Revolutionizing Photoinitiator Production: Continuous Flow Synthesis for Alpha-Hydroxyketones
Revolutionizing Photoinitiator Production: Continuous Flow Synthesis for Alpha-Hydroxyketones
The chemical manufacturing landscape for high-performance photoinitiators is undergoing a significant transformation driven by the adoption of continuous flow chemistry. Patent CN111056934A introduces a groundbreaking method for preparing α-hydroxyketone photoinitiators utilizing microreactor technology, addressing critical bottlenecks in safety, efficiency, and product quality that have long plagued traditional batch processes. This innovation is particularly relevant for the production of key industry standards such as Photoinitiator 1173 (2-hydroxy-2-methyl-1-phenylacetone), which serves as a cornerstone for UV curing applications in coatings, inks, and adhesives. By shifting from intermittent kettle-type reactors to a continuous micro-reaction system, this technology enables precise control over exothermic chlorination and subsequent hydrolysis steps, resulting in a process that is not only safer but also capable of delivering superior chemical purity and yield consistency.
For R&D directors and process engineers, the implications of this patent extend beyond simple yield improvements; it represents a fundamental shift in how hazardous halogenation reactions are managed. The traditional reliance on large-volume batch reactors for chlorination poses inherent risks due to the accumulation of heat and the handling of toxic chlorine gas. In contrast, the microreactor approach described in CN111056934A leverages the high heat and mass transfer coefficients of micro-channels to maintain isothermal conditions even during highly exothermic steps. This capability ensures that side reactions, such as over-chlorination or decomposition, are minimized, leading to a cleaner reaction profile. Furthermore, the continuous nature of the process allows for real-time monitoring and adjustment of parameters, providing a level of process analytical technology (PAT) integration that is difficult to achieve in batch systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
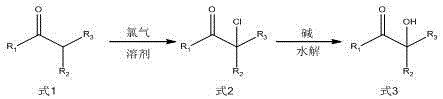
Traditional manufacturing routes for α-hydroxyketone photoinitiators typically rely on Friedel-Crafts halohydrolysis or batch-wise chlorination followed by alkaline hydrolysis in large stirred-tank reactors. These conventional methods suffer from several intrinsic disadvantages that impact both operational safety and economic efficiency. Firstly, the chlorination step is highly exothermic, and in a large batch vessel, heat removal is often inefficient, leading to hot spots that promote the formation of di-chlorinated byproducts and other impurities. Secondly, the handling of chlorine gas in large headspaces increases the risk of leakage and exposure, necessitating complex and costly scrubbing systems. Thirdly, the batch process requires long cycle times, including heating, cooling, and extensive work-up procedures like extraction and washing, which significantly increase energy consumption and solvent waste. The comparative examples in the patent data highlight these issues, showing that batch processes often result in lower selectivity and require prolonged reaction times, sometimes extending to several hours for a single step, which limits overall plant throughput.
The Novel Approach
The novel approach disclosed in patent CN111056934A overcomes these limitations by implementing a continuous flow strategy using microreactors. This method involves pumping the ketone substrate and chlorine gas into a micro-mixer where they are instantly homogenized before entering a micro-channel reactor. The small channel dimensions (typically 50-1000 μm) ensure that the diffusion distance is minimal, allowing for rapid mixing and heat exchange. This results in a narrow residence time distribution, meaning every molecule experiences nearly identical reaction conditions, which drastically improves selectivity towards the mono-chlorinated intermediate. Following chlorination, the stream is directly fed into a hydrolysis module where it meets an aqueous sodium hydroxide solution. This telescoped process eliminates the need for isolating the unstable chloro-intermediate, reducing handling risks and processing time. The continuous removal of product from the reaction zone prevents further degradation, ensuring that the final α-hydroxyketone is obtained with exceptional purity and yield, as evidenced by the patent's experimental data showing yields reaching up to 96%.
Mechanistic Insights into Microreactor-Mediated Chlorination and Hydrolysis
The core of this technological advancement lies in the precise manipulation of reaction kinetics through engineering control. In the chlorination stage, the reaction proceeds via a radical mechanism where chlorine atoms substitute the alpha-hydrogen of the ketone. In a batch reactor, the concentration of chlorine can fluctuate locally, leading to regions of high chlorine concentration that favor poly-chlorination. However, in the microreactor system, the stoichiometric ratio of chlorine to ketone is strictly controlled at the inlet, typically between 1:1 and 1:1.2. The rapid mixing ensures that the chlorine is consumed almost immediately upon contact with the substrate, maintaining a low local concentration of free chlorine throughout the channel. This kinetic control is crucial for suppressing the formation of di-chlorinated impurities, which are difficult to separate and can negatively affect the performance of the final photoinitiator in UV curing applications. The patent specifies reaction temperatures between 20°C and 70°C, a range that is easily maintained in a microreactor due to its high surface-area-to-volume ratio, preventing thermal runaway.
Following the formation of the alpha-chloro ketone intermediate, the stream undergoes nucleophilic substitution hydrolysis. The mechanism involves the attack of the hydroxide ion on the carbon bearing the chlorine atom, displacing the chloride ion to form the alpha-hydroxy group. In traditional batch processes, mass transfer limitations between the organic phase (containing the chloro-ketone) and the aqueous phase (containing NaOH) can slow down this reaction, often requiring phase transfer catalysts or prolonged stirring. The microreactor environment enhances this interfacial contact significantly. The high shear forces generated within the micro-channels create fine emulsions or segmented flows, maximizing the interfacial area between the two phases. This intensification allows the hydrolysis to proceed rapidly, often completing within minutes rather than hours. Furthermore, the continuous removal of the salt byproduct (NaCl) and the product from the reaction zone drives the equilibrium forward, ensuring high conversion rates. The result is a process that not only delivers high purity but also minimizes the generation of wastewater and solvent waste associated with extensive extraction steps.
How to Synthesize Alpha-Hydroxyketone Photoinitiators Efficiently
The synthesis of high-purity alpha-hydroxyketone photoinitiators via this microreactor method involves a streamlined sequence of continuous operations that replace traditional batch unit operations. The process begins with the precise metering of raw materials, where the ketone substrate and chlorine gas are introduced into the system under controlled pressure and flow rates. The reaction conditions are optimized to ensure that the chlorination proceeds with maximal selectivity, avoiding the formation of unwanted byproducts. Once the chlorination is complete, the intermediate stream is not isolated but is instead directly transferred to the hydrolysis module. Here, it is mixed with a controlled stream of sodium hydroxide solution. The entire process is monitored to ensure that residence times and temperatures remain within the optimal window defined by the patent. For a detailed breakdown of the specific operational parameters, equipment configurations, and step-by-step protocols required to implement this technology, please refer to the standardized synthesis guide below.
- Prepare the chlorination feed by pumping isobutyrophenone (or substituted phenyl ketone) and chlorine gas into a micro-mixer, ensuring precise molar ratios between 1: 1 and 1:1.2.
- Pass the mixture through a micro-channel reactor maintained at 20-70°C for rapid chlorination, achieving high selectivity for the alpha-chloro ketone intermediate.
- Immediately pump the chlorinated intermediate and aqueous sodium hydroxide into a second micro-reactor for hydrolysis, followed by separation and purification to obtain the final photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the microreactor technology described in CN111056934A offers compelling strategic advantages that go beyond simple technical metrics. The transition from batch to continuous flow fundamentally alters the cost structure and risk profile of photoinitiator manufacturing. By eliminating the need for large, expensive batch reactors and the associated safety infrastructure for handling bulk chlorine gas, capital expenditure (CAPEX) can be significantly optimized. Moreover, the continuous nature of the process allows for a smaller physical footprint, enabling production facilities to be located closer to key markets or integrated into existing sites with space constraints. The reduction in reaction time from hours to minutes translates directly into higher asset utilization rates, meaning that the same production capacity can be achieved with fewer resources. This efficiency gain is critical for meeting the growing global demand for UV-curable materials without the need for massive facility expansions.
- Cost Reduction in Manufacturing: The implementation of microreactor technology leads to substantial cost savings through multiple mechanisms. Firstly, the dramatic improvement in reaction selectivity reduces the consumption of raw materials, as less feedstock is wasted on forming byproducts. Secondly, the elimination of intermediate isolation steps and the reduction in solvent usage for work-ups lower the variable costs associated with solvents and waste disposal. Thirdly, the energy efficiency of microreactors is superior to batch systems due to reduced heating and cooling loads, further driving down operational expenses. While specific percentage savings depend on the scale of operation, the qualitative impact on the margin structure is profound, allowing suppliers to offer more competitive pricing for high-purity photoinitiators.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the long lead times and batch-to-batch variability inherent in traditional chemical manufacturing. The continuous flow process mitigates these risks by providing a steady, uninterrupted stream of product. The robustness of the microreactor system against fluctuations in reaction conditions ensures consistent quality, reducing the likelihood of batch failures that can disrupt supply. Additionally, the modular nature of the equipment allows for rapid scale-up through numbering-up, meaning that production capacity can be increased quickly to meet surges in demand without the long lead times associated with constructing new large-scale reactors. This agility is a significant advantage in a volatile market environment.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, particularly regarding the emission of volatile organic compounds (VOCs) and the handling of hazardous gases. The microreactor process addresses these concerns by operating in a closed system with minimal headspace, virtually eliminating fugitive emissions of chlorine gas. The high efficiency of the reaction also means less waste is generated per unit of product, simplifying wastewater treatment and reducing the environmental footprint. From a scalability perspective, the linear relationship between the number of reactor modules and output capacity makes it easy to predict and plan for future production needs. This predictability, combined with the inherent safety of the technology, ensures long-term compliance and operational stability.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of the microreactor synthesis method for alpha-hydroxyketone photoinitiators. These answers are derived directly from the technical disclosures and experimental data provided in patent CN111056934A, offering clarity on how this technology compares to established industry practices. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: How does microreactor technology improve the safety of chlorination reactions?
A: Microreactors drastically reduce the hold-up volume of hazardous reagents like chlorine gas and exothermic intermediates. The high surface-to-volume ratio allows for immediate heat dissipation, preventing thermal runaways common in batch kettle reactors.
Q: What purity levels can be achieved with this continuous flow method?
A: Experimental data from the patent indicates that this method can achieve product purities exceeding 99.6%, significantly higher than the 97.7% typically seen in traditional batch processes, due to precise residence time control.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for number-up scaling rather than vessel size increase. By parallelizing microreactor units, manufacturers can achieve tonnage-scale production while maintaining the same reaction efficiency and selectivity observed in the lab.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxyketone Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of advanced photo-initiating compounds. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate the innovative methods described in patent CN111056934A into commercial reality. Our facilities are equipped with state-of-the-art microreactor systems capable of handling hazardous chemistries like chlorination with the utmost safety and precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality materials. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that our photoinitiators perform flawlessly in your UV curing formulations.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain. Whether you are looking to secure a reliable source of Photoinitiator 1173 or explore custom synthesis routes for novel derivatives, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our microreactor-based solutions can enhance your product performance while optimizing your total cost of ownership.
