Scalable Reductive Coupling Technology for High-Purity 5,5'-Diaminobiphenyl Derivatives
Introduction to Advanced Reductive Coupling Technology
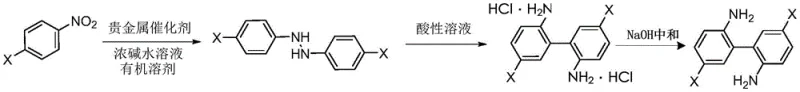
The chemical industry is constantly seeking more efficient pathways to synthesize complex biaryl diamines, which serve as critical building blocks for high-performance polymers and pharmaceutical agents. Patent CN111574380B introduces a groundbreaking method for preparing 5,5'-disubstituted-2,2'-diaminobiphenyl and its hydrochloride salts through a novel reductive coupling strategy. Unlike traditional approaches that rely on multi-step functionalization, this technology leverages the direct catalytic hydrogenation of readily available 4-substituted nitrobenzenes. The process initiates with the formation of a 1,2-diphenylhydrazine intermediate under mild alkaline conditions, followed by a precise acid-mediated rearrangement to construct the biphenyl core. This innovation represents a significant leap forward in synthetic efficiency, offering a robust alternative to legacy methods that often suffer from low atom economy and high operational complexity.
For R&D directors and process chemists, the implications of this patent are profound, as it simplifies the supply chain for key intermediates used in polyimide and polyurethane production. The methodology described in CN111574380B utilizes standard industrial equipment such as stainless steel autoclaves, making it highly adaptable for existing manufacturing facilities. By shifting the synthetic logic from cross-coupling of halides to the reductive dimerization of nitro compounds, the process inherently reduces the generation of stoichiometric metal waste. This alignment with green chemistry principles not only addresses environmental compliance concerns but also streamlines the downstream purification workflow, ensuring that the final diaminobiphenyl products meet the rigorous quality standards demanded by global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2'-diaminobiphenyl derivatives has been dominated by palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura coupling, or oxidative coupling strategies. These conventional routes typically require the preparation of specialized precursors like aryl boronic acids or aryl halides, which are not only expensive but also introduce additional synthetic steps prior to the main coupling event. Furthermore, oxidative coupling methods often necessitate the use of stoichiometric oxidants and generate significant amounts of hazardous waste, complicating the disposal process and increasing the overall environmental footprint. The reliance on precious metal catalysts in these traditional methods also poses a risk of metal contamination in the final product, necessitating costly and time-consuming purification steps to meet regulatory limits for residual metals in pharmaceutical and electronic grade materials.
The Novel Approach
In stark contrast, the method disclosed in CN111574380B circumvents these bottlenecks by employing a direct reductive coupling mechanism starting from commodity chemicals. The process begins with the catalytic hydrogenation of 4-substituted nitrobenzenes in the presence of a strong base and a supported noble metal catalyst, efficiently generating the 1,2-diphenylhydrazine intermediate in situ. This intermediate then undergoes a spontaneous rearrangement in an acidic ammonium salt solution to form the desired biphenyl skeleton. This telescoped approach eliminates the need for pre-functionalized coupling partners, drastically reducing the raw material costs and the number of unit operations required. The ability to use simple nitrobenzenes as feedstocks transforms the economic model of producing these valuable diamines, making large-scale production financially viable while maintaining exceptional control over regioselectivity and product purity.
Mechanistic Insights into Noble Metal-Catalyzed Reductive Coupling
The core of this innovative synthesis lies in the selective partial reduction of the nitro group to a hydrazine linkage, a transformation that requires precise control over reaction conditions to prevent over-reduction to anilines. Under the specified conditions of 1.0 to 2.0 MPa hydrogen pressure and temperatures ranging from 45 to 120°C, the noble metal catalyst facilitates the coupling of two nitrobenzene molecules. The presence of a concentrated sodium hydroxide solution is critical, as it promotes the condensation of nitroso and hydroxylamine intermediates on the catalyst surface, steering the reaction pathway towards the formation of the azo or hydrazine bond rather than the fully reduced amine. This mechanistic nuance ensures high conversion rates to the 1,2-diphenylhydrazine species, which serves as the essential precursor for the subsequent skeletal rearrangement.
Following the hydrogenation step, the reaction mixture is subjected to an acidic environment containing hydrochloric acid and ammonium salts, triggering a benzidine-type rearrangement. This acid-catalyzed migration reconstructs the carbon-carbon bond between the two aromatic rings, yielding the 5,5'-disubstituted-2,2'-diaminobiphenyl hydrochloride. The inclusion of ammonium salts in the acidic medium plays a vital role in stabilizing the transition state and suppressing side reactions that could lead to polymeric byproducts or isomeric impurities. Subsequent neutralization and recrystallization from toluene further refine the product, removing any residual catalyst particles or unreacted starting materials. This multi-stage purification protocol ensures that the final API intermediate or polymer monomer achieves a purity level exceeding 99%, suitable for the most demanding applications in advanced materials science.
How to Synthesize 5,5'-Disubstituted-2,2'-diaminobiphenyl Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for manufacturing these complex diamines with high reproducibility. The process is designed to be operationally simple, relying on standard filtration, phase separation, and crystallization techniques that are familiar to plant operators. By optimizing parameters such as the ratio of organic solvent to aqueous base and the specific loading of the noble metal catalyst, manufacturers can fine-tune the reaction kinetics to maximize throughput. The detailed标准化 synthesis steps见下方的指南 ensure that every batch meets consistent quality metrics, minimizing the risk of batch-to-batch variability that often plagues complex organic syntheses.
- Catalytic hydrogenation of 4-substituted nitrobenzene using noble metal catalysts in alkaline organic solvent to form 1,2-diphenylhydrazine.
- Acidic rearrangement of the hydrazine intermediate in hydrochloric acid-ammonium salt mixture to yield the diaminobiphenyl hydrochloride crude.
- Neutralization, solvent extraction, and recrystallization to achieve high-purity final product suitable for industrial applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to this reductive coupling methodology offers substantial opportunities for cost optimization and supply chain stabilization. The primary driver of cost reduction is the substitution of expensive, custom-synthesized coupling partners with bulk commodity nitrobenzenes, which are available from a wide range of global suppliers. This diversification of the raw material base mitigates the risk of supply disruptions and price volatility associated with specialized reagents. Additionally, the process design incorporates multiple recycling loops for both the catalyst and the organic solvents, which significantly lowers the consumption of consumables per kilogram of product. The ability to recover and reuse toluene and ethyl acetate not only reduces direct material costs but also diminishes the volume of hazardous waste requiring treatment, leading to further savings in environmental compliance expenditures.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling reagents and the use of recyclable heterogeneous catalysts fundamentally alter the cost structure of diaminobiphenyl production. By avoiding the purchase of costly aryl halides and boronic acids, the raw material bill is drastically reduced, allowing for more competitive pricing in the final market. Furthermore, the catalyst's longevity means that the amortization of the precious metal cost is spread over a much larger production volume, enhancing the overall margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials like 4-substituted nitrobenzenes ensures a robust and resilient supply chain that is less susceptible to geopolitical or logistical bottlenecks. The simplicity of the reaction conditions, which do not require extreme cryogenic temperatures or ultra-high vacuum systems, allows for production in a broader range of facilities, increasing the total available capacity in the market. This flexibility is crucial for securing long-term supply agreements and ensuring continuity of supply for downstream customers in the pharmaceutical and polymer industries.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in stainless steel autoclaves that are standard in fine chemical manufacturing. The mild reaction conditions and the absence of toxic heavy metal reagents simplify the safety profile of the plant, reducing the need for specialized containment infrastructure. Moreover, the efficient recovery of solvents and the minimization of aqueous waste streams align with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against evolving sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reductive coupling technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary advantages of this reductive coupling method over Suzuki coupling?
A: This method utilizes inexpensive 4-substituted nitrobenzenes as starting materials instead of costly aryl halides and boronic acids required for Suzuki coupling. It significantly reduces step count and eliminates the need for complex pre-functionalization, leading to lower overall production costs and simplified waste management.
Q: Can the noble metal catalyst be recovered and reused in this process?
A: Yes, the patent explicitly states that the solid noble metal catalyst (such as Pt/C or Pd/C) can be filtered off after the hydrogenation step and reused multiple times without significant loss of catalytic activity, which is a critical factor for cost-effective large-scale manufacturing.
Q: What level of purity can be achieved with this synthesis route?
A: Through a combination of ethyl acetate dissolution, toluene recrystallization, and controlled acid precipitation, the process consistently achieves product purity greater than 99%, meeting the stringent specifications required for high-performance polymer and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,5'-Disubstituted-2,2'-diaminobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111574380B for delivering high-value intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of 5,5'-disubstituted-2,2'-diaminobiphenyl meets the exacting standards required for polymer synthesis and pharmaceutical development. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical chemical building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative reductive coupling route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this technology offers compared to your current sourcing strategies. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver superior value through scientific excellence and operational precision.
