Scalable Synthesis of 4-Chloro-2-fluoro-5-nitroacetophenone for Hepatitis C Drug Intermediates
Scalable Synthesis of 4-Chloro-2-fluoro-5-nitroacetophenone for Hepatitis C Drug Intermediates
The rapid evolution of direct-acting antiviral therapies for Hepatitis C has placed immense pressure on the global supply chain for key pharmaceutical intermediates, particularly those required for the synthesis of pibrentasvir, a critical component of the blockbuster combination drug Mavyret. In response to the urgent need for more efficient manufacturing pathways, patent CN110668948B discloses a groundbreaking preparation method for 4-chloro-2-fluoro-5-nitroacetophenone that fundamentally restructures the synthetic logic away from scarce precursors. This technical breakthrough leverages m-fluoroaniline, a readily accessible commodity chemical, as the foundational building block, thereby circumventing the historical reliance on expensive and hard-to-source fluorinated benzoic acid derivatives. By integrating a strategic sequence of acetyl protection, regioselective Friedel-Crafts acylation, and classic Sandmeyer chlorination, this novel route offers a robust solution for high-purity intermediate production. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and supply chain implications of this patent is essential for securing a competitive advantage in the antiviral market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chloro-2-fluoro-5-nitroacetophenone has been plagued by significant upstream supply chain vulnerabilities associated with the choice of starting materials. Traditional routes, as illustrated in the prior art, typically initiate from 4-chloro-2-fluorobenzoic acid or its nitrated derivative, 4-chloro-2-fluoro-5-nitrobenzoic acid. These fluorinated aromatic acids are not only characterized by exorbitant market prices due to complex fluorination processes but also suffer from limited commercial availability, creating a single point of failure for large-scale manufacturing. Furthermore, the conversion of these carboxylic acid derivatives into the corresponding acetophenone structure often requires harsh conditions or multiple activation steps that degrade overall process efficiency. The economic burden of sourcing these niche starting materials directly impacts the cost of goods sold (COGS) for the final API, making the conventional pathway unsustainable for cost-sensitive generic production or large-volume contracts.
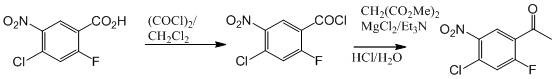
The Novel Approach
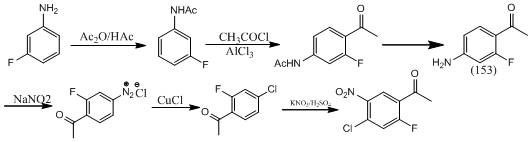
In stark contrast to the legacy methods, the innovative strategy outlined in the patent data pivots to m-fluoroaniline, a bulk chemical with a mature and stable global supply network. This new methodology employs a five-step linear synthesis that maximizes atom economy while minimizing the need for specialized reagents. The process begins with the acetylation of the amine to protect the reactive center, followed by a highly regioselective Friedel-Crafts acylation that installs the acetyl group precisely at the para-position relative to the nitrogen. Subsequent hydrolysis reveals the free amine, which is then transformed into the chloro substituent via a Sandmeyer reaction, a transformation known for its reliability in introducing halogens onto aromatic rings. The final nitration step completes the substitution pattern, delivering the target molecule with high purity. This logical disconnection transforms a supply-constrained problem into a commodity-driven solution, significantly enhancing the feasibility of commercial scale-up.
Mechanistic Insights into Friedel-Crafts Acylation and Sandmeyer Chlorination
The success of this synthetic route hinges critically on the precise control of regioselectivity during the Friedel-Crafts acylation step, where the acetyl protecting group plays a dual role as both a deactivator against over-reaction and a director for para-substitution. In the presence of a Lewis acid catalyst such as aluminum trichloride, the acetamido group directs the incoming acyl electrophile exclusively to the position para to the nitrogen, avoiding the formation of ortho-isomers which would be difficult to separate and would constitute a critical quality attribute failure. The reaction conditions, utilizing solvents like carbon disulfide or nitrobenzene at controlled temperatures between 0°C and 50°C, ensure that the kinetic product is favored while minimizing polymerization or resinification of the aniline derivative. This level of control is paramount for maintaining a clean impurity profile, as downstream purification of structural isomers in fluorinated aromatics is notoriously difficult and yield-destructive.

Following the installation of the ketone functionality, the transformation of the amino group to a chloro group via the Sandmeyer reaction represents another pivotal mechanistic checkpoint for ensuring product integrity. This process involves the generation of a diazonium salt intermediate at low temperatures, typically below -5°C, to prevent premature decomposition and the formation of phenolic byproducts. The subsequent treatment with cuprous chloride facilitates the radical replacement of the diazonium moiety with chlorine, a reaction that proceeds with high fidelity on the electron-deficient acetophenone ring. The patent data indicates that careful control of the pH and temperature during the diazotization phase is essential to suppress the formation of azo-coupling impurities. By optimizing the stoichiometry of sodium nitrite and the copper catalyst, the process achieves excellent conversion rates, ensuring that the final nitration step proceeds on a uniform substrate, thereby maximizing the overall yield of the final nitro-acetophenone product.
How to Synthesize 4-Chloro-2-fluoro-5-nitroacetophenone Efficiently
Implementing this synthesis at an industrial level requires strict adherence to the optimized reaction parameters defined in the patent examples to ensure consistent quality and safety. The process is designed to be executed in standard glass-lined or stainless steel reactors capable of handling mild exotherms and corrosive acidic conditions. Operators must pay particular attention to the temperature profiles during the diazotization and nitration steps, as thermal runaway in these exothermic transformations can lead to hazardous situations and degraded product quality. The workup procedures involve standard liquid-liquid extractions and crystallizations, utilizing common solvents like dichloromethane, ethyl acetate, and hexanes, which simplifies solvent recovery and waste management protocols. For a detailed breakdown of the specific operational parameters, reagent grades, and isolation techniques required to replicate the high yields reported in the patent literature, please refer to the standardized technical guide below.
- Acetylate m-fluoroaniline with acetic anhydride to form 3-fluoroacetanilide.
- Perform Friedel-Crafts acylation using acetyl chloride and AlCl3 to obtain 2-fluoro-4-acetamidoacetophenone.
- Hydrolyze the acetamido group using hydrochloric acid to yield 2-fluoro-4-aminoacetophenone.
- Execute Sandmeyer reaction via diazotization and cuprous chloride treatment to introduce the chloro group.
- Conduct final nitration using potassium nitrate and sulfuric acid at low temperature to afford the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this m-fluoroaniline-based route offers profound advantages in terms of cost stability and supply chain resilience. By decoupling the production of this critical intermediate from the volatile market for fluorinated benzoic acids, manufacturers can secure long-term supply agreements based on commodity pricing models rather than specialty chemical premiums. The elimination of expensive starting materials translates directly into a reduced raw material cost base, allowing for more competitive bidding on downstream API contracts. Furthermore, the use of ubiquitous reagents such as acetic anhydride, acetyl chloride, and potassium nitrate ensures that the supply chain is not vulnerable to the shortages of niche catalysts or proprietary reagents that often plague complex pharmaceutical syntheses. This robustness is essential for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of high-value fluorinated acids with low-cost m-fluoroaniline, which drastically lowers the entry price for the synthesis. Additionally, the high regioselectivity of the Friedel-Crafts step minimizes the formation of isomeric impurities, reducing the need for costly and yield-loss-inducing purification steps such as preparative chromatography. The ability to recover and recycle solvents like carbon disulfide or nitrobenzene further contributes to the economic efficiency of the process, lowering the overall environmental and financial burden of solvent consumption. Consequently, the total cost of production is significantly optimized, providing a substantial margin advantage for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Relying on m-fluoroaniline as the starting material mitigates the risk of supply disruptions, as this chemical is produced by multiple major vendors globally with ample capacity. Unlike the specialized fluorinated benzoic acids which may have limited suppliers and long lead times, m-fluoroaniline can be sourced with short notice, enabling just-in-time manufacturing strategies. The robustness of the synthetic steps, which do not require sensitive organometallic catalysts or inert atmosphere conditions beyond standard practice, ensures that production can be maintained across different geographic locations without technology transfer friction. This flexibility empowers supply chain managers to diversify their manufacturing footprint and reduce dependency on single-source providers.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations that are standard in the fine chemical industry, such as reflux, filtration, and distillation. The absence of heavy metal catalysts, with the exception of the catalytic amount of copper in the Sandmeyer step which is easily removed, simplifies the waste stream treatment and reduces the environmental compliance burden. The high yields reported in the patent examples indicate a material-efficient process that generates less waste per kilogram of product, aligning with green chemistry principles. This ease of scale-up from pilot plant to multi-ton commercial production ensures that the technology can rapidly meet surging market demand for Hepatitis C therapeutics without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived from a detailed analysis of the patent specifications and process chemistry. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these specifics is crucial for making informed decisions about technology transfer and vendor qualification.
Q: Why is m-fluoroaniline preferred over 4-chloro-2-fluorobenzoic acid as a starting material?
A: m-Fluoroaniline is a widely available commodity chemical with stable pricing, whereas fluorinated benzoic acid derivatives are often expensive, difficult to source, and subject to supply chain bottlenecks.
Q: How does this process control impurities during the Friedel-Crafts step?
A: The acetyl protection group on the amine directs the acylation specifically to the para-position, preventing ortho-substitution and poly-acylation side reactions common in unprotected anilines.
Q: Is this synthesis route suitable for multi-ton commercial production?
A: Yes, the route utilizes standard unit operations such as reflux, crystallization, and extraction without requiring exotic catalysts or extreme pressure conditions, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-fluoro-5-nitroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN110668948B can be reliably replicated at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to monitor every critical process parameter, guaranteeing that every batch of 4-chloro-2-fluoro-5-nitroacetophenone meets the exacting standards required for API synthesis. Our commitment to quality assurance extends beyond mere compliance, as we proactively manage impurity profiles to facilitate smoother regulatory filings for our clients.
We invite forward-thinking pharmaceutical companies and procurement leaders to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain security. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing excellence can drive value and reliability in your Hepatitis C drug development programs.
