Advanced Green Synthesis of Cefuroxime Acid Intermediate DCC for Global Pharma Supply Chains
The pharmaceutical industry is constantly evolving towards greener, more efficient manufacturing processes, particularly for critical antibiotic intermediates like those used in cephalosporin production. A pivotal advancement in this domain is detailed in patent CN109988183B, which outlines an environment-friendly preparation method for a cefuroxime acid intermediate. This technology addresses long-standing challenges associated with traditional synthesis routes, specifically targeting the elimination of hazardous solvents and the reduction of inorganic waste. By shifting from phosphorus pentachloride to a triphosgene-based activation system and completely abandoning dimethylacetamide (DMAC), this process offers a robust pathway for producing high-purity DCC (the key intermediate). For global supply chain leaders, this represents a significant opportunity to enhance the sustainability profile of their antibiotic portfolios while securing a more stable and cost-effective source of raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of cefuroxime acid intermediates has relied heavily on phosphorus pentachloride (PCl5) as a chlorinating agent and dimethylacetamide (DMAC) as a solvent. While effective in driving the reaction, this conventional approach suffers from severe environmental and operational drawbacks that impact the bottom line. The use of PCl5 generates substantial amounts of chloride salts and phosphates in the wastewater, creating a heavy burden on effluent treatment facilities and increasing compliance costs. Furthermore, DMAC is a high-boiling solvent with a boiling point of 161°C, making its recovery energy-intensive and economically inefficient. As a Class 2 solvent under ICH guidelines, DMAC also poses toxicity risks, necessitating strict control limits to prevent residual contamination in the final active pharmaceutical ingredient (API). These factors combine to create a process that is not only environmentally taxing but also prone to yield fluctuations due to the difficulty in completely removing solvent residues.
The Novel Approach
The innovative methodology described in the patent data introduces a paradigm shift by utilizing triphosgene as a safer and more efficient alternative to phosphorus pentachloride. This new route operates under milder conditions and employs common organic solvents that are easier to manage and recover. Crucially, the process completely eliminates the use of DMAC, thereby removing the associated toxicity risks and solvent recovery bottlenecks. The reaction utilizes a catalytic amount of imidazole to facilitate the depolymerization of triphosgene, ensuring a thorough conversion to the reactive acyl chloride species. This results in a cleaner reaction profile with fewer impurities introduced into the system. By optimizing the condensation step between the activated side chain and the beta-lactam nucleus, the novel approach achieves superior product quality with consistently low moisture content, directly addressing the purity concerns that plague older manufacturing techniques.
Mechanistic Insights into Triphosgene-Mediated Acylation
The core of this green synthesis lies in the precise activation of the methoxyimino furan acetic acid side chain. The process begins with the depolymerization of triphosgene in an organic solvent, catalyzed by imidazole at low temperatures ranging from -40°C to 20°C. This step generates the reactive phosgene equivalents in situ, which immediately react with the methoxyimino furan ammonium acetate (SMIA ammonium salt) to form the corresponding acyl chloride. The use of imidazole is critical here, as it acts as a nucleophilic catalyst that accelerates the formation of the acyl chloride while minimizing side reactions. The reaction progress is tightly monitored via HPLC, ensuring that the residual starting material is reduced to less than 0.5% before proceeding. This rigorous control prevents the carryover of unreacted acids into the subsequent condensation step, which is vital for maintaining high stereochemical integrity and overall yield.
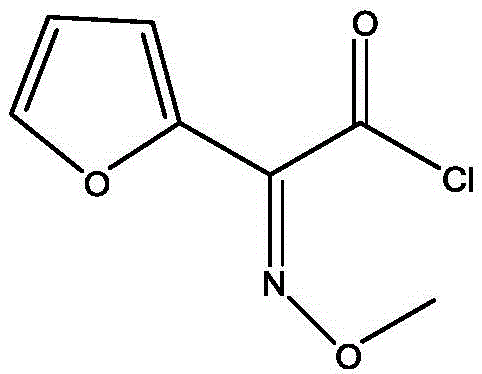
Following the formation of the SMIA acyl chloride, the process moves to the condensation phase with D-7-ACA (hydroxymethyl-7-aminocephalosporanic acid). The D-7-ACA is dissolved in water with pH adjustment to ensure solubility and stability of the beta-lactam ring. The acyl chloride solution is then added dropwise, and the pH is carefully maintained between 5.0 and 9.0 to facilitate the nucleophilic attack of the amine on the carbonyl carbon. This aqueous workup strategy is a significant departure from organic-heavy traditional methods, simplifying the isolation of the product. The resulting DCC intermediate precipitates upon acidification to pH 1.0-3.0, allowing for easy filtration and drying. The structural integrity of the final DCC molecule is preserved throughout this gentle aqueous processing, ensuring that the sensitive beta-lactam core remains intact for the final conversion to cefuroxime acid.
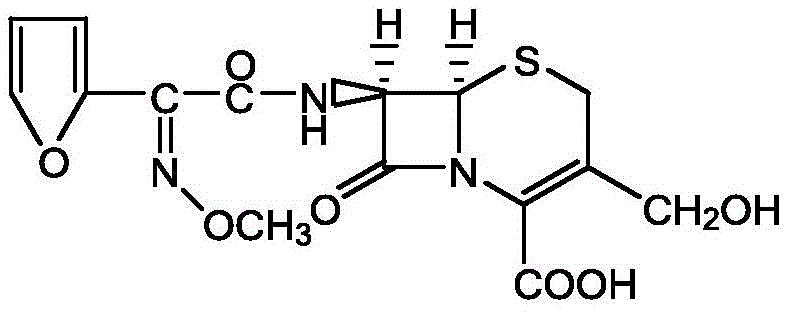
How to Synthesize Cefuroxime Acid Intermediate DCC Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometry to maximize the benefits of the triphosgene system. The process is designed to be scalable, moving seamlessly from laboratory optimization to industrial production without the need for specialized high-pressure equipment. Operators must ensure that the triphosgene concentration is maintained within the optimal range of 0.04 g/ml to 0.50 g/ml to balance reaction rate with safety. The subsequent crystallization step is equally critical, where controlling the cooling rate and stirring time ensures the formation of uniform crystals with low moisture content. For a detailed breakdown of the specific operational parameters and safety protocols required for this synthesis, please refer to the standardized guide below.
- Prepare SMIA acyl chloride solution by reacting triphosgene with imidazole and SMIA ammonium salt at low temperature (-40 to 20°C).
- Dissolve D-7-ACA in water with alkali solution, adjusting pH to 7.0-10.0 and cooling to -10 to 15°C.
- Condense the SMIA acyl chloride solution with the D-7-ACA solution, adjust pH to 5.0-9.0, then acidify to pH 1.0-3.0 for crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic advantages beyond mere regulatory compliance. The elimination of DMAC removes a major bottleneck in solvent recovery, significantly reducing energy consumption and utility costs associated with distillation. Furthermore, the reduction in inorganic waste (chlorides and phosphates) lowers the expense of wastewater treatment, contributing to a leaner manufacturing cost structure. The improved purity profile of the intermediate also reduces the risk of batch rejection during downstream API synthesis, enhancing overall supply reliability. By securing a supply of intermediates produced via this greener route, companies can future-proof their operations against tightening environmental regulations while achieving substantial cost savings through process efficiency.
- Cost Reduction in Manufacturing: The substitution of phosphorus pentachloride with triphosgene and the removal of DMAC drastically simplify the post-reaction workup. Traditional methods require extensive washing and energy-intensive distillation to remove high-boiling solvents, whereas this new method allows for easier phase separation and crystallization. This simplification reduces the consumption of utilities and labor hours per batch, leading to a more economical production process without compromising on the quality of the cefuroxime acid intermediate.
- Enhanced Supply Chain Reliability: The robustness of the triphosgene-mediated reaction ensures consistent yields and high purity, typically exceeding 98%. High-purity intermediates reduce the variability in the final API synthesis, minimizing the need for reprocessing or discarding off-spec batches. This consistency is crucial for maintaining uninterrupted supply lines for essential antibiotics, ensuring that pharmaceutical manufacturers can meet market demand without the disruption caused by quality failures or extended purification cycles.
- Scalability and Environmental Compliance: This process is inherently designed for scale-up, utilizing standard reactor configurations and avoiding hazardous reagents that require special handling infrastructure. The significant reduction in toxic solvent usage and inorganic waste generation aligns perfectly with modern ESG (Environmental, Social, and Governance) goals. Manufacturers adopting this technology can demonstrate a commitment to sustainable chemistry, potentially qualifying for green incentives and improving their corporate standing with environmentally conscious stakeholders and regulators.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the safety, efficiency, and quality implications of switching to this triphosgene-based method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is replacing DMAC significant in cefuroxime intermediate production?
A: DMAC is a high-boiling point solvent (161°C) that is difficult to recover and classified as a toxic Class 2 solvent by ICH. Eliminating it reduces solvent residue in the final API and lowers wastewater treatment costs significantly.
Q: How does the triphosgene method improve product purity compared to phosphorus pentachloride?
A: The triphosgene method avoids the introduction of high concentrations of chloride and phosphate salts found in wastewater from phosphorus pentachloride processes, leading to higher purity DCC with moisture content below 2%.
Q: What are the critical pH control points in this synthesis?
A: Critical pH controls include maintaining pH 7.0-10.0 for dissolving D-7-ACA, pH 5.0-9.0 during the condensation reaction, and adjusting to pH 1.0-3.0 with acid to induce crystallization of the final DCC product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, eco-friendly synthesis routes to meet the evolving demands of the global pharmaceutical market. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can deliver the cefuroxime acid intermediate DCC with the highest standards of consistency and purity. We leverage our rigorous QC labs and stringent purity specifications to guarantee that every batch meets the exacting requirements necessary for downstream API manufacturing. Our commitment to green chemistry aligns with the principles outlined in patent CN109988183B, allowing us to offer a product that is not only high-performing but also environmentally responsible.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to our DMAC-free intermediate. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of quality, sustainability, and reliability.
