Scalable Metal-Free Synthesis of 4-Iodo-N-Arylpyrazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN114539157A introduces a significant advancement in this domain by disclosing a novel iodine-promoted oxidation method for preparing 4-iodo-N-arylpyrazole compounds. This technology addresses the growing demand for efficient halogenated heterocycles, which are indispensable precursors in cross-coupling reactions for drug discovery. By utilizing N-aryl-3-pyrazolidone derivatives as starting materials, the process achieves a tandem dehydroaromatization and iodination sequence under remarkably mild conditions. For R&D directors and procurement specialists, this represents a strategic opportunity to streamline the supply chain for complex pyrazole intermediates while adhering to strict regulatory standards regarding impurity profiles.
The significance of this invention extends beyond mere academic interest; it offers a practical solution for the mass preparation of compounds that are foundational to modern medicinal chemistry. Pyrazole rings are ubiquitous in top-selling drugs such as Celebrex and agrochemical agents like Fipronil. The ability to directly install an iodine atom at the 4-position of the pyrazole ring facilitates downstream diversification through Suzuki, Heck, or Sonogashira couplings. Consequently, adopting this methodology allows chemical manufacturers to offer high-purity pharmaceutical intermediates with reduced lead times and enhanced cost competitiveness in a global market that increasingly values sustainability and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-arylpyrazole derivatives has relied heavily on the condensation of N-arylhydrazines with 1,3-dicarbonyl compounds or their equivalents. While this Knorr-type cyclization is well-established, it often suffers from significant drawbacks when applied to the synthesis of specifically halogenated derivatives. Conventional halogenation strategies frequently require harsh reaction conditions, strong acidic or basic media, and the use of stoichiometric amounts of hazardous halogenating agents. Furthermore, achieving regioselective iodination at the 4-position without affecting other sensitive functional groups on the aryl ring remains a persistent challenge. These limitations often result in complex purification processes, lower overall yields, and the generation of substantial chemical waste, which drives up manufacturing costs and complicates environmental compliance for large-scale production facilities.
The Novel Approach
In stark contrast, the method disclosed in patent CN114539157A utilizes a transition-metal-free oxidative strategy that fundamentally simplifies the synthetic workflow. By employing molecular iodine as both the oxidant and the iodine source in dimethyl sulfoxide (DMSO), the reaction proceeds through a concerted dehydroaromatization and iodination mechanism. This approach eliminates the need for expensive and toxic transition metal catalysts such as palladium or copper, which are traditionally associated with high costs and rigorous removal requirements in pharmaceutical manufacturing. The reaction operates effectively at temperatures between 100-120°C under an air atmosphere, removing the necessity for inert gas protection systems. This simplicity translates directly into operational advantages, including reduced equipment complexity, lower energy consumption, and a significantly simplified post-reaction workup procedure involving standard extraction and chromatography.
Mechanistic Insights into Iodine-Promoted Oxidative Aromatization
The core innovation of this technology lies in the dual role of iodine within the reaction system. In the presence of DMSO, iodine acts not merely as an electrophilic iodinating agent but also facilitates the oxidative dehydrogenation of the saturated pyrazolidone ring to form the aromatic pyrazole system. The mechanism likely involves the initial formation of an iodonium species or an iodine-DMSO complex that activates the alpha-position of the carbonyl group. Subsequent elimination steps lead to the formation of the double bond required for aromatization, concurrent with the electrophilic substitution of the iodine atom at the 4-position of the newly formed pyrazole ring. This tandem process is highly efficient because it combines two distinct chemical transformations into a single operational step, thereby minimizing intermediate isolation and handling losses.
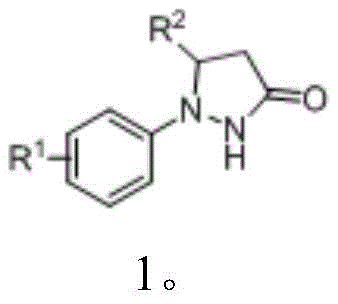
Furthermore, the reaction demonstrates exceptional functional group tolerance, accommodating a wide variety of substituents on the N-aryl ring, including halogens, alkyl groups, and trifluoromethyl moieties. This robustness is critical for process chemists who must design synthetic routes that remain stable despite variations in raw material quality or slight deviations in reaction parameters. The absence of transition metals also means that the final product is free from heavy metal residues, a critical quality attribute for active pharmaceutical ingredients (APIs). The mechanistic pathway ensures that the iodine atom is installed regioselectively, providing a consistent and reliable source of 4-iodo-N-arylpyrazoles that can be confidently used in subsequent metal-catalyzed coupling reactions without the risk of catalyst poisoning from residual impurities.
How to Synthesize 4-Iodo-N-Arylpyrazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The process begins with the dissolution of the N-aryl-3-pyrazolidone substrate in DMSO, followed by the addition of molecular iodine. The reaction mixture is then heated to maintain a temperature range of 100-120°C for a duration of 18-24 hours. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the optimal endpoint for specific substrates. Upon completion, the workup involves quenching with saturated sodium thiosulfate solution to reduce excess iodine, followed by extraction with ethyl acetate. The detailed standardized synthesis steps, including precise stoichiometric ratios and purification protocols, are outlined in the guide below.
- Dissolve N-aryl-3-pyrazolidone starting material in dimethyl sulfoxide (DMSO) solvent within a reaction vessel equipped with magnetic stirring.
- Add molecular iodine (I2) as both the catalyst and iodinating reagent to the reaction mixture under ambient air conditions.
- Heat the reaction mixture to 100-120°C for 18-24 hours, then perform standard aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-promoted oxidation method offers tangible benefits that extend beyond technical performance. The elimination of transition metal catalysts represents a major cost-saving opportunity, as it removes the need for expensive noble metals and the associated downstream purification technologies required to meet strict residual metal limits. Additionally, the use of commodity chemicals like iodine and DMSO ensures a stable and secure supply of raw materials, mitigating the risks associated with sourcing specialized reagents. The simplicity of the reaction conditions, particularly the ability to run the process under air rather than inert atmosphere, reduces capital expenditure on specialized reactor equipment and lowers operational overheads related to nitrogen or argon consumption.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the significant reduction in reagent costs and waste disposal fees. By avoiding precious metal catalysts, manufacturers can drastically lower the bill of materials for each batch. Furthermore, the simplified workup procedure, which relies on standard liquid-liquid extraction and column chromatography, reduces the labor hours and solvent volumes required for purification. This streamlined workflow enhances overall throughput, allowing facilities to produce larger quantities of high-purity pharmaceutical intermediates within the same timeframe, effectively driving down the unit cost of production without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable chemical inputs. Iodine and DMSO are bulk commodities with established global supply networks, reducing the vulnerability to shortages that often plague specialized catalytic systems. The robustness of the reaction against atmospheric moisture and oxygen further simplifies logistics, as there is no need for stringent exclusion of air during storage or transfer of reagents. This reliability ensures consistent production schedules and shorter lead times for delivering critical intermediates to downstream customers, fostering stronger partnerships and trust within the pharmaceutical supply network.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by improving atom economy and reducing the toxicity of the catalytic system. The absence of heavy metals simplifies wastewater treatment and solid waste disposal, lowering the environmental footprint of the manufacturing process. The reaction conditions are mild enough to be safely scaled from kilogram to multi-ton production levels without encountering significant exothermic hazards or pressure buildup. This scalability ensures that the technology can grow with market demand, providing a sustainable long-term solution for the commercial manufacture of complex pyrazole derivatives needed for next-generation therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted oxidation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, impurity profiles, and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this method into existing production workflows or new product development pipelines.
Q: Does this synthesis method introduce heavy metal impurities?
A: No, the process utilizes molecular iodine and DMSO, completely avoiding transition metal catalysts like palladium or copper, which ensures the final product meets stringent pharmaceutical heavy metal specifications without requiring complex scavenging steps.
Q: What is the typical yield range for this oxidative iodination process?
A: According to patent data, the reaction yields vary based on substrate substituents, generally ranging from 28% to 88%, with electron-neutral and electron-deficient substrates typically performing better than electron-rich ones.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method operates under air atmosphere without inert gas protection and uses common solvents like DMSO, making it highly scalable and cost-effective for commercial manufacturing of pyrazole intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Iodo-N-Arylpyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. By leveraging innovative technologies like the iodine-promoted oxidation method described in patent CN114539157A, we provide a competitive edge in the supply of complex heterocyclic building blocks.
We invite you to collaborate with us to optimize your supply chain for pyrazole-based projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for custom synthesis projects. Let us be your partner in accelerating the journey from laboratory discovery to commercial reality.
