Advanced Manufacturing of 1,1,1-Trifluoro-2-Methyl-2-Propanol for Global Pharmaceutical Supply Chains
The global demand for fluorine-containing fine chemicals has surged dramatically in recent years, driven by the critical role these molecular building blocks play in modern drug discovery and agrochemical development. Among these essential compounds, 1,1,1-trifluoro-2-methyl-2-propanol stands out as a pivotal intermediate, valued for the unique electronic and steric properties imparted by its trifluoromethyl group. However, traditional manufacturing routes have long been plagued by inefficiencies, including low yields, harsh reaction conditions, and significant safety hazards associated with volatile raw materials. A groundbreaking technical solution detailed in patent CN113735684A addresses these historical bottlenecks by introducing a novel catalytic system that fundamentally transforms the production landscape. This report provides a deep technical and commercial analysis of this advanced synthesis method, demonstrating how it enables reliable pharmaceutical intermediates supplier capabilities through superior process chemistry. By leveraging a gallium trichloride-catalyzed Grignard reaction, manufacturers can now achieve unprecedented levels of efficiency and purity, directly translating to enhanced supply chain stability and cost competitiveness for downstream partners seeking high-purity OLED material or pharmaceutical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1,1-trifluoro-2-methyl-2-propanol has been fraught with significant technical challenges that hindered its widespread industrial adoption and economic viability. Early literature from the mid-20th century described routes utilizing trifluoroacetyl chloride or trifluoroacetic anhydride reacting with methyl magnesium bromide, but these methods suffered from abysmal yields ranging merely from 40% to 50%. Furthermore, the raw materials involved, such as trifluoroacetyl chloride, possess low boiling points and high volatility, making them extremely difficult and dangerous to transport and handle on a commercial scale. Subsequent attempts involving catalytic hydrogenation required extreme temperatures up to 170°C and high-pressure equipment, imposing heavy capital expenditure burdens and safety risks on manufacturing facilities. Other reported pathways using trifluoromethyl iodide resulted in Grignard reagent yields as low as 9%, rendering the process economically unfeasible for any serious cost reduction in fine chemical manufacturing. These legacy methods also frequently produced complex by-product mixtures with boiling points similar to the target product, necessitating energy-intensive and technically difficult purification steps that further eroded profit margins and extended production lead times.
The Novel Approach
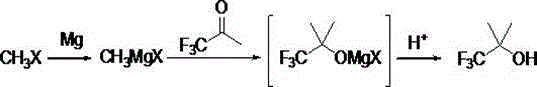
In stark contrast to these inefficient legacy protocols, the innovative method disclosed in CN113735684A introduces a streamlined and highly efficient pathway that resolves the core thermodynamic and kinetic limitations of previous art. This novel approach utilizes methyl halide, specifically methyl chloride, as a readily available and cost-effective starting material to generate the necessary methyl magnesium halide Grignard reagent in situ. The key breakthrough lies in the introduction of gallium trichloride as a specialized Lewis acid catalyst during the coupling reaction with 1,1,1-trifluoroacetone. This catalytic intervention not only accelerates the reaction kinetics but also directs the selectivity towards the desired alcohol product, thereby drastically minimizing the formation of unwanted side products. The process operates under remarkably mild conditions, typically between -20°C and 10°C, eliminating the need for high-pressure reactors or extreme thermal inputs. Consequently, this methodology facilitates the commercial scale-up of complex polymer additives and pharmaceutical intermediates by offering a robust, safe, and high-yielding alternative that simplifies the entire production workflow from raw material intake to final isolation.
Mechanistic Insights into GaCl3-Catalyzed Grignard Cyclization
The core of this technological advancement rests on the sophisticated interplay between the Grignard reagent and the ketone substrate, mediated by the gallium trichloride catalyst. In the absence of a catalyst, the nucleophilic attack of the methyl magnesium halide on the carbonyl carbon of 1,1,1-trifluoroacetone can be sluggish or non-selective due to the strong electron-withdrawing nature of the trifluoromethyl group. The introduction of GaCl3 acts as a potent Lewis acid, coordinating with the oxygen atom of the carbonyl group to significantly enhance its electrophilicity. This activation lowers the energy barrier for the nucleophilic addition, allowing the reaction to proceed rapidly even at sub-ambient temperatures. The mechanistic pathway ensures that the methyl group is transferred efficiently to the carbonyl carbon, forming a stable magnesium alkoxide intermediate which is subsequently protonated during the quenching phase to yield the final alcohol. This precise control over the reaction trajectory is crucial for maintaining high stereochemical integrity and preventing the degradation of the sensitive trifluoromethyl moiety, which is often prone to defluorination under harsher conditions found in uncatalyzed variants.
Beyond mere yield enhancement, the catalytic mechanism plays a pivotal role in impurity control, a factor of paramount importance for R&D Directors focused on purity and impurity profiles. Traditional methods often generated significant amounts of coupling by-products or reduction side-products that were structurally similar to the target molecule, complicating purification. The GaCl3-catalyzed system promotes a cleaner reaction profile, ensuring that the crude reaction mixture contains a overwhelming majority of the desired 1,1,1-trifluoro-2-methyl-2-propanol. This high selectivity reduces the burden on downstream purification units, such as distillation columns, allowing for simpler separation protocols. The use of n-butyl ether as a solvent further aids in this regard, as its boiling point differs sufficiently from the product to allow for easy removal via normal pressure distillation. This mechanistic elegance translates directly into a final product with purity levels consistently exceeding 99%, meeting the rigorous specifications required for sensitive pharmaceutical applications where trace impurities can compromise drug safety and efficacy.
How to Synthesize 1,1,1-Trifluoro-2-Methyl-2-Propanol Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized operational parameters defined in the patent to ensure maximum efficiency and safety. The process begins with the careful generation of the Grignard reagent under an inert nitrogen atmosphere to prevent moisture ingress, followed by the controlled addition of the ketone substrate in the presence of the catalyst. Temperature management is critical during the addition phase, where maintaining the reaction between -20°C and 10°C ensures optimal selectivity and prevents exothermic runaways. The subsequent workup involves a standard acidic quench followed by phase separation and distillation, procedures that are well-understood in industrial organic synthesis yet optimized here for minimal loss. For process engineers and plant managers looking to adopt this technology, the detailed standardized synthesis steps outlined below provide a clear roadmap for translating this laboratory-scale innovation into a robust commercial manufacturing protocol that guarantees consistent quality and output.
- Preparation of Grignard Reagent: React magnesium chips with methyl halide in an ether solvent under nitrogen protection, initiating with iodine and heat.
- Catalytic Addition: Mix 1,1,1-trifluoroacetone with gallium trichloride catalyst in ether, cool to -20 to 10°C, and slowly add the Grignard reagent.
- Workup and Purification: Quench with acidic aqueous solution, separate layers, wash, dry, and distill at normal pressure to collect the product at 80°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this GaCl3-catalyzed manufacturing process represents a strategic opportunity to optimize costs and secure supply continuity. Unlike legacy methods that relied on expensive or hazardous reagents like trifluoroacetyl chloride, this new route utilizes methyl chloride, a commodity chemical that is abundantly available and significantly cheaper. The elimination of high-pressure and high-temperature requirements reduces the strain on production equipment, lowering maintenance costs and extending the lifespan of capital assets. Furthermore, the simplified purification process means that production cycles are shorter, allowing for faster turnover and reduced inventory holding costs. These factors combine to create a manufacturing environment that is not only more economical but also more resilient to market fluctuations, ensuring that partners can rely on a stable source of high-purity pharmaceutical intermediates without the risk of supply disruptions caused by complex or fragile production processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly raw materials with inexpensive commodity chemicals and the drastic improvement in reaction yield. By achieving yields exceeding 90% compared to the historical average of 40-50%, the amount of raw material required per unit of product is nearly halved, leading to substantial cost savings in material procurement. Additionally, the use of n-butyl ether as a solvent facilitates easy recovery and recycling, further reducing operational expenditures related to solvent consumption and waste disposal. The avoidance of expensive transition metal catalysts or complex ligand systems also contributes to a leaner cost structure, making the final product highly competitive in the global market while maintaining healthy margins for manufacturers.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the robustness and simplicity of the new synthetic route. The reliance on widely available raw materials like methyl chloride and magnesium eliminates the risk of shortages associated with specialized or niche reagents used in older methods. Moreover, the mild reaction conditions reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, ensuring consistent production output. The high purity of the crude product minimizes the need for extensive reprocessing or second-run distillations, which often act as bottlenecks in chemical manufacturing. This operational reliability allows supply chain planners to forecast delivery timelines with greater accuracy, reducing lead time for high-purity pharmaceutical intermediates and enabling just-in-time delivery models for downstream clients.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages that align with modern green chemistry principles and regulatory standards. The process generates fewer by-products and waste streams compared to traditional routes, simplifying effluent treatment and reducing the environmental footprint of the manufacturing facility. The absence of high-pressure operations lowers the safety risk profile, facilitating easier regulatory approval for capacity expansions. The straightforward workup procedure, involving simple aqueous quenching and distillation, is easily adaptable from pilot scale to multi-ton production without requiring fundamental changes to the process design. This inherent scalability ensures that manufacturers can rapidly respond to increasing market demand, supporting the commercial scale-up of complex pharmaceutical additives while maintaining strict adherence to environmental and safety regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the patent data and practical manufacturing considerations, providing clarity for stakeholders evaluating this process for their supply chains. Understanding these details is essential for making informed decisions about adopting this method for large-scale production of fluorine-containing intermediates.
Q: What are the primary advantages of the GaCl3 catalyzed method over conventional synthesis?
A: The GaCl3 catalyzed method significantly improves yield to over 90% compared to historical methods yielding 21-50%, while operating under milder temperature conditions and using more stable raw materials.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes easily obtained raw materials like methyl chloride and n-butyl ether, features convenient post-treatment purification, and avoids high-pressure equipment, making it ideal for commercial scale-up.
Q: What purity levels can be achieved with this manufacturing process?
A: The optimized protocol consistently achieves product purity exceeding 99% via GC area normalization, meeting stringent requirements for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,1-Trifluoro-2-Methyl-2-Propanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chemical intermediates requires more than just a patented reaction; it demands a partner with the infrastructure and expertise to execute it flawlessly. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the GaCl3-catalyzed route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that every kilogram of 1,1,1-trifluoro-2-methyl-2-propanol we deliver meets the exacting requirements of global pharmaceutical and agrochemical clients, providing a foundation of trust and reliability for your most critical projects.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to validate the performance of our material against your internal benchmarks. By choosing NINGBO INNO PHARMCHEM, you are not just purchasing a chemical; you are securing a strategic alliance dedicated to driving innovation and efficiency in your production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
