Advanced One-Pot Synthesis of 2,4,6-Triphenylpyran Tetrafluoroborate for High-Performance Photoelectric Applications
Advanced One-Pot Synthesis of 2,4,6-Triphenylpyran Tetrafluoroborate for High-Performance Photoelectric Applications
The rapidly evolving landscape of optoelectronics and advanced material science has created an unprecedented demand for high-purity organic precursors that exhibit superior photophysical properties. Among these specialized compounds, pyrylium salts have emerged as critical components due to their unique absorption capabilities in the visible light spectrum and their exceptional stability in excited states. Specifically, 2,4,6-triphenylpyran tetrafluoroborate serves as a vital PET (photo-induced electron transfer) sensitizer, finding extensive applications in laser materials, Q-switches, and high-stability organic luminescent devices. Addressing the historical challenges associated with the low-yield and complex purification of this compound, the Chinese patent CN112979601B, published in January 2023, introduces a groundbreaking one-pot synthesis methodology. This technical breakthrough not only streamlines the manufacturing process but also significantly enhances the economic viability of producing this high-value photoelectric material, positioning it as a cornerstone for next-generation nano-devices and fluorescent sensors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in recent patent literature, the synthesis of 2,4,6-triphenylpyran tetrafluoroborate was predominantly governed by inefficient multi-step protocols that hindered large-scale commercialization. As documented in earlier academic works, such as those by Marcii V. Baldovi et al., the traditional route necessitated the preliminary synthesis of chalcone intermediates through the condensation of acetophenone and benzaldehyde. This intermediate then had to be isolated and subsequently reacted with additional acetophenone in the presence of a Lewis acid to form the final pyrylium salt. This two-step approach suffered from severe drawbacks, most notably a dismal reaction yield ranging merely from 30% to 40%, which drastically inflated the cost of goods sold. Furthermore, the generation of various stubborn byproducts during the intermediate stage made purification exceptionally difficult, often requiring resource-intensive chromatographic techniques that are impractical for ton-scale manufacturing. The cumulative effect of these inefficiencies was a supply chain bottleneck that limited the availability of high-purity material for critical photoelectric applications.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the novel methodology disclosed in patent CN112979601B revolutionizes the production landscape by adopting a direct, one-pot synthetic strategy. This innovative approach bypasses the isolation of the chalcone intermediate entirely, instead facilitating a direct cyclization reaction between benzaldehyde and acetophenone mediated by boron trifluoride diethyl etherate. By carefully controlling the reaction parameters, specifically maintaining an initial temperature of 15-25°C during reagent addition and subsequently heating to 50-80°C, the process achieves a remarkable mass yield of up to 82.1%. This represents a more than twofold improvement in efficiency compared to conventional methods. Moreover, the workup procedure is elegantly simplified; the reaction mixture is poured into stirring ethyl acetate to induce precipitation of the yellow solid, followed by a straightforward filtration and recrystallization step. This elimination of intermediate isolation steps not only reduces solvent consumption but also minimizes the operational time and labor costs associated with the manufacturing process.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this technological advancement lies in the precise manipulation of Lewis acid catalysis to drive the condensation and cyclization reactions simultaneously within a single reactor vessel. The mechanism initiates with the activation of the carbonyl groups in benzaldehyde and acetophenone by the strong Lewis acid, boron trifluoride diethyl etherate. This activation enhances the electrophilicity of the carbonyl carbon, facilitating a nucleophilic attack that leads to the formation of the carbon-carbon bonds necessary for the pyrylium ring structure. The patent data indicates that the reaction temperature plays a pivotal role in this mechanistic pathway; while the initial addition must be kept cool (15-25°C) to manage the exothermic nature of the complexation, the subsequent heating phase (50-80°C) provides the necessary activation energy to overcome the kinetic barriers of ring closure. Experimental data from the patent examples demonstrates a clear correlation between temperature and yield, with yields increasing as the temperature rises to 80°C, suggesting that the cyclization step is thermodynamically favored at elevated temperatures within this specific range.
Controlling the impurity profile is equally critical in the production of photoelectric materials, where trace contaminants can quench fluorescence or alter absorption spectra. The novel process addresses this through a robust purification protocol involving recrystallization from a mixed solvent system of acetone and water. This specific solvent combination exploits the differential solubility of the target pyrylium salt versus potential organic byproducts, ensuring that the final product meets stringent purity specifications required for sensitive optical applications. The structural integrity of the resulting cation, stabilized by the non-coordinating tetrafluoroborate anion, is confirmed through spectroscopic analysis, revealing the characteristic symmetric arrangement of the phenyl groups which is essential for its electronic properties.
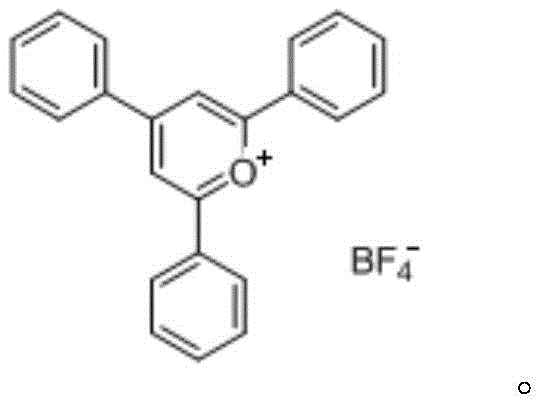
How to Synthesize 2,4,6-Triphenylpyran Tetrafluoroborate Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding stoichiometry and thermal management to ensure reproducibility and safety. The process begins with the precise weighing of benzaldehyde and acetophenone at a molar ratio of 1:3, which is crucial for driving the equilibrium towards the desired triphenyl-substituted product while minimizing side reactions. The addition of the catalyst must be performed dropwise over a period of approximately 1.5 hours to prevent localized overheating, which could lead to polymerization or degradation of the sensitive intermediates. Following the reaction period of 15 hours, the quenching step involves pumping the reaction liquid into a large volume of ethyl acetate, a technique that ensures rapid and uniform precipitation of the product. For a comprehensive, step-by-step guide including exact quantities and safety precautions, please refer to the standardized synthesis protocol provided below.
- Mix benzaldehyde and acetophenone at a molar ratio of 1: 3 and control the temperature between 15-25°C.
- Dropwise add boron trifluoride diethyl etherate over 1.5 hours, then heat the system to 50-80°C for 15 hours.
- Pour the reaction liquid into stirred ethyl acetate at 40°C to precipitate the solid, followed by filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the traditional two-step synthesis to this novel one-pot method offers profound strategic advantages that extend far beyond simple yield improvements. The consolidation of the synthesis into a single reactor run fundamentally alters the cost structure of the material by eliminating the need for intermediate storage, handling, and quality control testing of the chalcone precursor. This reduction in unit operations translates directly into lower overhead costs and a smaller physical footprint required for production, making it an ideal candidate for facilities looking to optimize their existing infrastructure without significant capital expenditure on new equipment. Furthermore, the use of commodity chemicals like benzaldehyde and acetophenone as starting materials ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or exotic reagents that are prone to market volatility.
- Cost Reduction in Manufacturing: The most significant economic driver of this new process is the drastic improvement in mass yield, which climbs from a historical average of roughly 35% to over 82% under optimized conditions. This near-doubling of efficiency means that for every kilogram of raw material purchased, the output of saleable product is significantly higher, effectively halving the raw material cost per unit of finished good. Additionally, the simplification of the purification process removes the need for expensive chromatographic resins or extensive solvent exchanges, further reducing the variable costs associated with consumables and waste disposal. The ability to achieve high purity through simple recrystallization also lowers the energy intensity of the drying and finishing stages, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: By relying on a one-pot synthesis, the lead time for production batches is substantially shortened, as the time-consuming isolation and characterization of intermediates are completely eradicated. This agility allows manufacturers to respond more rapidly to fluctuations in market demand, ensuring that critical photoelectric materials are available when needed for downstream device assembly. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures (50-80°C) without significant loss in yield, adds a layer of operational reliability, reducing the likelihood of batch failures due to minor process deviations. Consequently, this stability fosters a more predictable supply schedule, which is essential for long-term planning in the electronics and optical materials sectors.
- Scalability and Environmental Compliance: The process design is inherently scalable, utilizing standard unit operations such as mechanical stirring, dropwise addition, and filtration that are easily transferred from laboratory glassware to industrial-scale reactors. The avoidance of complex separation techniques simplifies the engineering requirements for scale-up, reducing the technical risk associated with expanding production capacity from kilograms to tons. From an environmental perspective, the higher atom economy and reduced solvent usage align with increasingly stringent global regulations on chemical manufacturing emissions. The ability to recycle the ethyl acetate used in the precipitation step further enhances the sustainability profile of the process, appealing to environmentally conscious stakeholders and helping companies meet their corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2,4,6-triphenylpyran tetrafluoroborate, based on the detailed disclosures within the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this material into new product lines or for procurement specialists assessing supplier capabilities. The answers provided reflect the specific advantages of the one-pot methodology over legacy processes, focusing on yield, purity, and operational simplicity.
Q: What is the primary advantage of the one-pot method described in CN112979601B?
A: The primary advantage is the significant increase in yield (up to 82.1%) compared to conventional two-step methods (30-40%), alongside a simplified purification process that eliminates the need for isolating unstable chalcone intermediates.
Q: What are the critical reaction conditions for maximizing purity?
A: Critical conditions include maintaining the initial addition temperature between 15-25°C to control exothermicity, reacting at 60-80°C for optimal conversion, and utilizing a specific acetone-water recrystallization system to remove impurities.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is highly scalable as it utilizes readily available raw materials like benzaldehyde and acetophenone, avoids complex chromatographic purification, and relies on simple precipitation and filtration unit operations suitable for large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-Triphenylpyran Tetrafluoroborate Supplier
As the global demand for advanced photoelectric materials continues to surge, partnering with a manufacturer that possesses both the technical expertise and the production capacity to deliver high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep chemical engineering knowledge to translate complex laboratory patents into robust commercial processes. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of handling diverse synthetic pathways, ensuring that we can scale production seamlessly from 100 kgs to 100 MT/annual commercial production volumes. We understand that in the field of optoelectronics, consistency is key; therefore, we adhere to stringent purity specifications to guarantee that every batch of 2,4,6-triphenylpyran tetrafluoroborate meets the exacting standards required for high-performance laser and sensor applications.
We invite international partners to collaborate with us to unlock the full potential of this advanced material for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized synthesis route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our supply chain solutions can support your long-term growth in the competitive photoelectric materials market.
