Advanced Synthesis of Bactericidal Thiazole Derivatives for Commercial Pharmaceutical Applications
Introduction to Novel Thiazole-Pyrimidine Hybrids
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer enhanced biological activity and improved safety profiles. Patent CN110172067B discloses a groundbreaking class of thiazole drug molecules possessing potent bactericidal activity, specifically targeting Gram-negative and Gram-positive bacteria. These compounds feature a unique fused thiazolo[3,2-a]pyrimidin-5-one core, which combines the pharmacological benefits of both thiazole and pyrimidine rings. The structural versatility of this scaffold allows for substitution at the phenyl ring (R = methyl, hydroxyl, or nitro), enabling fine-tuning of physicochemical properties for specific therapeutic applications. As a leading reliable pharmaceutical intermediate supplier, understanding the synthesis of such high-value scaffolds is critical for developing next-generation antibiotics.
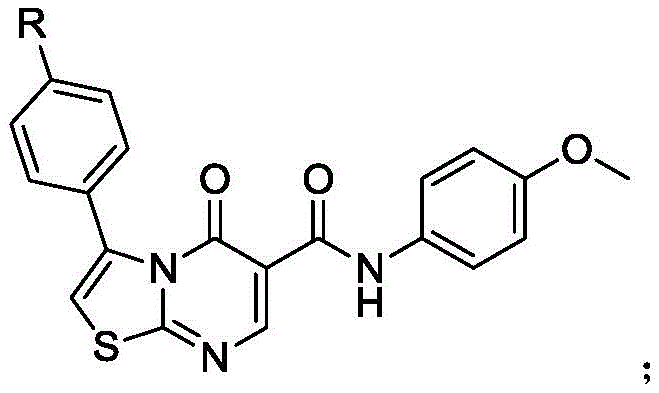
The significance of this technology lies not only in its biological potential but also in its synthetic accessibility. The patent outlines a robust six-step pathway that transforms simple acetophenone derivatives into complex, bioactive molecules. This approach addresses the growing demand for high-purity pharmaceutical intermediates capable of combating resistant bacterial strains. By leveraging efficient cyclization and oxidation strategies, this method provides a viable route for the commercial production of advanced antibacterial agents, positioning it as a key asset for R&D teams focused on infectious disease therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of fused thiazole-pyrimidine systems often relies on harsh reaction conditions that compromise yield and purity. Conventional routes frequently utilize stoichiometric amounts of toxic heavy metal oxidants or require extreme temperatures that lead to decomposition of sensitive intermediates. Furthermore, older methodologies may involve multi-step protection and deprotection sequences, drastically increasing the overall process mass intensity (PMI) and generating substantial chemical waste. These inefficiencies create significant bottlenecks in cost reduction in antibacterial drug manufacturing, making it difficult to scale production without incurring prohibitive expenses. Additionally, the use of non-selective reagents can result in complex impurity profiles that are challenging to remove, thereby delaying regulatory approval and market entry.
The Novel Approach
In contrast, the methodology described in CN110172067B introduces a streamlined and environmentally conscious strategy. The process employs a dual catalytic system involving pyrrolidine and p-toluenesulfonic acid to facilitate the initial thiazole ring formation under relatively mild conditions. A standout feature of this novel approach is the utilization of molecular oxygen (O2) mediated by N-hydroxyphthalimide (NHPI) for the oxidation steps. This green chemistry tactic eliminates the need for expensive and hazardous oxidizing agents like chromium or manganese salts. By integrating efficient one-pot transformations and utilizing commodity starting materials, this route significantly simplifies the operational complexity. This innovation directly supports the commercial scale-up of complex pharmaceutical intermediates by ensuring a safer, cleaner, and more economically viable production process.
Mechanistic Insights into Catalytic Cyclization and Oxidation
The core of this synthetic route lies in the construction of the fused heterocyclic system, which proceeds through a carefully orchestrated sequence of condensation and cyclization events. The formation of the thiazolo[3,2-a]pyrimidin-5-one scaffold begins with the reaction of the thiazole-2-amine intermediate with ethyl 3-oxopropionate. This step is catalyzed by p-toluenesulfonic acid and involves the nucleophilic attack of the amine nitrogen on the ester carbonyl, followed by intramolecular cyclization. The presence of sodium methoxide in the subsequent stage facilitates the elimination of ethanol and aromatization of the pyrimidine ring. This mechanism ensures high regioselectivity and minimizes the formation of isomeric byproducts, which is crucial for maintaining the stringent purity specifications required for API intermediates.
![Cyclization reaction forming the thiazolo[3,2-a]pyrimidin-5-one core](/insights/img/thiazole-drug-synthesis-pharma-supplier-20260305192932-05.webp)
Furthermore, the oxidation strategy employed in this patent represents a significant mechanistic advancement. The conversion of the hydroxymethyl group to the carboxylic acid is achieved via a two-step oxidation process involving silver nitrate/potassium persulfate and subsequently NHPI/O2. The NHPI-catalyzed aerobic oxidation operates through a radical mechanism where the phthalimide-N-oxyl (PINO) radical abstracts a hydrogen atom from the aldehyde intermediate. This generates an acyl radical that reacts with molecular oxygen to form a peroxy radical, eventually yielding the carboxylic acid. This catalytic cycle is highly efficient and operates at moderate pressures (0.1-0.2 MPa), demonstrating excellent atom economy. Such mechanistic precision allows for tight control over the reaction endpoint, preventing over-oxidation or degradation of the sensitive thiazole ring.
How to Synthesize Thiazole Drug Molecules Efficiently
The synthesis of these bactericidal thiazole derivatives follows a logical six-step progression that balances yield optimization with operational simplicity. The process begins with the condensation of substituted acetophenones with sulfur and cyanamide, followed by ring closure to form the bicyclic ketone. Subsequent functionalization involves hydroxymethylation, oxidation to the aldehyde and acid, and finally, amide coupling. Each step has been optimized in the patent examples to provide reproducible results with high purity. For detailed operational parameters, including exact molar ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Condense 4-substituted acetophenone with sulfur and cyanamide using pyrrolidine/p-TsOH catalysis to form the thiazole amine scaffold.
- Cyclize the thiazole amine with ethyl 3-oxopropionate under reflux with p-TsOH and sodium methoxide to form the fused pyrimidinone ring.
- Perform hydroxymethylation using paraformaldehyde and triethylamine, followed by sequential oxidation to aldehyde and carboxylic acid.
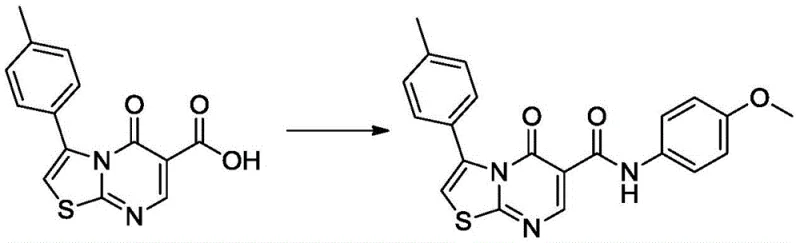
- Complete the synthesis via amide coupling with p-anisidine using EDC/HOBt activation to yield the final bactericidal target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route outlined in CN110172067B offers distinct advantages that translate into tangible business value. The reliance on readily available bulk chemicals such as acetophenones, elemental sulfur, and paraformaldehyde ensures a stable supply base that is not subject to the volatility associated with exotic reagents. This stability is paramount for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to market demands. Moreover, the elimination of heavy metal catalysts in the oxidation steps simplifies the downstream purification process, reducing the burden on waste treatment facilities and lowering the overall environmental compliance costs.
- Cost Reduction in Manufacturing: The implementation of catalytic aerobic oxidation significantly lowers raw material costs by replacing expensive stoichiometric oxidants with inexpensive oxygen gas. This shift not only reduces the direct cost of goods sold (COGS) but also minimizes the expense associated with the disposal of hazardous heavy metal waste. The high atom efficiency of the cyclization steps further contributes to yield maximization, ensuring that a greater proportion of input materials are converted into valuable product. Consequently, this process enables substantial cost savings in the large-scale production of antibacterial drug candidates without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The use of commodity starting materials mitigates the risk of supply chain disruptions caused by the scarcity of specialized reagents. Since the key building blocks are produced globally in large volumes, procurement teams can secure long-term contracts with multiple suppliers, ensuring continuity of supply. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high pressures, further enhances manufacturability. This reliability is essential for maintaining consistent production schedules and meeting the rigorous delivery timelines expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with steps such as reflux condensation and filtration being easily adaptable from laboratory to pilot and commercial scales. The avoidance of toxic solvents and reagents aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing site. By minimizing the generation of hazardous byproducts, the process simplifies effluent treatment and helps companies meet increasingly stringent environmental regulations. This sustainable approach not only protects the environment but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these thiazole derivatives. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing product pipelines. The information provided covers aspects ranging from biological efficacy to process safety and scalability.
Q: What is the antibacterial spectrum of the synthesized thiazole derivatives?
A: According to patent CN110172067B, the target compounds exhibit significant inhibitory activity against Escherichia coli and Bacillus subtilis, with the nitro-substituted variant showing superior efficacy compared to methyl or hydroxyl analogs.
Q: How does the novel oxidation method improve process safety?
A: The process utilizes molecular oxygen (O2) catalyzed by N-hydroxyphthalimide (NHPI) for the aldehyde-to-acid conversion, replacing hazardous stoichiometric heavy metal oxidants and significantly reducing toxic waste generation.
Q: Are the starting materials commercially available for scale-up?
A: Yes, the synthesis begins with commodity chemicals such as 4-methylacetophenone, elemental sulfur, and cyanamide, ensuring a robust and cost-effective supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazole Drug Molecule Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the thiazole-pyrimidine scaffold described in CN110172067B for the development of novel antibiotics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, including pressurized oxidation reactors and advanced purification suites. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative synthetic route for your antibacterial drug programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Together, we can accelerate the development of life-saving medicines while optimizing your supply chain efficiency and cost structure.
