Advanced Synthesis of N-Substituted Hydroxycycloalkylamine Derivatives for Commercial Scale-Up
The landscape of pharmaceutical intermediate synthesis has been significantly altered by the transformative methodologies disclosed within patent CN1323293A, which introduces a robust pathway for the preparation of N-substituted hydroxycycloalkylamine derivatives. This intellectual property outlines a sophisticated chemical strategy that leverages 1,2-cyclosulfinyl alkyl halides as pivotal intermediates, effectively bridging the gap between simple polyol starting materials and complex heterocyclic amines required for modern drug development. By utilizing 1,2-dihydroxyalkyl alcohols as the foundational feedstock, the invention achieves a level of regioselectivity and stereoselectivity that was previously difficult to attain without resorting to prohibitively expensive enzymatic resolutions or multi-step protection-deprotection sequences. The technical breakthrough lies in the differential activation of hydroxyl groups, where the adjacent diols are selectively converted into a cyclic sulfite moiety, leaving the terminal hydroxyl group available for specific halogenation and subsequent nucleophilic displacement. This approach not only streamlines the synthetic route but also ensures that the chiral integrity of the molecule is preserved throughout the transformation, a critical parameter for the production of optically active pharmaceutical ingredients such as carbapenem antibiotics and antihypertensives. For industry stakeholders, this patent represents a viable solution to the longstanding challenges of yield optimization and impurity control in the manufacturing of high-value nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-substituted-3-hydroxypyrrolidine derivatives has been plagued by inefficient synthetic routes that rely on harsh reagents and complex purification protocols, creating substantial bottlenecks for reliable pharmaceutical intermediate supplier operations. One of the most prevalent traditional methods involves the reduction of N-benzyl-3-hydroxysuccinimide using lithium aluminum hydride (LAH), a reagent that is not only costly but also poses significant safety hazards due to its pyrophoric nature and sensitivity to moisture. Furthermore, alternative pathways involving the decarboxylation of hydroxy-proline derivatives or the enzymatic resolution of racemic mixtures often suffer from low overall yields and require specialized biocatalysts that are difficult to recover and recycle on a commercial scale. The reliance on expensive brominating agents for selective halogenation of triols, as seen in older literature, frequently results in poor regiocontrol, leading to the formation of unwanted trihaloalkane by-products that are arduous to separate from the desired intermediate. These conventional processes often necessitate cryogenic conditions or high-pressure reactors, driving up capital expenditure and operational complexity while failing to guarantee the high optical purity demanded by regulatory agencies for final drug substances. Consequently, the cumulative effect of these limitations is a supply chain that is vulnerable to disruptions, with extended lead times and inflated production costs that ultimately erode profit margins for downstream API manufacturers.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a thionyl halide-mediated activation strategy that fundamentally simplifies the molecular architecture assembly while drastically enhancing process efficiency. By reacting 1,2-dihydroxyalkyl alcohols with thionyl chloride or bromide in the presence of a mild base catalyst such as pyridine, the synthesis creates a stable 1,2-cyclosulfinyl alkyl halide intermediate that serves as a highly reactive electrophile for subsequent amination. This method allows for the precise differentiation of hydroxyl reactivity, ensuring that the nucleophilic amine attacks the terminal carbon bearing the halogen rather than the cyclic sulfite protected positions, thereby achieving exceptional regioselectivity. The process operates under relatively mild thermal conditions, typically ranging from -20°C to 10°C for intermediate formation and reflux temperatures for cyclization, which reduces energy consumption and minimizes the risk of thermal degradation of sensitive functional groups. Moreover, the by-products generated during the reaction, primarily sulfur dioxide and hydrogen halides, are gaseous or easily neutralized, simplifying waste treatment and aligning with modern green chemistry principles for cost reduction in API manufacturing. The ability to isolate the intermediate in high purity via simple vacuum distillation further empowers manufacturers to implement a modular production strategy, where the intermediate can be stored and transported, decoupling the upstream alcohol processing from the downstream amine cyclization steps.
Mechanistic Insights into Thionyl Halide-Mediated Cyclization
The core mechanistic advantage of this technology resides in the unique electronic properties of the 1,2-cyclosulfinyl group, which acts as a temporary protecting group that simultaneously activates the adjacent carbon framework for nucleophilic substitution. When the 1,2-dihydroxyalkyl alcohol reacts with thionyl halide, the two vicinal hydroxyl groups form a five-membered cyclic sulfite ring, which exerts a strong electron-withdrawing effect that stabilizes the molecule against unwanted side reactions at the C1 and C2 positions. Concurrently, the terminal hydroxyl group at the end of the alkyl chain is converted into a halide, creating a primary electrophilic site that is significantly more susceptible to nucleophilic attack than the secondary carbons within the cyclic sulfite ring. This differential reactivity is the key to the high regioselectivity observed, as the incoming amine nucleophile preferentially displaces the terminal halogen to form a secondary amine, which then undergoes an intramolecular cyclization by attacking the electrophilic sulfur or carbon centers of the cyclic sulfite moiety. The release of sulfur dioxide gas during the final ring-closing step provides a strong thermodynamic driving force that pushes the equilibrium towards the formation of the desired N-substituted hydroxycycloalkylamine, effectively suppressing the formation of azetidine or aziridine by-products. 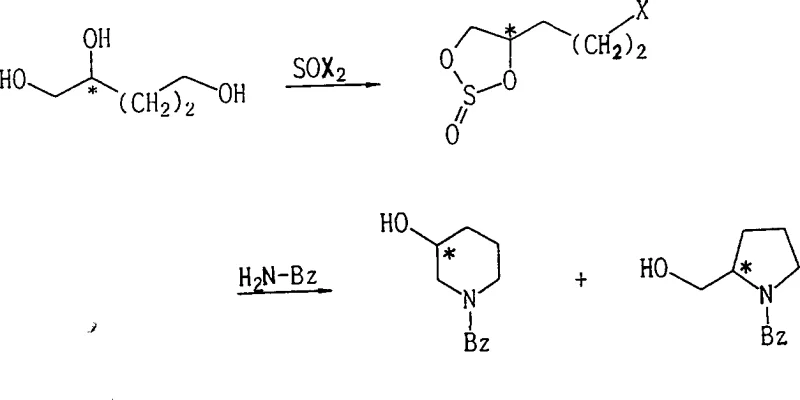
From an impurity control perspective, the stability of the 1,2-cyclosulfinyl alkyl halide intermediate is paramount, as it allows for rigorous quality assurance testing before the final cyclization step is initiated. Unlike transient intermediates in LAH reductions that must be consumed immediately, this halide species remains stable at room temperature for several months, enabling manufacturers to identify and remove any trace isomers or unreacted starting materials prior to the critical ring-closing reaction. This capability is essential for maintaining the stringent purity specifications required for high-purity OLED material or pharmaceutical intermediate production, where even minor structural analogs can act as potent catalyst poisons or toxic impurities in the final drug product. The stereoselectivity is inherently preserved because the reaction proceeds through an SN2-type displacement mechanism at the chiral center, which, when starting from optically pure 1,2-dihydroxyalkyl alcohols, ensures inversion of configuration without racemization. By carefully controlling the stoichiometry of the thionyl halide to prevent over-halogenation of the cyclic sulfite ring, the process minimizes the generation of trihaloalkane impurities, resulting in a cleaner reaction profile that simplifies downstream crystallization and distillation operations.
How to Synthesize N-Substituted Hydroxycycloalkylamine Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to precise operational parameters regarding solvent selection, temperature control, and reagent stoichiometry to maximize yield and purity. The process begins with the dissolution of the 1,2-dihydroxyalkyl alcohol in an aprotic polar solvent such as acetonitrile or dichloromethane, followed by the controlled addition of a base catalyst like pyridine to scavenge the acidic by-products generated during the reaction.
- React 1,2-dihydroxyalkyl alcohol with thionyl halide and base catalyst at low temperature to form 1,2-cyclosulfinyl alkyl halide.
- Isolate the stable intermediate via vacuum distillation or chromatography to ensure high purity before cyclization.
- Perform nucleophilic ring substitution with an amine compound under reflux conditions to yield the final N-substituted hydroxycycloalkylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this thionyl halide-based synthesis route offers profound strategic benefits that extend far beyond simple chemical yield improvements, directly impacting the bottom line through operational excellence. The elimination of hazardous reducing agents like lithium aluminum hydride removes a significant cost center associated with specialized handling equipment, safety training, and hazardous waste disposal, leading to substantial cost savings in facility maintenance and regulatory compliance. Furthermore, the use of commodity chemicals such as thionyl chloride and pyridine, which are readily available in bulk quantities from global chemical suppliers, mitigates the risk of raw material shortages that often plague processes relying on bespoke enzymes or exotic metal catalysts. The robustness of the intermediate allows for the creation of strategic inventory buffers, ensuring that production schedules for critical API intermediates can be maintained even during fluctuations in upstream feedstock availability or unexpected maintenance downtime. This resilience translates into enhanced supply chain reliability, providing downstream partners with the confidence of consistent delivery timelines and reducing the need for expensive expedited shipping or safety stock premiums.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents with cost-effective thionyl halides significantly lowers the direct material cost per kilogram of the final product, while the simplified workup procedures reduce labor and utility expenses associated with complex purification steps. By avoiding the use of precious metal catalysts or biocatalysts that require strict temperature and pH controls, the process reduces the capital intensity of the manufacturing plant, allowing for higher throughput in existing reactor trains without major retrofitting investments. The high atom economy of the reaction, driven by the evolution of gaseous sulfur dioxide which leaves the reaction mixture spontaneously, minimizes the volume of liquid waste streams that require costly treatment and disposal, further contributing to overall operational expenditure reduction. Additionally, the ability to recycle solvents like acetonitrile and dichloromethane through standard distillation columns enhances the economic viability of the process, making it highly competitive for large-scale commercial production of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The stability of the 1,2-cyclosulfinyl alkyl halide intermediate serves as a critical buffer in the supply chain, allowing manufacturers to produce and store key building blocks in advance of demand spikes for the final active pharmaceutical ingredient. This decoupling of synthesis steps means that delays in the final amination stage do not halt the entire production line, as the intermediate can be stockpiled safely, ensuring continuous operation and maximizing asset utilization rates. The reliance on widely available industrial chemicals rather than niche specialty reagents reduces the vulnerability of the supply chain to geopolitical disruptions or single-source supplier failures, fostering a more resilient and diversified procurement strategy. Moreover, the predictable reaction kinetics and high reproducibility of the process facilitate accurate demand forecasting and production planning, enabling supply chain heads to optimize inventory levels and reduce working capital tied up in unfinished goods.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production due to the manageable exotherm of the thionyl halide reaction and the absence of sensitive catalytic systems that often fail upon scale-up. The generation of sulfur dioxide as a by-product, while requiring appropriate scrubbing systems, is a well-understood industrial challenge with established abatement technologies, ensuring full compliance with environmental regulations regarding air emissions and effluent discharge. The high selectivity of the reaction minimizes the formation of complex organic by-products, reducing the load on wastewater treatment facilities and lowering the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly becoming a key criterion for selection by major multinational pharmaceutical companies seeking responsible commercial scale-up of complex polymer additives and fine chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing manufacturing portfolios. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, highlighting the practical advantages over legacy methods.
Q: What are the advantages of using 1,2-cyclosulfinyl alkyl halides over traditional LAH reduction methods?
A: The use of 1,2-cyclosulfinyl alkyl halides eliminates the need for hazardous and expensive reducing agents like lithium aluminum hydride (LAH), significantly improving operational safety and reducing raw material costs while maintaining high stereochemical integrity.
Q: Does this synthesis method preserve optical purity during the cyclization step?
A: Yes, the process is designed to be highly stereoselective, ensuring that optically active starting materials yield optically pure products without racemization at the chiral center, which is critical for pharmaceutical applications.
Q: How does the stability of the intermediate impact supply chain reliability?
A: The 1,2-cyclosulfinyl alkyl halide intermediate is stable at room temperature for several months, allowing manufacturers to stockpile key intermediates and decouple production steps, thereby enhancing supply continuity and reducing lead times for final API delivery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Hydroxycycloalkylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes like the one described in CN1323293A to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the absence of regioisomers and optical impurities in every batch. Our infrastructure is designed to handle the specific safety requirements of thionyl halide chemistry, providing a secure and compliant environment for the manufacture of these valuable pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs, offering a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior synthetic route. By collaborating with us, you gain access to specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality N-substituted hydroxycycloalkylamine derivatives on schedule and within budget. Let us help you optimize your supply chain and reduce your manufacturing costs with our proven expertise in regioselective cyclization technologies.
