Advanced Photocatalytic Synthesis of Sulfonyl-Substituted Alpha-Cyclopropyl Styrenes for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures, particularly those containing strained rings and polar functional groups. Patent CN110483346B introduces a groundbreaking approach for the synthesis of sulfonyl-substituted α-cyclopropyl styrene compounds, utilizing clean and sustainable visible light as the energy source. This technology addresses a critical challenge in medicinal chemistry: the simultaneous introduction of cyclopropyl and sulfonyl groups while retaining the valuable exocyclic double bond structure. The cyclopropyl moiety is renowned for its metabolic stability and ability to modulate lipophilicity, found in blockbuster drugs like ciprofloxacin and celecoxib, while the sulfonyl group enhances solubility and hydrogen bonding capabilities. By leveraging a photoredox catalytic system, this invention offers a pathway to high-purity intermediates that were previously difficult to access without compromising structural integrity.
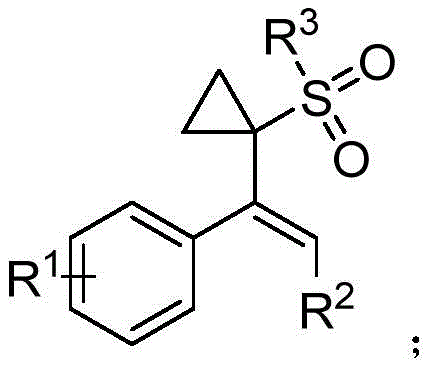
For R&D Directors focused on novel scaffold exploration, the ability to access these specific chemical spaces is invaluable. The general structure disclosed allows for significant diversification at the R1, R2, and R3 positions, enabling the rapid generation of libraries for structure-activity relationship (SAR) studies. This level of flexibility is crucial during the lead optimization phase of drug discovery, where minor structural changes can drastically improve pharmacokinetic profiles. Furthermore, the retention of the double bond provides a versatile handle for subsequent transformations, such as hydrogenation or epoxidation, expanding the utility of these intermediates beyond their immediate application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of molecules containing both cyclopropyl and sulfonyl functionalities has been fraught with difficulties due to the inherent instability of the cyclopropane ring under harsh reaction conditions. Traditional synthetic routes often rely on strong bases, strong acids, or stoichiometric amounts of aggressive oxidants to facilitate sulfonylation. However, these rigorous conditions frequently lead to the undesired ring-opening of the cyclopropyl group or the degradation of the sensitive exocyclic double bond. Such side reactions not only reduce the overall yield of the target molecule but also generate complex impurity profiles that are challenging and costly to remove during purification. For procurement managers, this translates to higher raw material consumption and increased waste disposal costs, while for supply chain heads, it introduces variability in batch-to-batch consistency and potential delays in production timelines.
The Novel Approach
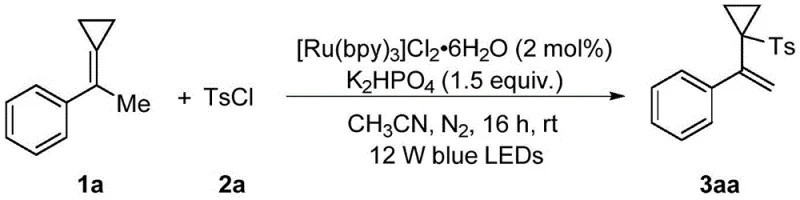
The methodology described in CN110483346B represents a paradigm shift by employing visible-light photocatalysis to drive the reaction under exceptionally mild conditions. By utilizing methylenecyclopropane compounds as substrates and reacting them with benzenesulfonyl chloride in the presence of a photocatalyst like [Ru(bpy)3]Cl2·6H2O, the process successfully installs the sulfonyl group without destroying the cyclopropyl ring. This approach operates at room temperature and atmospheric pressure, eliminating the need for energy-intensive heating or cooling systems. The reaction demonstrates excellent step economy and substrate universality, accommodating various electronic and steric environments on the aromatic rings. This technological advancement ensures that the final products retain their intended structural features, providing a reliable source of high-quality intermediates for downstream pharmaceutical manufacturing.
Mechanistic Insights into Visible-Light Photocatalytic Sulfonylation
The core of this innovation lies in the photoredox catalytic cycle initiated by the absorption of visible light by the ruthenium complex. Upon irradiation with blue LEDs, the photocatalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the sulfonyl chloride substrate. This interaction generates a sulfonyl radical species, which is highly reactive yet selective enough to add to the electron-rich double bond of the methylenecyclopropane without inducing immediate ring fragmentation. The subsequent steps involve radical recombination or further oxidation/reduction events that stabilize the intermediate, ultimately yielding the sulfonyl-substituted product while preserving the strained three-membered ring. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters such as light intensity, catalyst loading, and solvent choice to maximize efficiency and minimize byproduct formation.
From an impurity control perspective, the mild nature of the radical mechanism significantly reduces the formation of thermal degradation products commonly seen in traditional ionic reactions. The use of a base like K2HPO4 helps to neutralize the hydrochloric acid byproduct generated during the reaction, preventing acid-catalyzed ring opening of the cyclopropane. This careful balance of reactivity ensures a cleaner reaction profile, simplifying the downstream purification process. For quality assurance teams, this means that the resulting intermediates meet stringent purity specifications with less effort, reducing the risk of genotoxic impurities or heavy metal residues that might arise from harsher reagents. The ability to tune the reaction by adjusting the molar ratios of substrates, as demonstrated in the optimization experiments, further empowers manufacturers to tailor the process for specific scale-up requirements.
How to Synthesize Sulfonyl-Substituted Alpha-Cyclopropyl Styrenes Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible method for producing these valuable compounds. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent oxygen quenching of the excited photocatalyst. Key reagents include the methylenecyclopropane substrate, the sulfonyl chloride coupling partner, a catalytic amount of the ruthenium complex, and a mild inorganic base. The reaction is typically conducted in anhydrous acetonitrile, although other polar aprotic solvents may also be suitable depending on substrate solubility. Following the irradiation period, the workup involves simple solvent removal and chromatographic purification, making it accessible for both laboratory-scale discovery and pilot-scale production. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reactor with methylenecyclopropane compound, benzenesulfonyl chloride, photocatalyst (e.g., [Ru(bpy)3]Cl2·6H2O), base (e.g., K2HPO4), and anhydrous solvent under nitrogen.
- Irradiate the reaction mixture with visible light (e.g., 12W blue LEDs) while stirring at room temperature for 16 to 30 hours.
- Remove the solvent via rotary evaporation and purify the crude product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this visible-light mediated synthesis strategy offers substantial strategic benefits for organizations focused on cost efficiency and supply chain resilience. The shift away from extreme reaction conditions directly impacts the total cost of ownership for manufacturing these intermediates. By operating at room temperature, facilities can significantly reduce energy consumption associated with heating mantles or cryogenic cooling systems, leading to a lower carbon footprint and reduced utility expenses. Furthermore, the simplicity of the workup procedure minimizes the requirement for specialized extraction equipment or complex distillation setups, allowing existing infrastructure to be utilized effectively. This operational simplicity translates into faster turnaround times and improved throughput capabilities for contract development and manufacturing organizations (CDMOs).
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the use of catalytic amounts of readily available photocatalysts drastically lowers the raw material costs per kilogram of product. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and associated piping, extending the lifespan of capital equipment. The high atom economy of the reaction ensures that a greater proportion of the starting materials ends up in the final product, minimizing waste generation and the associated costs of hazardous waste disposal. These factors collectively contribute to a more economically viable manufacturing process that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as methylenecyclopropanes and benzenesulfonyl chlorides, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to variations in environmental factors, ensuring consistent output quality regardless of seasonal changes or facility location. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery demands of downstream pharmaceutical clients. Moreover, the scalability of the photochemical process has been well-documented in recent literature, providing confidence that laboratory success can be translated to commercial tonnage without significant re-engineering.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this green chemistry approach positions manufacturers favorably for future compliance. The use of visible light as a traceless reagent aligns with the principles of green chemistry, avoiding the generation of toxic byproducts common in traditional oxidation methods. The solvent systems employed are standard and can be easily recovered and recycled, further enhancing the environmental profile of the process. For supply chain heads, this means easier permitting for new production lines and a stronger corporate social responsibility (CSR) narrative when engaging with eco-conscious partners. The ability to scale this reaction from grams to kilograms using flow chemistry or large batch reactors ensures that supply can meet growing market demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details helps in assessing the feasibility of integrating this method into existing production workflows.
Q: What are the key advantages of this photocatalytic method over traditional synthesis?
A: Unlike traditional methods requiring strong acids or oxidants that degrade the sensitive cyclopropyl ring, this visible-light protocol operates under mild room temperature conditions, preserving both the cyclopropyl group and the exocyclic double bond structure essential for downstream functionalization.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes simple operational steps, commercially available starting materials like methylenecyclopropanes and sulfonyl chlorides, and standard LED lighting, making it highly suitable for scale-up from laboratory to industrial manufacturing without complex high-pressure equipment.
Q: What is the substrate scope regarding the aromatic ring substituents?
A: The method demonstrates excellent universality, tolerating a wide range of substituents including alkyl, alkoxy, halogens (F, Cl, Br, I), trifluoromethyl, cyano, and nitro groups on the phenyl ring, allowing for the synthesis of diverse drug-like scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonyl-Substituted Alpha-Cyclopropyl Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modern drug synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with specialized photochemical reactors and rigorous QC labs capable of handling sensitive intermediates with stringent purity specifications. We are committed to delivering high-quality sulfonyl-substituted alpha-cyclopropyl styrenes that meet the exacting standards of the global pharmaceutical industry, supporting your pipeline from early-stage development through to commercial launch.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your time-to-market while maximizing value efficiency.
