Advanced Asymmetric Catalysis for High-Purity Benazepril Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust pathways for cardiovascular therapeutics, particularly for Angiotensin Converting Enzyme (ACE) inhibitors like Benazepril Hydrochloride. A pivotal development in this domain is documented in Chinese patent CN113292495B, which discloses a novel synthesis method for the key chiral intermediate, (S)-3-amino-2,3,4,5-tetrahydro-2-oxo-1H-1-benzazepine-1-acetic acid tert-butyl ester. This specific intermediate is critical for constructing the active (S,S)-configuration of Benazepril, ensuring optimal therapeutic efficacy. The technical breakthrough lies in replacing traditional resolution techniques with a direct asymmetric catalytic transamination strategy. 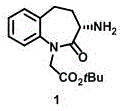 By establishing the chiral center directly through organocatalysis rather than separating racemates, the process fundamentally alters the economic and operational landscape for manufacturers. This report analyzes the technical depth of this innovation, providing actionable insights for R&D directors and supply chain leaders aiming to optimize their API production pipelines.
By establishing the chiral center directly through organocatalysis rather than separating racemates, the process fundamentally alters the economic and operational landscape for manufacturers. This report analyzes the technical depth of this innovation, providing actionable insights for R&D directors and supply chain leaders aiming to optimize their API production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this benzazepine derivative has been plagued by inherent inefficiencies associated with classical resolution strategies. For instance, US patent US4575503 describes a route utilizing 3-chloro-benzazepine substrates subjected to azidation and subsequent resolution. 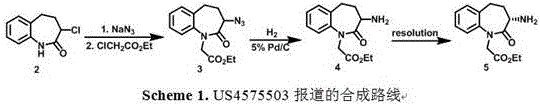 This approach presents severe industrial hazards due to the use of sodium azide, an explosive raw material that complicates safety protocols and increases insurance and handling costs. Furthermore, other reported methods, such as those in CN1844102 and CN101538242, rely on bromo-substituted precursors and chiral resolution using tartaric acid or Cbz-L-phenylalanine. These resolution-based processes suffer from a theoretical maximum yield of 50% for the desired enantiomer, effectively wasting half of the synthesized material. Additionally, the requirement for stoichiometric amounts of expensive chiral resolving agents significantly inflates the raw material costs, making these routes less competitive for large-scale generic drug manufacturing where margin compression is a constant pressure.
This approach presents severe industrial hazards due to the use of sodium azide, an explosive raw material that complicates safety protocols and increases insurance and handling costs. Furthermore, other reported methods, such as those in CN1844102 and CN101538242, rely on bromo-substituted precursors and chiral resolution using tartaric acid or Cbz-L-phenylalanine. These resolution-based processes suffer from a theoretical maximum yield of 50% for the desired enantiomer, effectively wasting half of the synthesized material. Additionally, the requirement for stoichiometric amounts of expensive chiral resolving agents significantly inflates the raw material costs, making these routes less competitive for large-scale generic drug manufacturing where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the methodology outlined in CN113292495B introduces a streamlined five-step sequence that bypasses these historical bottlenecks. The process initiates with a simple Aldol condensation between 2-aminobenzaldehyde and pyruvic acid, followed by catalytic hydrogenation and a DCC-mediated cyclization to form the core benzazepine scaffold. 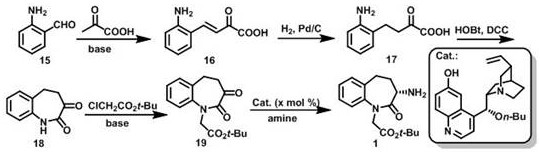 The true innovation occurs in the final step, where a quinine-derived organocatalyst facilitates an asymmetric transamination reaction. This step directly installs the amino group with high stereoselectivity, achieving enantiomeric excess (ee) values as high as 99.5%. By constructing the chiral center directly, the process eliminates the need for resolution, theoretically doubling the yield compared to racemic separation methods. This shift from stoichiometric resolution to catalytic asymmetry represents a paradigm shift in process chemistry, aligning with green chemistry principles by reducing waste generation and improving atom economy.
The true innovation occurs in the final step, where a quinine-derived organocatalyst facilitates an asymmetric transamination reaction. This step directly installs the amino group with high stereoselectivity, achieving enantiomeric excess (ee) values as high as 99.5%. By constructing the chiral center directly, the process eliminates the need for resolution, theoretically doubling the yield compared to racemic separation methods. This shift from stoichiometric resolution to catalytic asymmetry represents a paradigm shift in process chemistry, aligning with green chemistry principles by reducing waste generation and improving atom economy.
Mechanistic Insights into Quinine-Derived Asymmetric Transamination
The core of this technological advancement is the specialized organocatalyst derived from quinine, which orchestrates the stereochemical outcome of the transamination reaction. The catalyst functions by activating the ketone substrate (compound 19) through hydrogen bonding interactions while simultaneously coordinating the amine donor. This dual activation creates a highly organized transition state that favors the formation of the (S)-enantiomer over the (R)-enantiomer. The patent details the preparation of this catalyst through the alkylation of quinine followed by demethylation, resulting in a structure capable of precise steric control. 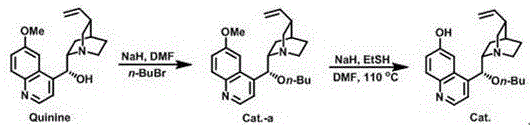 The reaction proceeds under mild conditions, typically between 0-10°C, which helps suppress background non-catalyzed reactions that could lead to racemization. The use of o-chlorobenzylamine as the amine source is particularly effective, likely due to its electronic properties which enhance nucleophilicity without compromising the stability of the intermediate imine species.
The reaction proceeds under mild conditions, typically between 0-10°C, which helps suppress background non-catalyzed reactions that could lead to racemization. The use of o-chlorobenzylamine as the amine source is particularly effective, likely due to its electronic properties which enhance nucleophilicity without compromising the stability of the intermediate imine species.
From an impurity control perspective, this catalytic mechanism offers superior predictability compared to resolution. In resolution processes, the mother liquor contains significant amounts of the unwanted enantiomer, which can be difficult to purge completely and may lead to cross-contamination in subsequent batches. Here, the high enantioselectivity (up to 99.5% ee) ensures that the crude product is already of high optical purity, simplifying downstream purification. The reaction solvent, typically dichloromethane or ethyl acetate, allows for easy workup, and the catalyst loading is kept low (0.5-5 mol%), minimizing the burden of removing metal or organic residues. This level of control is essential for meeting the stringent regulatory requirements for chiral pharmaceutical intermediates, where even trace amounts of the wrong enantiomer can be toxicologically significant.
How to Synthesize (S)-Amino Benazepril Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the cyclization and asymmetric steps. The initial Aldol condensation is exothermic and requires temperature control between 20-30°C to prevent polymerization of the aldehyde. Subsequent hydrogenation uses standard Pd/C catalysts at moderate pressures (0.2 MPa), a unit operation familiar to most multipurpose plants. The cyclization step utilizes DCC and HOBt, necessitating efficient filtration to remove dicyclohexylurea byproducts.
- Perform Aldol condensation of 2-aminobenzaldehyde with pyruvic acid under alkaline conditions to form compound 16.
- Execute Pd/C catalytic hydrogenation of compound 16 to generate the saturated intermediate compound 17.
- Conduct DCC-mediated self-condensation of compound 17 to construct the benzazepine ring system (compound 18).
- Alkylate compound 18 with tert-butyl chloroacetate to install the ester side chain, yielding compound 19.
- Complete the synthesis via asymmetric transamination of compound 19 using a quinine-derived catalyst to obtain the final chiral amino compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic route offers tangible strategic benefits beyond mere technical elegance. The elimination of resolution steps fundamentally changes the cost structure of the intermediate, removing the need to purchase and recover expensive chiral acids or bases. Furthermore, the avoidance of hazardous azides reduces the regulatory burden and safety infrastructure costs associated with handling explosive precursors. This route enables a more resilient supply chain by relying on commodity chemicals like 2-aminobenzaldehyde and pyruvic acid, which are widely available and less prone to supply shocks compared to specialized chiral pool starting materials.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in yield efficiency. By abandoning the 50% yield ceiling imposed by resolution, the overall throughput of the manufacturing plant is effectively doubled for the same amount of starting material input. Additionally, the use of a catalytic amount of chiral inducer rather than a stoichiometric resolver significantly lowers the bill of materials cost. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to reduced utility costs per kilogram of product. These factors combined result in substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the starting materials are bulk commodities rather than niche chiral building blocks. 2-Aminobenzaldehyde and tert-butyl chloroacetate are produced by numerous global suppliers, reducing the risk of single-source dependency. The simplified process flow, with fewer unit operations and no complex resolution crystallizations, shortens the overall cycle time from raw material intake to finished goods. This agility allows manufacturers to respond more quickly to fluctuations in market demand for Benazepril, ensuring consistent delivery schedules to downstream API formulators.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor types and avoiding exotic conditions that are difficult to replicate at the 100 MT scale. From an environmental standpoint, the reduction in waste is significant; eliminating the mother liquor from resolution steps cuts solvent waste volumes drastically. The absence of heavy metal catalysts (using organic catalysis instead) and explosive azides simplifies wastewater treatment and effluent compliance. This alignment with green chemistry principles not only reduces disposal costs but also enhances the sustainability profile of the final pharmaceutical product, a growing requirement for major multinational buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer.
Q: How does this new method improve upon traditional resolution processes?
A: Traditional methods rely on resolution which theoretically limits yield to 50% and requires expensive chiral resolving agents. This patent utilizes direct asymmetric catalysis, achieving up to 99.5% ee and significantly higher overall yields by avoiding the discard of the unwanted enantiomer.
Q: What are the safety advantages regarding raw materials?
A: Prior art routes often utilize sodium azide, an explosive hazard difficult to manage on an industrial scale. This new protocol avoids azides entirely, relying on safer reagents like pyruvic acid and tert-butyl chloroacetate, thereby enhancing industrial safety profiles.
Q: Is the quinine-derived catalyst reusable or expensive?
A: The catalyst is used in catalytic amounts (0.5-5 mol%), which is far more economical than stoichiometric chiral auxiliaries. While the preparation involves specific modification of quinine, the low loading factor drastically reduces the cost contribution per kilogram of product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Amino Benazepril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric catalytic route described in CN113292495B for the production of high-purity cardiovascular intermediates. Our R&D team has extensively evaluated similar organocatalytic pathways and possesses the expertise to adapt this chemistry for robust commercial manufacturing. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity observed in the lab is maintained at the plant scale. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the 99.5% ee target is consistently met.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their Benazepril supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this catalytic route for your volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and detailed route feasibility assessments tailored to your project timelines.
