Industrial Scale Synthesis of 4-Chloro-2-Trifluoromethyl-Acetophenone Intermediates via Safe Malonate Coupling
Introduction to Advanced Intermediate Synthesis
The global demand for high-performance triazole fungicides, particularly chlorofluoroether oxazoles like Revysol, has necessitated a rigorous re-evaluation of supply chains for critical building blocks. Patent CN113968775A introduces a transformative methodology for producing 4-chloro-2-trifluoromethyl-acetophenone, a pivotal intermediate in this value chain. This intellectual property shifts the paradigm from hazardous, multi-step halogenation sequences to a streamlined, industrially viable process centered on malonate coupling. For R&D directors and procurement strategists, this represents a significant opportunity to de-risk manufacturing operations while securing a stable supply of high-purity agrochemical intermediates. The technology leverages abundant raw materials and avoids the severe safety liabilities associated with traditional organometallic approaches, positioning it as a superior choice for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chloro-2-trifluoromethyl-acetophenone has relied heavily on Grignard chemistry, a pathway fraught with operational hazards and supply chain fragility. As illustrated in the prior art reaction schemes, the conventional route typically begins with 2-bromo-5-chlorotrifluoromethylbenzene, a specialized raw material that is neither commoditized nor easily sourced in bulk quantities. The preparation of this brominated precursor itself involves a treacherous sequence of nitration, reduction, and Sandmeyer reactions, each step introducing potential safety incidents and generating substantial toxic waste streams. Furthermore, the core Grignard exchange reaction using isopropyl magnesium chloride requires strictly anhydrous conditions and poses significant thermal runaway risks, making it ill-suited for large-scale continuous manufacturing without expensive engineering controls.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes 3,4-dimethyl chlorobenzene as a foundational feedstock, a commodity chemical that offers superior availability and cost stability. The new synthetic strategy replaces the dangerous Grignard coupling with a robust acylation-coupling sequence involving dialkyl malonates. This approach not only bypasses the need for explosive nitration steps but also eliminates the handling of moisture-sensitive organometallic reagents entirely. The process flow is characterized by a logical progression of chlorination, fluorine-chlorine exchange, and hydrolysis, culminating in a high-yielding coupling reaction. This structural redesign of the synthesis tree fundamentally alters the risk profile of the manufacturing process, enabling safer operations and more predictable production schedules for supply chain managers.
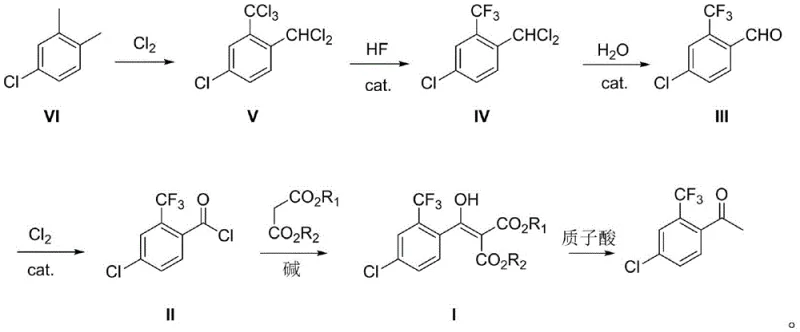
Mechanistic Insights into Fluorine-Chlorine Exchange and Malonate Coupling
The chemical elegance of this process lies in its efficient utilization of halogen exchange mechanics and nucleophilic acyl substitution. The transformation of the trichloromethyl group into a trifluoromethyl moiety is achieved via a catalytic fluorine-chlorine exchange reaction using hydrogen fluoride (HF) and antimony catalysts (SbCl3 or SbCl5). This Swarts-type reaction is highly selective under the specified conditions of 100-120°C and moderate pressure, ensuring minimal formation of poly-fluorinated byproducts. Following this, the dichloromethyl group is hydrolyzed to an aldehyde using Lewis acids like zinc chloride or ferric chloride, which then undergoes radical chlorination to form the reactive benzoyl chloride. This specific activation of the carbonyl carbon is crucial for the subsequent coupling step, as it creates a highly electrophilic center ready for nucleophilic attack by the malonate enolate.
The final construction of the carbon skeleton involves the coupling of the benzoyl chloride with dialkyl malonate in the presence of strong bases such as sodium hydride or sodium tert-butoxide. This reaction forms a beta-keto ester intermediate, specifically diethyl 2-(4-chloro-2-trifluoromethylbenzoyl) malonate, which serves as the protected form of the target ketone. The subsequent hydrolysis and decarboxylation step, catalyzed by mineral acids like sulfuric acid at elevated temperatures (110-130°C), cleanly removes the ester groups to reveal the final acetophenone structure. This two-step "couple-then-deprotect" strategy effectively masks the reactivity of the ketone during the harsh fluorination and chlorination stages, thereby preventing side reactions and ensuring high overall purity of the final API intermediate.
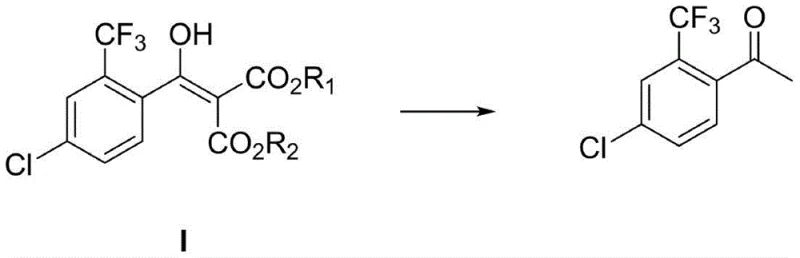
How to Synthesize 4-Chloro-2-Trifluoromethyl-Acetophenone Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly during the fluorination and coupling stages where exotherms can occur. The patent outlines a detailed protocol starting with the photo-chlorination of 3,4-dimethyl chlorobenzene, followed by the critical HF exchange step which demands corrosion-resistant equipment. The subsequent coupling with malonate allows for flexibility in base selection, with sodium ethoxide and sodium tert-butoxide showing excellent results in various solvents including toluene and chlorobenzene. For process engineers looking to replicate this efficiency, the standardized operating procedures below detail the specific stoichiometry, temperature ramps, and workup conditions necessary to achieve the reported yields of over 90% in the final hydrolysis step.
- Chlorinate 3,4-dimethyl chlorobenzene under light to form trichloromethyl/dichloromethyl derivatives, then perform fluorine-chlorine exchange using HF and antimony catalyst.
- Hydrolyze the dichloromethyl group to an aldehyde using Lewis acid catalysts, followed by further chlorination to generate the benzoyl chloride.
- Couple the benzoyl chloride with dialkyl malonate using a base like NaH or sodium tert-butoxide, then hydrolyze and decarboxylate to obtain the final ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages in terms of cost structure and supply chain resilience. By shifting the starting material from a niche brominated aromatic to a widely available dimethyl chlorobenzene, manufacturers can drastically reduce raw material procurement costs and mitigate the risk of supply disruptions. The elimination of the Grignard step removes the need for expensive, high-purity magnesium turnings and specialized dry solvents, while also reducing the energy consumption associated with maintaining strict anhydrous environments. Furthermore, the avoidance of nitration and Sandmeyer reactions significantly lowers the burden on wastewater treatment facilities, as the process generates fewer nitrogenous organic wastes and heavy metal contaminants, leading to substantial operational expenditure savings in environmental compliance.
- Cost Reduction in Manufacturing: The replacement of complex, multi-step precursor synthesis with a direct chlorination-fluorination sequence significantly lowers the cost of goods sold. By utilizing commodity chemicals and avoiding precious metal catalysts or exotic reagents, the process achieves a leaner cost structure. The ability to telescope steps, such as using the crude benzoyl chloride directly in the coupling reaction without distillation, further reduces utility costs and solvent losses, driving down the overall manufacturing expense per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing 3,4-dimethyl chlorobenzene is far more reliable than procuring custom-synthesized brominated intermediates, which often have long lead times and limited supplier bases. This shift ensures a more robust supply chain capable of withstanding market fluctuations. Additionally, the simplified reaction conditions reduce the likelihood of batch failures due to sensitivity issues, thereby improving on-time delivery performance and allowing for more accurate production planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvent-free or low-solvent conditions in key steps like the initial chlorination and fluorination. This minimizes the volume of volatile organic compounds (VOCs) released, simplifying permitting and regulatory approval for new production lines. The high atom economy of the malonate coupling and the straightforward acidic workup facilitate easy scale-up from pilot plant to multi-ton commercial production without requiring disproportionate increases in reactor footprint or waste handling capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, yield, and operational feasibility for potential licensees or manufacturing partners.
Q: What are the safety advantages of this new synthesis route compared to traditional Grignard methods?
A: The new route described in CN113968775A eliminates the use of hazardous Grignard reagents (isopropyl magnesium chloride) and avoids the explosive risks associated with nitration and Sandmeyer reactions required for preparing brominated starting materials. It utilizes safer chlorination and fluorination steps suitable for industrial containment.
Q: How does the malonate coupling strategy improve yield and purity?
A: By using dialkyl malonate coupling followed by hydrolysis and decarboxylation, the process achieves high conversion rates (up to 90% in examples) and simplifies purification. The intermediate malonate derivative can often be used directly in the next step without isolation, reducing solvent waste and processing time.
Q: Is the starting material 3,4-dimethyl chlorobenzene readily available for large-scale production?
A: Yes, 3,4-dimethyl chlorobenzene is a commodity chemical that is significantly easier and cheaper to source than the specialized brominated aromatics (like 2-bromo-5-chlorotrifluoromethylbenzene) required in conventional pathways, ensuring better supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-Trifluoromethyl-Acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of secure and efficient supply chains for critical agrochemical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 4-chloro-2-trifluoromethyl-acetophenone meets the exacting standards required for downstream fungicide synthesis. Our commitment to process safety and environmental stewardship aligns perfectly with the advantages offered by this novel synthetic route.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology for their supply needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your production goals are met with the highest levels of quality and reliability.
