Advanced Synthesis of 19-Hydroxyandrostenedione Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for complex steroid intermediates, particularly those serving as precursors for contraceptives and hormonal therapies. Patent CN110172078B introduces a groundbreaking preparation method for 19-hydroxycortodoxone derivatives and the critical intermediate 19-hydroxyandrostenedione. This technology addresses the longstanding challenge of modifying the sterically hindered C19 position on the steroid skeleton, a transformation that is notoriously difficult due to the inertness of the C-H bond in that region. By utilizing 19-hydroxycortodoxone as a versatile starting material, the disclosed method enables precise structural modifications on the A and B rings, selective substitution of hydroxyl groups at positions 17, 21, and 19, and efficient excision of the side chain. This represents a significant leap forward in steroid chemistry, offering a reliable pathway to high-purity intermediates that are essential for the synthesis of norethisterone-class contraceptives.
The strategic value of this patent lies in its ability to bypass the limitations of indirect hydroxylation methods. Traditionally, introducing a hydroxyl group at the C19 position required cumbersome multi-step sequences involving specialized functional groups or harsh photochemical conditions. The approach detailed in CN110172078B simplifies this landscape by starting with a biologically or chemically pre-hydroxylated scaffold and focusing on downstream diversification. This shift not only streamlines the synthetic route but also enhances the overall atom economy of the process. For R&D teams evaluating new entry points into the steroid market, this methodology provides a flexible platform for generating diverse analogues with potential biological activity, thereby accelerating drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C19 methyl group in steroids has been a bottleneck in organic synthesis. Conventional strategies such as the Suárez reaction, Norrish-Type II photoreactions, and Barton nitrite photoreactions have dominated the literature, yet they suffer from significant drawbacks that hinder industrial adoption. These methods often necessitate the pre-synthesis of specific, unstable functional groups, adding unnecessary steps and cost to the overall process. Furthermore, photochemical reactions typically require specialized equipment and suffer from poor scalability due to light penetration issues in large reactors. The harsh conditions associated with these traditional routes frequently lead to low yields and complex impurity profiles, complicating downstream purification and increasing the environmental footprint of the manufacturing process. Consequently, there has been an urgent demand for a more direct, efficient, and scalable transformation method.
The Novel Approach
The methodology presented in patent CN110172078B offers a compelling alternative by leveraging 19-hydroxycortodoxone as a robust substrate for diversification. Instead of struggling to install the C19 hydroxyl group de novo, this approach utilizes it as a handle for further chemical manipulation. The patent describes a suite of reactions including epoxidation of the A-ring using hydrogen peroxide and sodium hydroxide, hydrogenation of the double bond using palladium on carbon, and oxidative cleavage of the side chain using sodium periodate. 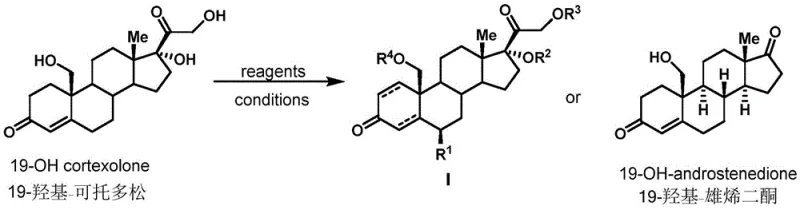 This modular strategy allows chemists to access a wide array of derivatives, designated as Formula I, by simply varying the reagents and conditions. The ability to perform these transformations under relatively mild conditions, such as 0°C to room temperature, drastically reduces energy consumption and safety risks compared to high-temperature or high-pressure alternatives. This novel approach effectively decouples the difficulty of C19 functionalization from the rest of the synthesis, enabling a more focused optimization of the final drug substance.
This modular strategy allows chemists to access a wide array of derivatives, designated as Formula I, by simply varying the reagents and conditions. The ability to perform these transformations under relatively mild conditions, such as 0°C to room temperature, drastically reduces energy consumption and safety risks compared to high-temperature or high-pressure alternatives. This novel approach effectively decouples the difficulty of C19 functionalization from the rest of the synthesis, enabling a more focused optimization of the final drug substance.
Mechanistic Insights into Oxidative Side-Chain Cleavage and Ring Modification
A deeper mechanistic understanding of the transformations described in the patent reveals why this process is so effective for industrial application. The oxidative cleavage of the side chain to generate 19-hydroxyandrostenedione is particularly noteworthy. This transformation proceeds via a two-step sequence involving initial reduction of the C20 ketone with sodium borohydride, followed by oxidative cleavage with sodium periodate. The mechanism likely involves the formation of a glycol intermediate upon reduction, which is then susceptible to periodate oxidation, resulting in the clean scission of the C17-C20 bond. This avoids the use of hazardous ozonolysis or heavy metal oxidants that are often employed in similar degradations. The high yield of 84% reported for this specific transformation underscores the efficiency of the redox manifold employed, minimizing waste generation and maximizing reactor throughput.
In addition to side-chain cleavage, the patent details sophisticated modifications of the steroid core that are critical for tuning biological activity. For instance, the introduction of an epoxide on the A-ring using alkaline hydrogen peroxide demonstrates a high degree of regioselectivity, preserving the sensitive C19 hydroxyl group while modifying the enone system. Similarly, the use of trimethyliodosilane (TMSI) for deoxygenation or protection strategies highlights the versatility of silyl chemistry in managing the multiple hydroxyl functionalities present on the molecule. These mechanistic pathways are designed to be orthogonal, meaning that one functional group can be modified without affecting others, a crucial feature for the synthesis of complex pharmaceutical intermediates. The control over stereochemistry, evidenced by the specific beta/alpha ratios reported in certain examples, further ensures that the resulting intermediates possess the correct spatial configuration for subsequent biological interactions.
How to Synthesize 19-Hydroxyandrostenedione Efficiently
The synthesis of 19-hydroxyandrostenedione from 19-hydroxycortodoxone is a prime example of how modern organic synthesis can optimize legacy pathways. The process begins with the dissolution of the starting material in a mixed solvent system of dichloromethane and ethanol, providing a homogeneous medium for the reaction. The addition of sodium borohydride at controlled low temperatures ensures selective reduction without over-reduction of other sensitive moieties. Following the quench with acetone, the oxidative cleavage step is initiated by the addition of sodium periodate, which acts as a powerful yet selective oxidant. 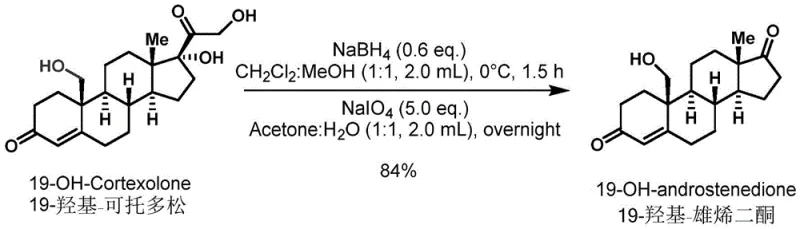 This sequence is operationally simple, requiring no exotic catalysts or extreme pressures, making it highly amenable to transfer from the laboratory to the pilot plant. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the synthesis guide below.
This sequence is operationally simple, requiring no exotic catalysts or extreme pressures, making it highly amenable to transfer from the laboratory to the pilot plant. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the synthesis guide below.
- Dissolve 19-hydroxycortodoxone in a dichloromethane and ethanol solvent mixture and react with sodium borohydride at 0°C to reduce the ketone functionality.
- Quench the reduction with acetone, then introduce sodium periodate and water to the reaction mixture at room temperature to effect oxidative side-chain cleavage.
- Filter the reaction mixture through celite, concentrate the filtrate under reduced pressure, and purify the crude 19-hydroxyandrostenedione via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the technology disclosed in CN110172078B offers substantial strategic advantages over conventional steroid synthesis routes. The primary benefit stems from the simplification of the synthetic sequence, which directly translates to reduced manufacturing costs and improved supply reliability. By eliminating the need for complex photochemical reactors or hazardous reagents like ozone, the capital expenditure required for setting up production lines is significantly lowered. Furthermore, the use of commodity chemicals such as hydrogen peroxide, sodium hydroxide, and palladium on carbon ensures that raw material sourcing is stable and不受 geopolitical fluctuations that often affect specialty reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis pathway leads to significant cost savings by reducing the total number of unit operations required. Traditional methods often involve lengthy protection and deprotection sequences that consume additional reagents and solvents, driving up the cost of goods sold. In contrast, the direct modification strategies outlined in the patent minimize these auxiliary steps. The high yields achieved, particularly in the side-chain cleavage step, mean that less starting material is wasted, improving the overall mass balance of the process. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and reduced utility costs. These factors combined create a highly competitive cost structure for the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable reagents. Unlike processes that depend on custom-synthesized catalysts or light-sensitive materials that degrade during storage, the reagents specified in this patent have long shelf lives and are sourced from multiple global suppliers. This redundancy mitigates the risk of supply disruptions caused by vendor-specific issues. Moreover, the robustness of the reaction conditions allows for greater flexibility in manufacturing locations, enabling companies to diversify their production footprint. The ability to scale these reactions from gram to kilogram quantities without significant re-optimization ensures that supply can be ramped up quickly to meet surges in market demand, securing the supply chain against volatility.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved compared to older technologies, facilitating easier regulatory compliance and permitting. The avoidance of heavy metal oxidants and the use of aqueous workups where possible simplify waste treatment protocols. The high selectivity of the reactions reduces the formation of by-products, leading to cleaner crude products that require less solvent-intensive purification. This reduction in solvent usage not only lowers costs but also aligns with green chemistry principles increasingly mandated by environmental regulations. The process is inherently scalable, as demonstrated by the successful execution of reactions in standard glassware without the need for specialized flow chemistry equipment, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN110172078B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D directors and procurement managers evaluating the feasibility of adopting this route for their specific product portfolios. The answers reflect the practical realities of scaling these chemical transformations in a GMP environment.
Q: What is the primary advantage of the side-chain cleavage method described in CN110172078B?
A: The method utilizes a sodium borohydride reduction followed by sodium periodate oxidation, achieving yields up to 84% under mild conditions, which significantly outperforms traditional harsh oxidative methods.
Q: Can this process be adapted for producing other 19-hydroxy steroid derivatives?
A: Yes, the patent outlines versatile modifications including A-ring epoxidation, B-ring hydrogenation, and hydroxyl protection, allowing for the synthesis of a diverse library of steroid intermediates.
Q: Are the reagents used in this synthesis commercially scalable?
A: Absolutely. The process relies on common industrial reagents such as hydrogen peroxide, palladium on carbon, and sodium periodate, ensuring easy sourcing and straightforward waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 19-Hydroxyandrostenedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert chemists has extensively analyzed the methodologies described in CN110172078B and possesses the technical capability to implement these advanced synthetic routes on a commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 19-hydroxyandrostenedione or its derivatives meets the highest industry standards. We are committed to being a partner that adds value through technical expertise and operational excellence.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will de-risk your supply chain. Contact us today to discuss how we can support your goals with reliable, cost-effective, and high-purity steroid intermediates, ensuring your projects move forward without delay.
