Advanced Disulfide Porphyrin Catalysts: Revolutionizing Industrial Oxidation Processes
The landscape of fine organic synthesis is undergoing a significant transformation driven by the urgent need for greener, more efficient oxidation technologies. Traditional methods relying on stoichiometric oxidants such as potassium permanganate or potassium dichromate are increasingly obsolete due to their severe environmental footprint and poor atom economy. Patent CN101870703B introduces a groundbreaking solution through the development of linked disulfide propyl porphyrin and its metal complexes. This innovation represents a paradigm shift towards biomimetic catalysis, where the catalyst structure itself integrates the functions of both the primary catalyst and the co-catalyst. By simulating the active center of monooxygenase cytochrome P450, this technology enables the direct use of molecular oxygen or air as the oxidant, drastically reducing waste generation and operational hazards associated with hazardous chemical oxidants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of hydrocarbons, alcohols, and sulfides in the pharmaceutical and agrochemical industries has relied heavily on stoichiometric oxidants that generate substantial amounts of inorganic salt waste. These traditional processes not only suffer from low atom efficiency but also require complex downstream purification steps to remove heavy metal residues and salt byproducts. Furthermore, many conventional metalloporphyrin catalysts lack sufficient stability or activity when used alone, often necessitating the addition of external co-catalysts. This addition complicates the reaction system, negatively impacts product selectivity, and creates significant challenges in separating the final product from the catalyst mixture, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
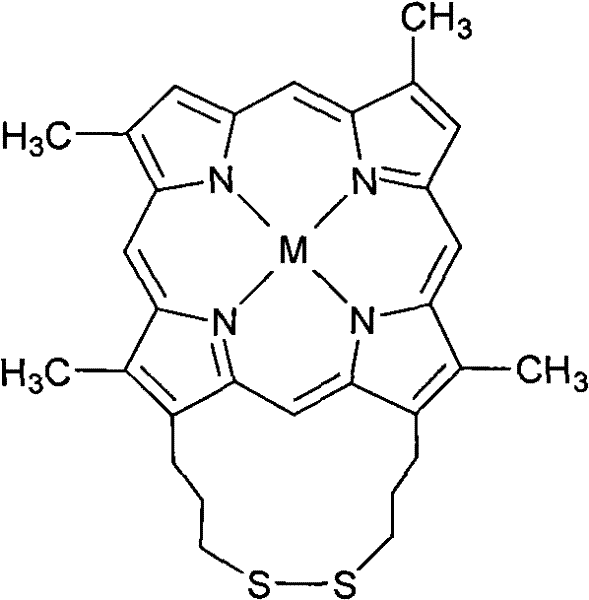
The novel approach detailed in the patent overcomes these hurdles by engineering a porphyrin molecule with an intrinsic disulfide bridge at the 13 and 17 positions. This structural modification creates a unified catalytic system where the disulfide group acts as an internal electron donor, effectively replacing the need for external co-catalysts. This integration significantly simplifies the reaction workflow and enhances the stability of the catalyst under oxidative conditions. Unlike previous generations of porphyrin catalysts that suffered from steric hindrance or instability due to complex bridging groups like cystine, this linked disulfide propyl design offers a more streamlined molecular architecture. The result is a robust catalyst capable of operating under milder temperatures and pressures while maintaining high conversion rates and exceptional selectivity for target oxidation products.
Mechanistic Insights into Biomimetic Oxidation Catalysis
The core innovation lies in the unique molecular architecture of the linked disulfide propyl porphyrin metal complex. The structure features a porphyrin macrocycle coordinated with a transition metal center (such as Cobalt, Manganese, or Iron), flanked by a flexible disulfide-containing side chain. This design is critical because it mimics the natural enzymatic environment where multiple functional groups work in concert to activate molecular oxygen. The disulfide bond serves as a redox-active site that facilitates electron transfer during the catalytic cycle, stabilizing high-valent metal-oxo intermediates that are responsible for the actual oxidation of the substrate. This synergistic effect between the metal center and the sulfur bridge allows the catalyst to activate inert C-H bonds in alkanes or oxidize sensitive functional groups like sulfides without over-oxidation.
Furthermore, the impurity profile of reactions utilizing this catalyst is markedly superior to traditional methods. Because the catalyst operates via a selective radical or concerted mechanism rather than non-specific harsh oxidation, side reactions such as ring opening or over-oxidation to carboxylic acids are minimized. The patent data indicates that for the oxidation of cyclohexane, the selectivity for cyclohexanone and cyclohexanol can reach levels between 90% and 97%. This high selectivity is crucial for R&D directors aiming to minimize downstream purification burdens. The stability of the disulfide linkage under the reaction conditions ensures that the catalyst maintains its integrity over multiple cycles, preventing the leaching of metal ions into the product stream, which is a common compliance issue in pharmaceutical manufacturing.
How to Synthesize Linked Disulfide Propyl Porphyrin Efficiently
The synthesis of this advanced catalyst is designed for practical scalability, utilizing readily available starting materials and standard organic transformations. The process begins with the reduction of deuteroporphyrin dimethyl ester, followed by bromination to activate the side chains for nucleophilic substitution. The key step involves the formation of the disulfide bridge using thiourea, a cost-effective reagent that ensures high purity of the intermediate. Finally, metallation with transition metal salts completes the catalyst preparation. This straightforward pathway avoids the need for exotic reagents or cryogenic conditions, making it highly attractive for commercial scale-up of complex specialty chemicals. For detailed standardized synthesis protocols and safety parameters, please refer to the guide below.
- Reduce deuteroporphyrin dimethyl ester using sodium borohydride and lithium chloride in tetrahydrofuran to obtain hydroxypropyl porphyrin.
- Convert the hydroxyl group to a bromide using phosphorus tribromide in dichloromethane to form bromopropyl porphyrin.
- React bromopropyl porphyrin with thiourea in ethanol followed by alkaline hydrolysis to form the linked disulfide propyl porphyrin ligand.
- Coordinate the porphyrin ligand with a transition metal salt (e.g., Cobalt, Manganese) in chloroform under reflux to yield the final metal complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst technology translates into tangible strategic benefits beyond mere technical performance. The shift from stoichiometric oxidants to catalytic aerobic oxidation fundamentally alters the cost structure of manufacturing oxidation-sensitive intermediates. By eliminating the consumption of expensive and hazardous oxidizing agents, manufacturers can achieve significant cost reduction in fine chemical manufacturing. Additionally, the ability to use air or oxygen as the terminal oxidant removes the logistical burden of sourcing, storing, and disposing of large quantities of chemical oxidants, thereby streamlining the supply chain and reducing inventory risks associated with hazardous materials.
- Cost Reduction in Manufacturing: The integrated design of the catalyst eliminates the need for purchasing and handling separate co-catalysts, which simplifies the bill of materials. Since the catalyst can be recovered and recycled after the reaction, the effective cost per kilogram of product is drastically lowered over time. The high selectivity reduces the loss of valuable starting materials to byproducts, maximizing raw material utilization and improving overall process economics without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthesis relies on commodity chemicals like sodium borohydride, thiourea, and common metal salts, which are widely available from multiple global suppliers. This diversification of raw material sources mitigates the risk of supply disruptions that often plague specialized reagent markets. Furthermore, the robustness of the catalyst allows for longer shelf life and easier transportation, ensuring consistent availability for continuous production lines and reducing the frequency of emergency procurement actions.
- Scalability and Environmental Compliance: The process generates minimal inorganic waste compared to traditional oxidation methods, significantly lowering the costs associated with wastewater treatment and hazardous waste disposal. This aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing process against regulatory changes. The mild reaction conditions also reduce energy consumption for heating and pressure maintenance, contributing to a lower carbon footprint and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of linked disulfide porphyrin catalysts in industrial settings. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these aspects is essential for technical teams evaluating the transition from legacy oxidation processes to this advanced biomimetic technology.
Q: How does the disulfide bridge improve catalytic performance compared to traditional metalloporphyrins?
A: The integrated disulfide bridge acts as an internal co-reducing group, mimicking the dual-active-center mechanism of cytochrome P450. This eliminates the need for external co-catalysts, simplifying product separation and enhancing selectivity in oxidation reactions.
Q: What are the primary industrial applications of this catalyst technology?
A: This catalyst is highly effective for the aerobic oxidation of cyclohexane to KA oil, benzyl alcohol to benzaldehyde, dehydroaromatization of dihydropyridines, and the selective oxidation of sulfides to sulfoxides, making it vital for pharmaceutical and fine chemical synthesis.
Q: Is the synthesis of this catalyst scalable for commercial production?
A: Yes, the patent outlines a robust four-step synthesis using common reagents like sodium borohydride and thiourea. The process avoids extreme conditions and utilizes standard workup procedures like extraction and recrystallization, ensuring high feasibility for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linked Disulfide Propyl Porphyrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in driving innovation within the pharmaceutical and fine chemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity linked disulfide propyl porphyrin catalysts that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to optimize your oxidation processes and achieve your sustainability targets. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in catalyst supply can enhance your competitive advantage in the global market.
