Industrial Scale Synthesis of Dasatinib via Novel Thiophene Cyclization Route
Introduction to Advanced Dasatinib Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value oncology drugs like Dasatinib, a potent tyrosine kinase inhibitor. Patent CN103420999A introduces a transformative preparation method that addresses critical bottlenecks in existing manufacturing technologies. This innovation focuses on a streamlined sequence starting from 4-amino-5-chloro-2-methylpyrimidine, proceeding through a thiourea derivative, and culminating in the formation of the thiazole-pyrimidine core without relying on precious metal catalysis. The structural integrity of the target molecule, Dasatinib (Compound I), is maintained through a series of highly selective thermal reactions that favor industrial scalability.
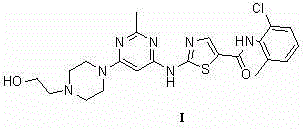
This technical breakthrough is particularly significant for supply chain stability, as it replaces complex cross-coupling reactions with more accessible thermal cyclization steps. By leveraging common reagents such as isothiocyanate derivatives and chloroacetamides, the process mitigates the supply risks associated with specialized catalysts. The methodology outlined in the patent provides a clear pathway for producing high-purity Dasatinib intermediates, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards for oncology treatments while optimizing the overall cost structure of production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Dasatinib, such as those disclosed in earlier patents like CN1980909, often rely heavily on palladium-catalyzed cross-coupling reactions to construct the critical carbon-nitrogen or carbon-carbon bonds. These methods, while chemically valid, present substantial hurdles for large-scale manufacturing due to the high cost of palladium catalysts and sophisticated ligands like BINAP. Furthermore, the removal of trace heavy metals from the final product requires additional purification steps, often involving column chromatography, which is notoriously difficult to scale and results in significant solvent waste and yield loss. The reliance on these expensive and operationally complex steps creates a fragile supply chain vulnerable to fluctuations in precious metal prices and availability.
The Novel Approach
In stark contrast, the method disclosed in CN103420999A utilizes a convergent strategy that builds the thiazole ring through a thermal cyclization mechanism. As illustrated in the reaction scheme below, the process begins with the formation of a thiourea intermediate, which is subsequently cyclized with a chloroacetamide derivative. This approach completely bypasses the need for transition metal catalysts, thereby simplifying the downstream processing requirements. The reaction conditions are mild, typically involving reflux in alcohols or ethers, and the products can often be isolated via simple crystallization or filtration. This shift from catalytic coupling to thermal cyclization represents a fundamental optimization in process chemistry, directly translating to lower operational expenditures and enhanced environmental compliance.
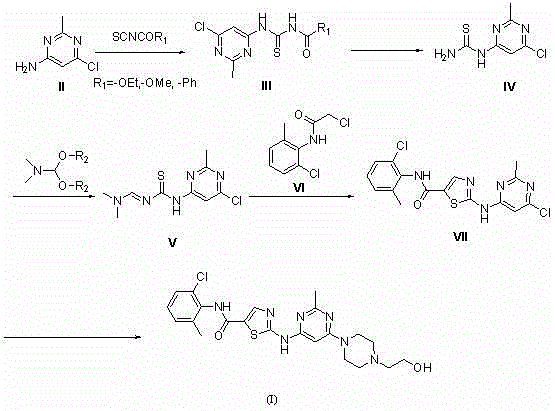
Mechanistic Insights into Thermal Thiophene Cyclization
The core of this innovative synthesis lies in the efficient construction of the thiazole moiety fused to the pyrimidine system. The mechanism initiates with the nucleophilic attack of the amino group on the pyrimidine ring onto an isothiocyanate derivative, forming a stable thiourea linkage. This intermediate is then subjected to basic conditions to generate a reactive species that undergoes cyclization upon exposure to an alpha-halo ketone or amide equivalent. The intramolecular cyclization is driven by the thermodynamic stability of the aromatic thiazole ring, a process that proceeds smoothly under reflux conditions without the need for external activation energy from metal catalysts. This mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric byproducts that often complicate purification in metal-catalyzed routes.
From an impurity control perspective, the absence of palladium eliminates a major class of genotoxic impurities concerns that plague many modern API syntheses. The purification strategy relies on the differential solubility of the intermediates in solvents like heptane, methanol, or isopropanol. For instance, the thiourea intermediate can be precipitated by adding non-polar solvents to the reaction mixture, allowing for the removal of soluble organic impurities before the cyclization step. This 'purify-as-you-go' approach prevents the carryover of impurities into the final stages, ensuring that the final Dasatinib product possesses a superior purity profile suitable for direct tableting or further formulation without extensive recrystallization cycles.
How to Synthesize Dasatinib Efficiently
The synthesis of Dasatinib via this novel route involves a sequential five-step process that transforms simple starting materials into the complex target molecule. The procedure begins with the preparation of a thiourea derivative from 4-amino-5-chloro-2-methylpyrimidine, followed by conversion to an imine intermediate using DMF acetals. The critical cyclization step involves reacting this imine with a chloroacetamide derivative to form the thiazole ring system. Finally, nucleophilic substitution with 1-(2-hydroxyethyl)piperazine completes the synthesis. The detailed standardized operating procedures, including specific stoichiometric ratios, temperature profiles, and workup instructions for each stage, are provided in the technical guide below.
- React 4-amino-5-chloro-2-methylpyrimidine with an isothiocyanate derivative to form the thiourea intermediate.
- Convert the thiourea derivative into an imine intermediate using DMF acetal under reflux conditions.
- Perform thermal cyclization with a chloroacetamide derivative followed by nucleophilic substitution with piperazine to yield Dasatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of precious metal catalysts fundamentally alters the cost structure of the manufacturing process, removing a volatile cost component linked to global commodity markets. Additionally, the simplified purification workflow reduces the consumption of chromatographic silica and vast quantities of solvents, leading to significant waste reduction and lower disposal costs. These factors combine to create a more resilient and cost-effective supply chain for Dasatinib intermediates, enabling manufacturers to offer competitive pricing while maintaining healthy margins.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of palladium catalysts and phosphine ligands, which are among the most expensive reagents in pharmaceutical synthesis. By replacing these with inexpensive thermal cyclization reagents, the direct material cost is drastically reduced. Furthermore, the avoidance of column chromatography in favor of crystallization and filtration significantly lowers processing time and solvent usage, resulting in substantial operational cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as 4-amino-5-chloro-2-methylpyrimidine and various isothiocyanates, are commodity chemicals with robust global supply networks. Unlike specialized catalysts that may have single-source suppliers or long lead times, these reagents are readily available from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers and pharmaceutical companies.
- Scalability and Environmental Compliance: The reaction conditions described, primarily involving reflux in standard organic solvents like ethanol or tetrahydrofuran, are inherently scalable from kilogram to multi-ton batches without requiring specialized high-pressure or low-temperature equipment. The simplified workup procedures generate less hazardous waste compared to metal-catalyzed processes, facilitating easier compliance with increasingly strict environmental regulations. This 'green chemistry' aspect not only reduces regulatory burden but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Dasatinib synthesis method. These answers are derived directly from the experimental data and claims presented in patent CN103420999A, providing clarity on the practical aspects of adopting this technology. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement teams assessing the long-term viability of the supply source.
Q: How does this new synthesis route improve upon traditional palladium-catalyzed methods?
A: The novel route described in patent CN103420999A eliminates the need for expensive palladium catalysts and ligands like BINAP, which are required in conventional cross-coupling methods. This significantly reduces raw material costs and removes the complex purification steps associated with removing heavy metal residues, thereby streamlining the manufacturing process.
Q: What are the key advantages for large-scale commercial production of Dasatinib?
A: The process utilizes mild reaction conditions, primarily involving reflux in common organic solvents like alcohols or ethers, which are easily manageable on a multi-ton scale. Furthermore, the isolation of intermediates is achieved through simple filtration and washing rather than column chromatography, drastically improving throughput and operational efficiency for industrial manufacturing.
Q: Does this method impact the purity profile of the final API?
A: Yes, by avoiding transition metal catalysts, the risk of metal contamination in the final active pharmaceutical ingredient is inherently minimized. The method emphasizes crystallization and filtration for purification, which typically yields a product with a cleaner impurity profile compared to methods requiring extensive chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dasatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving oncology medications. Our technical team has thoroughly analyzed the methodology presented in CN103420999A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to fruition. We are equipped with rigorous QC labs and advanced manufacturing facilities capable of meeting stringent purity specifications, ensuring that every batch of Dasatinib intermediate we produce adheres to the highest international quality standards.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and economics of your Dasatinib production.
