive capital in expensive, slow-moving inventory items.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the one-step nature of the reaction simplifies the engineering requirements for scale-up. Fewer unit operations mean less equipment footprint and reduced complexity in process control systems. Additionally, avoiding the use of heavy fluorine-containing oxidants reduces the burden on waste treatment facilities, as there is no need to manage hexafluorophosphate byproducts. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, facilitating smoother regulatory approvals and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, reagent substitutions, and process safety.
Q: How does the new preparation method improve yield compared to literature methods?
A: The novel one-step process achieves a 32% yield, significantly outperforming the traditional two-step literature method which only yields 23%.
Q: Why is avoiding nitrosohexafluorophosphoric acid (NOPF6) critical for cost reduction?
A: NOPF6 is an expensive imported reagent costing approximately 3500 RMB per 25g. The new method eliminates this reagent entirely, reducing total reagent costs for 5g of product from roughly 6000 RMB to under 300 RMB.
Q: What role does TMEDA play in the reaction mechanism?
A: TMEDA acts as a crucial stabilizer for the LDA base, ensuring efficient deprotonation of the hydrazine nitrogen at low temperatures (-78°C) prior to the copper-catalyzed alkylation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraisopropyl Hydrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the global fine chemicals market. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN109232301B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering stringent purity specifications for tetraisopropyl hydrazine, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for downstream pharmaceutical applications. Our infrastructure is designed to handle complex organometallic reactions safely and efficiently, ensuring that the benefits of this novel one-step process are fully realized at an industrial scale.
We invite potential partners to engage with our technical sales team to discuss how this optimized manufacturing route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this new method. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of this high-quality intermediate into your supply chain.
