Advanced Synthesis of 5-Fluoro-2-Hydroxyacetophenone for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 5-Fluoro-2-Hydroxyacetophenone for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable pathways for producing critical intermediates, particularly those serving as building blocks for beta-blocker medications. A significant breakthrough in this domain is detailed in Chinese patent CN114539042A, which discloses a novel, highly efficient synthesis process for 5-fluoro-2-hydroxyacetophenone. This compound acts as a pivotal precursor in the manufacture of various cardiovascular drugs, yet traditional production methods have long been plagued by prohibitive costs and severe environmental liabilities. The disclosed innovation shifts the paradigm by utilizing inexpensive o-hydroxyacetophenone as the starting material, bypassing the need for costly fluorinated aromatics. By integrating a mild iron-catalyzed halogenation step followed by a nucleophilic fluorination, this methodology achieves total yields exceeding 80% while drastically minimizing hazardous waste generation. For R&D directors and procurement strategists, this represents a transformative opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with a significantly reduced carbon footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-fluoro-2-hydroxyacetophenone has relied heavily on the Fries rearrangement of phenolic esters or the demethylation of fluoroanisoles, both of which present substantial operational hurdles. As illustrated in prior art such as WO2019/62802, the Fries rearrangement pathway necessitates the use of p-fluorophenol, a relatively expensive raw material, which is first esterified and then subjected to rearrangement using anhydrous aluminum trichloride at temperatures exceeding 130°C.
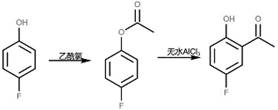
This high-temperature process not only poses significant safety risks due to the exothermic nature of the quenching step but also results in the evolution of large quantities of corrosive hydrogen chloride gas. Furthermore, the hydrolysis of the aluminum complexes post-reaction generates massive volumes of high-salt wastewater that are notoriously difficult and costly to treat. Similarly, alternative routes involving p-fluoroanisole, as seen in WO2018/161008, suffer from similar environmental drawbacks and higher raw material costs.
These legacy processes create a bottleneck for cost reduction in API manufacturing, as the extensive waste treatment infrastructure and specialized high-temperature reactors required drive up the overall production expenditure. The reliance on stoichiometric amounts of Lewis acids like AlCl3 further complicates the supply chain, introducing variability in waste disposal compliance and operational safety.
The Novel Approach
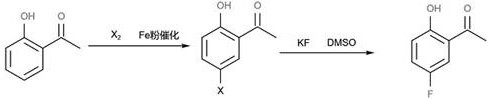
In stark contrast, the new process outlined in CN114539042A introduces a streamlined two-step sequence that fundamentally alters the economic and environmental profile of production. The strategy begins with the direct halogenation of o-hydroxyacetophenone, a commodity chemical that is cheap and abundantly available globally. By employing a catalytic amount of iron powder or ferric salts, the reaction proceeds smoothly at mild temperatures ranging from 0°C to 55°C, effectively installing a halogen atom at the 5-position with high regioselectivity. This intermediate is then subjected to a nucleophilic aromatic substitution using potassium fluoride in a polar aprotic solvent. This approach eliminates the need for harsh Lewis acids and high-temperature rearrangements, thereby avoiding the formation of tarry by-products and ensuring the final product possesses a light color and high purity. The transition from a rearrangement-based logic to a substitution-based logic exemplifies a modern approach to commercial scale-up of complex intermediates, prioritizing atom economy and process safety.
Mechanistic Insights into Iron-Catalyzed Halogenation and Halex Reaction
The success of this synthetic route hinges on the precise control of electrophilic aromatic substitution in the first step. The hydroxyl group on the o-hydroxyacetophenone ring acts as a strong activating group, directing the incoming electrophile (chlorine or bromine) primarily to the para-position relative to itself, which corresponds to the 5-position of the acetophenone skeleton. The inclusion of an iron catalyst, such as iron powder or ferric chloride, facilitates the generation of the active halogen species, allowing the reaction to proceed rapidly even at near-ambient temperatures. This mild condition is crucial for preventing poly-halogenation or oxidative degradation of the phenolic ring, which are common side reactions in uncontrolled halogenations. The patent data indicates that maintaining the temperature below 55°C is sufficient to drive the reaction to completion within 2 to 6 hours, demonstrating excellent kinetic control.
Following the isolation of the 5-halo intermediate, the second step involves a classic Halex (halogen-exchange) reaction. In this nucleophilic aromatic substitution, the electron-withdrawing acetyl group ortho to the leaving halogen activates the ring towards attack by the fluoride ion. The use of polar aprotic solvents like DMSO or DMF is critical here, as they effectively solvate the cation (potassium) while leaving the fluoride anion "naked" and highly nucleophilic. The addition of a copper fluoride co-catalyst, as noted in the examples, likely assists in the activation of the carbon-halogen bond, further enhancing the conversion rate to over 95%. This mechanistic pathway ensures that impurities arising from thermal decomposition are minimized, directly contributing to the high-purity 5-fluoro-2-hydroxyacetophenone required for downstream pharmaceutical applications. The ability to recrystallize the final product from simple hydrocarbons like petroleum ether confirms the low level of polar impurities generated during the reaction.
How to Synthesize 5-Fluoro-2-Hydroxyacetophenone Efficiently
Implementing this novel synthesis requires careful attention to reagent addition rates and temperature control to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for specialized high-pressure or cryogenic equipment, which makes it highly attractive for reducing lead time for high-purity intermediates. The following overview summarizes the critical operational phases derived from the patent examples, detailing the transition from raw material to the final crystalline solid. For a comprehensive standard operating procedure including exact molar ratios and safety protocols, please refer to the technical guide below.
- Perform electrophilic halogenation of o-hydroxyacetophenone using chlorine or bromine with an iron catalyst at 0-55°C to obtain 5-halo-2-hydroxyacetophenone.
- React the 5-halo intermediate with a fluorinating agent like potassium fluoride in a polar aprotic solvent such as DMSO or DMF under reflux.
- Quench the reaction, remove inorganic salts, and purify the crude product via recrystallization using petroleum ether or heptane to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages that extend beyond simple unit price reductions. The shift away from exotic starting materials like p-fluorophenol to commodity chemicals like o-hydroxyacetophenone fundamentally de-risks the supply chain. Since o-hydroxyacetophenone is produced on a massive scale for various industries, its availability is stable, and its price is subject to less volatility than specialized fluorinated aromatics. This stability ensures consistent production scheduling and mitigates the risk of raw material shortages that often plague niche intermediate manufacturing. Furthermore, the simplified reaction conditions reduce the dependency on specialized reactor types, allowing for greater flexibility in manufacturing site selection.
- Cost Reduction in Manufacturing: The elimination of stoichiometric aluminum trichloride and the avoidance of high-temperature operations result in substantial cost savings. Traditional methods require significant energy input to maintain temperatures above 130°C and incur high costs for neutralizing the acidic off-gases and treating saline wastewater. By operating at mild temperatures and avoiding aluminum salts, this new process drastically lowers utility costs and waste disposal fees. The removal of expensive metal scavengers typically needed to meet residual metal specifications in APIs further enhances the economic viability, providing a clear path for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including chlorine, bromine, and potassium fluoride, are bulk industrial chemicals with robust global supply networks. Unlike specialized catalysts or custom-synthesized starting materials, these inputs are unlikely to face supply disruptions. The robustness of the chemistry, demonstrated by high conversion rates (>95%) and yields (>80%), ensures predictable output volumes. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing production stoppages due to intermediate shortages and fostering long-term partnerships with a reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process is superior. The absence of high-salt wastewater and corrosive HCl gas simplifies the permitting process for new production lines and reduces the burden on existing effluent treatment plants. The mild reaction conditions minimize the risk of thermal runaways, enhancing plant safety. These factors make the process highly scalable from pilot kilogram batches to multi-ton commercial production without requiring disproportionate increases in environmental control infrastructure. This scalability supports the commercial scale-up of complex intermediates while adhering to increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary cost advantages of this new synthesis route compared to Fries rearrangement?
A: The new process utilizes o-hydroxyacetophenone, which is significantly cheaper and more readily available than p-fluorophenol or p-fluoroanisole used in conventional methods. Additionally, it eliminates the need for excessive amounts of anhydrous aluminum trichloride and avoids high-temperature conditions above 130°C, reducing energy consumption and equipment corrosion costs.
Q: How does this process address environmental concerns regarding wastewater treatment?
A: Conventional Fries rearrangement generates large volumes of high-salt wastewater due to the hydrolysis of aluminum complexes. This novel halogenation-fluorination route avoids aluminum salts entirely, resulting in substantially reduced saline waste and eliminating the release of hazardous hydrogen chloride gas during the quenching phase.
Q: What purity levels can be achieved with the described recrystallization method?
A: According to the patent data, the final product obtained after recrystallization from solvents like petroleum ether or n-heptane consistently achieves a GC purity of over 98%, with specific examples demonstrating purity levels reaching 99%. The mild reaction conditions also prevent high-temperature carbonization, ensuring a light-colored product that meets stringent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Fluoro-2-Hydroxyacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel process are fully realized at an industrial level. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this halogenation-fluorination sequence safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for beta-blocker intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can enhance your production efficiency and reduce overall manufacturing costs.
