Scalable Synthesis of Pyrano Thiazolo Pyrimidine Derivatives Using Recyclable Basic Ionic Liquids
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as critical intermediates for antiviral and anticancer agents. A significant breakthrough in this domain is detailed in patent CN114478580A, which discloses a novel synthetic method for producing pyrano[2,3-d][1,3]thiazolo[3,2-a]pyrimidine derivatives. This technology represents a paradigm shift from traditional multi-step, harsh condition syntheses to a streamlined, one-pot multicomponent reaction facilitated by a unique basic ionic liquid catalyst system. By leveraging a mixed solvent system comprising ethanol and 1-butyl-3-methylimidazolium bromide ([Bmim]Br), this method achieves exceptional reaction rates at mild temperatures ranging from 35-50°C. For R&D directors and process chemists, this innovation offers a robust route to high-purity intermediates with minimized environmental footprint, addressing the growing demand for sustainable manufacturing practices in the production of bioactive nitrogen and sulfur heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrano[2,3-d][1,3]thiazolo[3,2-a]pyrimidine derivatives has been plagued by significant inefficiencies that hinder large-scale industrial adoption. Conventional routes often rely on multi-step sequences involving harsh reaction conditions, such as high-temperature reflux in organic solvents like ethanol with stoichiometric amounts of organic bases like N,N-diisopropylethylamine. These traditional methods suffer from poor atom economy and generate substantial volumes of hazardous waste liquid, as the organic base catalysts cannot be effectively recovered or recycled. Furthermore, the lack of selectivity in these older processes frequently leads to the formation of difficult-to-remove by-products, complicating downstream purification and reducing the overall yield of the target pharmaceutical intermediate. The inability to recycle the catalyst and solvent system not only drives up raw material costs but also creates severe bottlenecks in continuous production capabilities, forcing manufacturers to rely on inefficient batch processing that struggles to meet modern supply chain demands.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a highly efficient one-pot three-component condensation reaction that dramatically simplifies the synthetic landscape. By utilizing a specialized basic ionic liquid as both a catalyst and a co-solvent component, the reaction proceeds rapidly within a narrow temperature window of 35-50°C, completing in as little as 4-8 minutes. This approach eliminates the need for toxic volatile organic compounds and non-recyclable organic bases, replacing them with a tunable ionic liquid system that enhances reaction kinetics through superior solubility and activation of reactants. The result is a process that not only delivers high yields exceeding 90% but also ensures product purities consistently above 98%, as verified by HPLC analysis. The simplicity of the work-up procedure, which involves mere filtration and washing, underscores the practical superiority of this ionic liquid-mediated pathway over legacy technologies.

Mechanistic Insights into Basic Ionic Liquid-Catalyzed Cyclization
The efficacy of this synthesis lies in the dual functionality of the basic ionic liquid catalyst, which acts as a potent base to activate the methylene group of malononitrile while simultaneously stabilizing transition states through hydrogen bonding interactions. The specific structure of the catalyst, featuring long alkyl chains and hydroxyl-functionalized morpholinium rings, facilitates a micro-environment that promotes the nucleophilic attack of the active methylene species onto the aldehyde carbonyl group. This initial Knoevenagel condensation is followed by a Michael addition and subsequent intramolecular cyclization with the 7-hydroxy-2,3-dihydro-5H-[1,3]thiazolo[3,2-a]pyrimidin-5-one. The ionic nature of the solvent system ensures that all polar intermediates remain in solution, preventing premature precipitation that could halt the reaction cascade. This mechanistic precision minimizes side reactions and polymerization, which are common pitfalls in conventional base-catalyzed condensations, thereby ensuring a clean reaction profile suitable for GMP manufacturing environments.
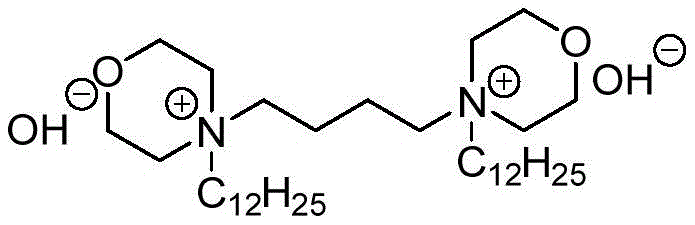
Impurity control is inherently built into this catalytic system due to the high selectivity of the ionic liquid medium. Unlike heterogeneous catalysts that may suffer from leaching or inconsistent active site availability, the homogeneous nature of the ionic liquid ensures uniform catalytic activity throughout the reaction volume. This uniformity prevents localized hot spots of high basicity that often lead to the degradation of sensitive functional groups on the aromatic aldehyde substrates. Furthermore, the ability to fine-tune the solvent polarity by adjusting the ratio of ethanol to [Bmim]Br allows process chemists to optimize the solubility of the final product, facilitating its spontaneous crystallization upon cooling. This "crystallization-driven" purification mechanism effectively excludes soluble impurities from the crystal lattice, resulting in a crude product of such high quality that extensive recrystallization steps are rendered unnecessary, streamlining the overall production timeline.
How to Synthesize Pyrano Thiazolo Pyrimidine Derivatives Efficiently
The operational protocol for this synthesis is designed for maximum simplicity and scalability, requiring standard laboratory equipment such as a three-necked flask equipped with a condenser and magnetic stirrer. The process begins by preparing a mixed solvent system of ethanol and [Bmim]Br, into which the three key reactants—aldehyde, malononitrile, and the thiazolo-pyrimidinone precursor—are introduced along with a catalytic amount of the basic ionic liquid. The reaction mixture is then gently heated to the optimal temperature range, where it is maintained for a brief period to ensure complete conversion. Upon completion, the reaction is simply allowed to cool, triggering the precipitation of the target compound, which is then isolated via filtration. This straightforward workflow minimizes operator exposure to hazardous chemicals and reduces the complexity of equipment requirements, making it an ideal candidate for technology transfer from lab to pilot plant.
- Combine aldehyde, malononitrile, and 7-hydroxy-2,3-dihydro-5H-[1,3]thiazolo[3,2-a]pyrimidin-5-one in a mixed solvent of ethanol and [Bmim]Br with a basic ionic liquid catalyst.
- Heat the reaction mixture in a water bath to a temperature range of 35-50°C and maintain magnetic stirring for 4-8 minutes until completion.
- Cool the mixture to room temperature to precipitate the solid product, then filter, wash with absolute ethanol, and vacuum dry to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based synthesis offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The most significant advantage is the drastic reduction in raw material costs achieved through the recyclability of the catalytic system. Since the ionic liquid and the mixed solvent can be recovered from the filtrate and reused directly for subsequent batches, the consumption of expensive reagents is minimized, leading to substantial cost savings over the lifecycle of the product. Additionally, the elimination of toxic organic base catalysts removes the need for costly waste treatment protocols associated with neutralizing and disposing of hazardous chemical waste, further enhancing the economic viability of the process. The mild reaction conditions also translate to lower energy consumption, as there is no requirement for high-temperature reflux or cryogenic cooling, aligning with corporate sustainability goals and reducing utility overheads.
- Cost Reduction in Manufacturing: The implementation of this technology fundamentally alters the cost structure of producing these complex heterocycles by removing the dependency on single-use catalysts. In traditional methods, the catalyst is lost to the waste stream, representing a direct financial loss with every batch produced. By contrast, the regenerable nature of the ionic liquid system means that the initial investment in the catalyst is amortized over dozens of production cycles. Furthermore, the simplified work-up procedure, which avoids complex extraction and chromatography steps, reduces labor hours and solvent usage, contributing to a leaner and more cost-effective manufacturing operation that improves overall margin potential.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of raw material markets and the regulatory pressures surrounding hazardous chemicals. This synthesis method mitigates such risks by utilizing readily available starting materials and a catalyst system that does not rely on scarce precious metals or strictly controlled substances. The robustness of the reaction, which tolerates a wide variety of aldehyde substrates including those with electron-withdrawing or electron-donating groups, ensures that production can continue even if specific raw material grades fluctuate. This flexibility allows manufacturers to source materials from a broader supplier base, reducing the risk of supply disruptions and ensuring a steady flow of high-quality intermediates to downstream API producers.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often exposes hidden inefficiencies, but this ionic liquid method is inherently scalable due to its homogeneous nature and mild thermal profile. The absence of exothermic spikes and the use of non-volatile ionic liquids significantly improve process safety, reducing the engineering controls required for large-scale reactors. From an environmental compliance perspective, the process generates minimal waste, as the solvent system is closed-loop and the catalyst is non-volatile and non-biodegradable in a way that prevents environmental contamination. This "green chemistry" profile simplifies the permitting process for new manufacturing facilities and helps companies meet increasingly stringent global environmental regulations regarding VOC emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this ionic liquid catalytic system. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or developing new synthetic routes for related analogues.
Q: What are the primary advantages of using basic ionic liquids over traditional organic bases in this synthesis?
A: Unlike traditional organic bases which generate significant waste and cannot be recycled, the basic ionic liquid catalyst described in patent CN114478580A allows for the regeneration and reuse of the catalytic system. This significantly reduces environmental pollution and lowers the overall cost of goods by minimizing solvent and catalyst consumption.
Q: How does the reaction temperature impact the yield and purity of the pyrano thiazolo pyrimidine derivatives?
A: The process operates efficiently at mild temperatures between 35-50°C. Maintaining this specific low-temperature range prevents thermal degradation of sensitive functional groups while ensuring rapid reaction kinetics, typically completing within 4-8 minutes. This results in consistently high purity levels exceeding 98% and yields often surpassing 90%.
Q: Can the solvent and catalyst system be reused for subsequent batches without loss of efficiency?
A: Yes, the filtrate containing the ethanol-[Bmim]Br mixed solvent and the ionic liquid catalyst can be directly reused for the next reaction cycle. Furthermore, after multiple uses, the system can be regenerated through distillation and washing, maintaining high catalytic activity and product purity over multiple cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano Thiazolo Pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN114478580A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to scale diverse synthetic pathways, ranging from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch. We understand that in the competitive landscape of API manufacturing, reliability and quality are paramount, and our team is dedicated to delivering intermediates that meet the highest international standards.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this efficient route can optimize your bill of materials. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for custom derivatives. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only robust and compliant but also driven by continuous innovation in green chemistry and process efficiency.
