Optimizing N-Methyl-2-Fluoroaniline Production: A Novel Diazotization Route for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity, better selectivity, and more sustainable processes. A significant advancement in this domain is detailed in patent CN109053486B, which discloses a novel synthesis method for N-methyl-2-fluoroaniline. This compound serves as a critical building block in the development of various active pharmaceutical ingredients (APIs) and agrochemicals, where the precise introduction of a methyl group onto an aniline nitrogen is often required. The patented methodology addresses longstanding challenges associated with traditional methylation techniques, offering a robust pathway that leverages diazotization chemistry to achieve exceptional selectivity. By shifting away from equilibrium-limited reductive aminations, this approach provides a reliable solution for producing high-purity secondary amines, positioning it as a valuable asset for any reliable pharma intermediate supplier looking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the monomethylation of primary amines has been fraught with selectivity issues, primarily due to the inherent reactivity of the resulting secondary amine product. The classical Eschweiler-Clark reaction, which utilizes formaldehyde and formic acid, is a widely known method for N-methylation. However, as illustrated in the reaction scheme below, this process suffers from a lack of control at the mono-substitution stage. Once the primary amine is converted to a secondary amine, the product remains nucleophilic and can readily undergo a second methylation cycle to form unwanted tertiary amine byproducts.
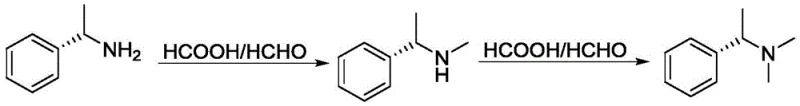
This tendency towards over-alkylation necessitates rigorous purification steps to separate the desired secondary amine from tertiary impurities, which significantly drives up processing costs and reduces overall yield. Furthermore, alternative methods using strong methylating agents like dimethyl sulfate often require specific additives, such as zeolite molecular sieves, to improve selectivity, adding complexity and material costs to the operation. Without these specialized catalysts, the reaction mixture typically yields a poor ratio of the target monomethylated product, rendering the process inefficient for large-scale cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these equilibrium-driven methods, the invention disclosed in CN109053486B introduces a fundamentally different mechanistic pathway that bypasses the issue of over-methylation entirely. By employing a diazotization strategy followed by coupling with methylamine, the process ensures that the nitrogen atom is functionalized in a single, controlled step. This route starts with 2-fluoroaniline, a commercially abundant feedstock, and converts it into a reactive diazonium salt intermediate under acidic conditions. This intermediate is then reacted with an aqueous methylamine solution, effectively transferring the methyl group without the risk of further alkylation. The result is a streamlined synthesis that delivers high yields—reported up to 95% in optimized examples—with minimal formation of tertiary amine impurities, thereby simplifying downstream purification and enhancing overall process economics.
Mechanistic Insights into Diazotization-Methylamination Coupling
The core of this technological breakthrough lies in the precise manipulation of reaction conditions to stabilize the highly reactive diazonium species before coupling. The first stage involves the treatment of 2-fluoroaniline with concentrated hydrochloric acid and sodium nitrite. Crucially, this diazotization step must be conducted at low temperatures, specifically maintained between -15°C and -5°C, to prevent the premature decomposition of the diazonium salt. The patent data indicates that maintaining the internal temperature below 5°C during the dropwise addition of sodium nitrite is essential for maximizing the stability of the intermediate. Deviations from this thermal window, such as allowing the temperature to rise to 15°C, can lead to a dramatic collapse in yield, dropping from over 90% to as low as 45%, highlighting the kinetic sensitivity of this transformation.
Following the formation of the diazonium salt, the second stage involves a nucleophilic substitution where the diazo group is displaced by methylamine. This step is performed at elevated temperatures, optimally between 50°C and 55°C, which provides the necessary activation energy for the coupling reaction to proceed efficiently. The stoichiometry is also carefully balanced, with a molar ratio of 2-fluoroaniline to methylamine ranging from 1:2.5 to 1:3, ensuring that the amine is in excess to drive the reaction to completion. As shown in the reaction pathway below, this two-step sequence effectively installs the methyl group while preserving the integrity of the aromatic ring and the fluorine substituent, which is critical for the biological activity of downstream drug candidates.
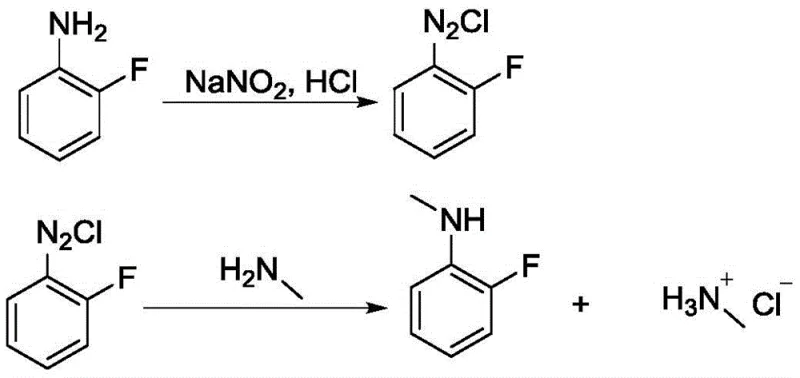
How to Synthesize N-Methyl-2-Fluoroaniline Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the diazonium salt in an ice-salt bath, followed by the controlled addition of this solution into a pre-heated methylamine reactor. The workup procedure is straightforward, involving extraction with ethyl acetate, washing with saturated brine to remove inorganic salts, and final distillation to isolate the pure product. For R&D teams looking to replicate or scale this chemistry, the following standardized protocol outlines the critical operational steps derived directly from the patent examples.
- Prepare 2-fluoroaniline diazonium salt by reacting 2-fluoroaniline with concentrated hydrochloric acid and sodium nitrite at temperatures between -15°C and -5°C.
- Add the diazonium salt solution dropwise into a heated methylamine aqueous solution maintained at 50°C to 55°C.
- Perform post-treatment via ethyl acetate extraction, brine washing, drying, and solvent distillation to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling advantages that directly address the pain points of sourcing complex organic intermediates. The shift from catalytic hydrogenation or complex reductive amination to a diazotization-based approach eliminates the dependency on expensive transition metal catalysts, such as palladium or platinum, which are not only costly but also subject to volatile market pricing and supply constraints. By removing the need for these precious metals, manufacturers can significantly reduce raw material expenditures and avoid the regulatory and environmental burdens associated with heavy metal residue testing and removal in the final API.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the utilization of commodity chemicals. 2-fluoroaniline, hydrochloric acid, sodium nitrite, and methylamine are all bulk industrial chemicals with stable supply chains and low unit costs. Unlike methods requiring specialized reagents or high-pressure hydrogenation equipment, this process operates at atmospheric pressure and moderate temperatures, reducing capital expenditure (CAPEX) on reactor infrastructure. The high selectivity of the reaction minimizes waste generation and reduces the solvent load required for purification, leading to substantial operational expenditure (OPEX) savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for global pharmaceutical supply chains. Because this method relies on widely available, non-proprietary raw materials, it mitigates the risk of bottlenecks associated with niche reagents. The robustness of the chemistry, demonstrated by consistent yields across multiple experimental groups in the patent, ensures that production schedules can be met reliably. Furthermore, the avoidance of sensitive catalysts means that the process is less susceptible to disruptions caused by catalyst deactivation or supply shortages, providing a more resilient manufacturing backbone for long-term contracts.
- Scalability and Environmental Compliance: The simplicity of the workup procedure—extraction and distillation—is inherently scalable from pilot plant to multi-ton production. The process generates manageable waste streams primarily consisting of saline water and spent solvents, which are easier to treat compared to the heavy metal waste from hydrogenation routes. This aligns well with modern green chemistry principles and stringent environmental regulations, facilitating smoother permitting and compliance audits. The ability to scale this process from 100 kgs to 100 MT annual commercial production without significant re-engineering makes it an ideal candidate for meeting growing market demand.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common inquiries regarding the reaction mechanism, safety considerations, and quality attributes of the final product. These insights are derived from the detailed experimental data provided in the patent documentation, offering a transparent view of the process capabilities and limitations for potential partners and technical evaluators.
Q: Why is the diazotization method preferred over Eschweiler-Clark methylation for this compound?
A: The Eschweiler-Clark reaction often leads to over-methylation, producing tertiary amines as impurities. The diazotization route described in patent CN109053486B offers superior selectivity for the secondary amine stage, ensuring higher purity without complex separation steps.
Q: What are the critical temperature controls for this synthesis?
A: Temperature control is vital. The diazotization step must be kept below 5°C (ideally -10°C to -5°C) to stabilize the diazonium salt. The subsequent coupling with methylamine requires heating to 50-55°C to drive the reaction to completion efficiently.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, readily available raw materials like 2-fluoroaniline and methylamine. It avoids expensive metal catalysts and achieves yields up to 95%, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-2-Fluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates like N-methyl-2-fluoroaniline in the development of next-generation therapeutics. Our technical team has extensively analyzed the diazotization pathway described in CN109053486B and possesses the expertise to implement this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, including tertiary amines and residual diazonium species, guaranteeing the integrity of your supply chain.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can support your commercial goals and accelerate your time to market.
