Advanced Synthetic Route for 3 Alpha-Hydroxy-5 Alpha-Cholanic Acid Enhancing Commercial Viability
Advanced Synthetic Route for 3 Alpha-Hydroxy-5 Alpha-Cholanic Acid Enhancing Commercial Viability
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for complex steroid intermediates, particularly those with specific stereochemical configurations that are historically difficult to access. A pivotal development in this domain is documented in patent CN109134577B, which discloses a highly efficient synthetic method for 3 alpha-hydroxy-5 alpha-cholanic acid, a critical isomer of lithocholic acid. This compound holds immense potential not only as a diagnostic marker for liver diseases but also as a versatile scaffold for synthesizing novel steroid derivatives with tumor inhibition properties. The disclosed technology addresses a significant gap in the prior art by providing a stable, reproducible route that bypasses the instability issues associated with traditional strong acid or base treatments which often compromise the chiral integrity of the steroid nucleus.
For R&D directors and process chemists, the significance of this patent lies in its ability to transform a scarce, high-value isomer into a commercially viable commodity. The method leverages hyodeoxycholic acid, a cheap and abundant natural product, as the starting material, thereby shifting the economic balance of production favorably. By achieving a total yield of up to 47% through a concise four-step sequence, this innovation offers a compelling alternative to legacy processes that were plagued by low throughput and excessive waste generation. As a reliable pharmaceutical intermediates supplier, understanding and adopting such patented efficiencies is crucial for maintaining competitiveness in the global supply chain for bile acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lithocholic acid and its isomers has been fraught with synthetic inefficiencies and stereochemical pitfalls. As early as 1946, literature described a laborious seven-step pathway starting from deoxycholic acid, which necessitated methyl esterification followed by multiple hydroxyl protection and deprotection cycles. This conventional approach, often involving benzoyl protection groups, was not only operationally cumbersome but also suffered from poor atom economy. The requirement to selectively remove protecting groups under harsh conditions frequently led to epimerization at the C-5 position, resulting in a mixture of 5-alpha and 5-beta isomers that were difficult to separate. Furthermore, the reliance on multiple chromatographic purifications and the use of stoichiometric amounts of expensive protecting reagents drove up the cost of goods significantly, making large-scale production economically unfeasible for many applications.
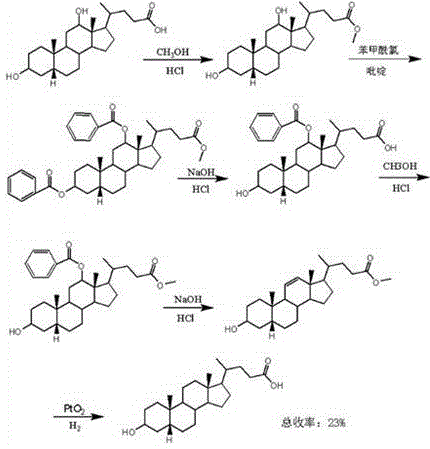
The structural complexity of the steroid backbone means that every additional synthetic step introduces a risk of yield loss and impurity formation. In the traditional seven-step route, the cumulative yield often dropped to negligible levels, rendering the final product prohibitively expensive for widespread research or clinical use. Additionally, the environmental footprint of such a process was substantial, generating significant volumes of organic solvent waste and hazardous byproducts from the protection-deprotection sequences. For procurement managers, these factors translated into volatile pricing and unreliable supply continuity, as few manufacturers were willing or able to commit to such inefficient production schedules. The inability to stably obtain the specific 3 alpha-hydroxy-5 alpha-cholanic acid isomer without contamination from its 5-beta counterpart remained a persistent bottleneck in the field of bile acid chemistry.
The Novel Approach
In stark contrast to the convoluted legacy methods, the novel approach detailed in the patent utilizes a strategic four-step cascade that maximizes efficiency while minimizing structural manipulation. By selecting hyodeoxycholic acid as the precursor, the synthesis capitalizes on the existing oxygenation pattern of the starting material, requiring only modification at the C-3 and C-6 positions rather than building the functionality from scratch. The process initiates with a straightforward esterification to protect the C-24 carboxyl group, followed by a dual oxidation that simultaneously converts the 3-alpha and 6-alpha hydroxyls into carbonyls. This is followed by a highly selective reduction that restores the 3-alpha hydroxyl group while leaving the 6-keto group intact for the final elimination step. This logical progression eliminates the need for orthogonal protecting groups entirely, streamlining the workflow and drastically reducing the number of unit operations required.
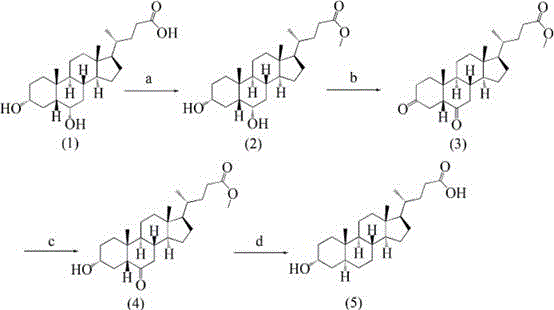
The culmination of this route is a xanthate reduction (a variation of the Wolff-Kishner reduction) that cleanly removes the C-6 oxygen functionality to yield the desired 5-alpha configuration. This final step is critical as it establishes the thermodynamic stability of the 5-alpha isomer without inducing the epimerization seen in older acid-catalyzed hydrogenation methods. The result is a process that delivers a total yield of approximately 47%, a figure that represents a monumental improvement over historical benchmarks. For supply chain heads, this translates to a more predictable manufacturing timeline and a substantial reduction in raw material consumption per kilogram of finished product. The simplicity of the route also facilitates easier technology transfer and scale-up, allowing manufacturers to respond more agilely to market demand for high-purity steroid intermediates.
Mechanistic Insights into the Oxidation-Reduction Cascade
The core chemical innovation of this synthesis lies in the precise control of oxidation states at the C-3 and C-6 positions of the steroid nucleus. In the second step, the use of oxidants such as Jones reagent (chromic acid) or PCC allows for the simultaneous conversion of both secondary alcohols into ketones. This double oxidation is mechanistically favorable because the hyodeoxycholic acid substrate presents both hydroxyl groups in an axial or equatorial orientation that is accessible to the oxidant, depending on the specific conformational flexibility of the A and B rings. The choice of solvent, typically acetone or dichloromethane, plays a vital role in solubilizing the lipophilic steroid while maintaining the reactivity of the oxidizing species. Controlling the temperature at 0°C during this phase is essential to prevent over-oxidation or degradation of the sensitive steroid skeleton, ensuring that the reaction stops cleanly at the dione stage.
Following oxidation, the selective reduction step employs mild reducing agents like sodium borohydride to differentiate between the C-3 and C-6 carbonyls. Although both are ketones, the steric environment around the C-3 position often favors the formation of the 3-alpha alcohol due to the approach of the hydride ion from the less hindered face of the molecule. This stereoselectivity is paramount for obtaining the correct biological activity in the final product. The subsequent xanthate reduction involves the formation of a thiocarbonate intermediate which, upon heating with hydrazine hydrate and a strong base like potassium hydroxide in a high-boiling solvent like diethylene glycol, undergoes homolytic cleavage to extrude nitrogen and sulfur gases. This radical mechanism effectively deoxygenates the C-6 position while locking the C-5 proton into the stable alpha-configuration, thereby preventing the thermodynamic equilibration to the 5-beta isomer that plagues other methods.
How to Synthesize 3 Alpha-Hydroxy-5 Alpha-Cholanic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the oxidation and reduction phases to maintain stereochemical integrity. The process is designed to be robust, utilizing common laboratory reagents that are readily available on an industrial scale, thus lowering the barrier to entry for manufacturing. Operators should monitor the reaction progress via HPLC or TLC to ensure complete conversion at each stage, especially during the selective reduction where over-reduction could lead to the formation of unwanted diol byproducts. The workup procedures involve standard extraction and washing protocols, making the process compatible with existing infrastructure in most fine chemical plants. For a detailed breakdown of the specific molar ratios, solvent volumes, and reaction times optimized in the patent examples, please refer to the standardized guide below.
- Perform esterification of hyodeoxycholic acid with methanol under acidic conditions to protect the carboxyl group.
- Execute a double oxidation reaction using an oxidant like Jones reagent to convert 3-alpha and 6-alpha hydroxyl groups into carbonyls.
- Conduct a selective reduction using sodium borohydride to restore the 3-alpha hydroxyl configuration while maintaining the 6-keto group.
- Finalize the synthesis via a xanthate reduction (Wolff-Kishner variant) using hydrazine hydrate and alkali to remove the 6-keto oxygen.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this four-step synthesis offers transformative benefits for cost structures and supply chain resilience in the steroid intermediate market. The shift from a seven-step to a four-step process inherently reduces the cumulative processing time, labor costs, and utility consumption associated with manufacturing. By eliminating the need for expensive protecting group reagents and the solvents required for their installation and removal, the variable cost per kilogram of product is significantly lowered. This efficiency gain allows suppliers to offer more competitive pricing models to downstream pharmaceutical clients, potentially expanding the addressable market for lithocholic acid derivatives in oncology and diagnostics. Furthermore, the use of hyodeoxycholic acid, a bulk commodity chemical, as the starting material insulates the supply chain from the volatility associated with specialized, low-volume precursors.
- Cost Reduction in Manufacturing: The streamlined nature of the synthetic route directly correlates to reduced operational expenditures. By cutting the number of isolation and purification steps nearly in half, manufacturers can achieve substantial savings in solvent usage and waste disposal fees, which are often the hidden drivers of high production costs in organic synthesis. The higher overall yield of 47% means that less raw material is required to produce the same amount of final product, further enhancing the margin profile. Additionally, the avoidance of precious metal catalysts or exotic reagents ensures that the cost of goods remains stable and predictable, shielding buyers from fluctuations in the prices of rare catalytic metals.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway enhances the reliability of supply for critical API intermediates. Because the reaction conditions are relatively mild and utilize standard equipment, the risk of batch failure due to equipment incompatibility or extreme process parameters is minimized. The availability of hyodeoxycholic acid from multiple global sources ensures that raw material shortages are unlikely to disrupt production schedules. This stability is crucial for pharmaceutical companies that require consistent, long-term supply agreements to support their own clinical trials and commercial launches, reducing the risk of stockouts that could delay time-to-market for new therapies.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production without the need for specialized high-pressure or cryogenic equipment. The reduction in chemical steps also aligns with green chemistry principles by lowering the E-factor (mass of waste per mass of product), facilitating easier compliance with increasingly stringent environmental regulations. The solvents used, such as methanol, acetone, and ethyl acetate, are widely recycled in industrial settings, further minimizing the environmental footprint. This sustainability profile is increasingly becoming a key differentiator for procurement teams evaluating suppliers based on ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3 alpha-hydroxy-5 alpha-cholanic acid. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances helps stakeholders make informed decisions regarding the integration of this intermediate into their broader drug development pipelines.
Q: Why is the synthesis of 3 alpha-hydroxy-5 alpha-cholanic acid considered challenging?
A: The challenge lies in the stereochemical control of the 5-H position. Conventional strong acid or base conditions often lead to configuration transformation at the chiral centers, specifically epimerization at C-5, making it difficult to stably obtain the 5-alpha isomer without significant impurities.
Q: What represents the primary advantage of the new 4-step method over historical routes?
A: The primary advantage is the drastic reduction in synthetic steps from seven to four. By utilizing hyodeoxycholic acid as a readily available starting material and employing a strategic oxidation-reduction sequence, the new method eliminates complex protection and deprotection cycles, significantly improving overall yield and operational efficiency.
Q: How does this synthesis method impact the purity profile of the final API intermediate?
A: The method utilizes highly selective reagents, such as sodium borohydride for specific carbonyl reduction and controlled Wolff-Kishner conditions, which minimize side reactions. This selectivity ensures a cleaner impurity profile, reducing the burden on downstream purification processes and ensuring high-purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3 Alpha-Hydroxy-5 Alpha-Cholanic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality bile acid intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3 alpha-hydroxy-5 alpha-cholanic acid meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that our clients receive materials with consistent impurity profiles, facilitating smoother regulatory filings and faster clinical progression.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthetic route can be leveraged to optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient manufacturing protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring a successful and profitable collaboration in the competitive landscape of fine chemical manufacturing.
