Advanced Catalytic Hydrogenation for Scalable Production of Pteridinone Intermediates
Advanced Catalytic Hydrogenation for Scalable Production of Pteridinone Intermediates
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as core structures for anti-proliferative agents. Patent CN101065381A introduces a groundbreaking modification in the preparation of fused piperazin-2-one derivatives, specifically targeting the efficient synthesis of 7,8-dihydro-5H-pteridin-6-one scaffolds. These structures are pivotal in the development of next-generation therapeutics, yet their historical synthesis has been plagued by impurity profiles that complicate purification. The disclosed technology leverages a sophisticated catalytic hydrogenation strategy that integrates noble metal catalysts with specific transition metal additives to achieve unprecedented selectivity. By addressing the limitations of traditional nitro-reduction pathways, this innovation offers a streamlined approach that enhances both chemical purity and process economics. For R&D teams and procurement specialists alike, understanding the nuances of this catalytic system is essential for securing a reliable supply chain of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7,8-dihydro-5H-pteridine-6-ketone derivatives has relied heavily on the reduction of corresponding nitro-compounds, a pathway fraught with significant technical challenges. Prior art, such as that described in WO03/020722, indicates that standard reduction techniques often result in the formation of deeply colored mixture products. These complex impurity profiles necessitate rigorous and labor-intensive aftertreatment and purification steps, which drastically increase manufacturing costs and extend lead times. Furthermore, earlier attempts described in documents like WO 96/36597 utilized noble metal catalysts in the presence of vanadium compounds but frequently failed to prevent the formation of unhindered amines or lacked the necessary control to ensure exclusive lactam formation. The inability to cleanly convert the nitro precursor directly to the desired cyclic urea structure without generating substantial byproducts has long been a bottleneck in the commercial production of these critical API intermediates.
The Novel Approach
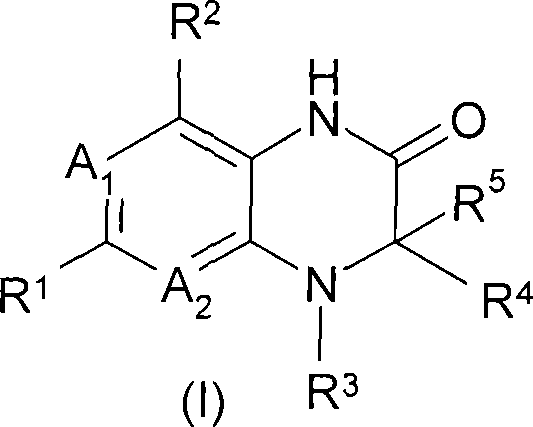
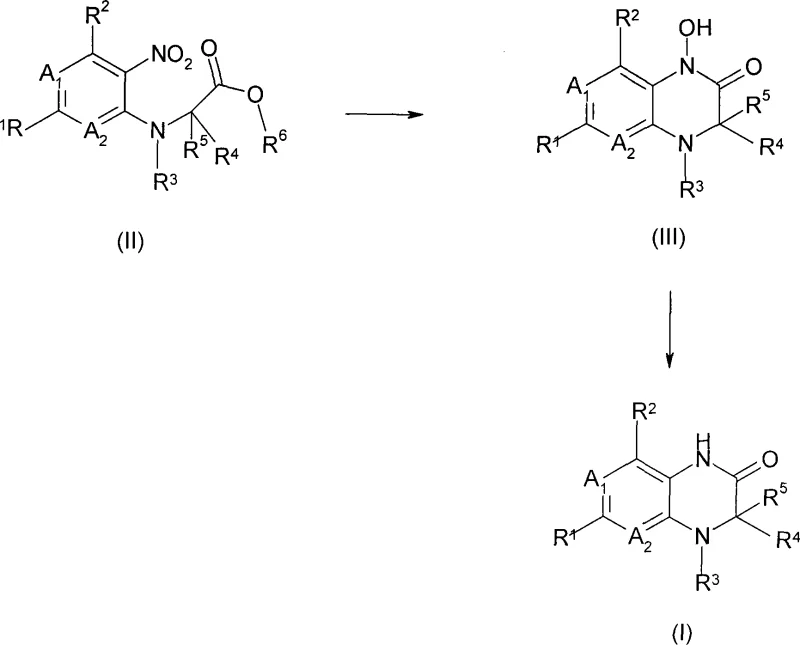
The present invention circumvents these historical hurdles by introducing a modified hydrogenation protocol that synergistically combines a hydrogenation catalyst with specific copper, iron, or vanadium compounds. This dual-catalyst system allows for the direct conversion of the nitro-precursor (Formula II) into the target fused piperazin-2-one (Formula I) with remarkable efficiency. The process can be executed either as a simultaneous one-pot reaction or as a sequential two-step procedure involving the isolation of a hydroxylamine intermediate (Formula III). By fine-tuning the reaction parameters, including the choice of solvent and the specific oxidation state of the metal additive, the method effectively suppresses the formation of dark-colored impurities. This results in a product that requires minimal purification, often achievable through simple crystallization, thereby representing a substantial leap forward in process chemistry for this class of compounds.
Mechanistic Insights into Vanadium-Promoted Catalytic Hydrogenation
The core of this technological advancement lies in the mechanistic interplay between the noble metal surface and the transition metal additive during the hydrogenation cycle. The reaction initiates with the adsorption of the nitro-group onto the catalyst surface, typically Platinum, Palladium, or Raney Nickel, where it undergoes stepwise reduction. The critical innovation is the presence of additives such as vanadium(IV) acetylacetonate, copper halides, or iron salts, which appear to modulate the reduction potential and prevent over-reduction to the open-chain amine. Instead, the system favors the intramolecular cyclization required to form the six-membered piperazin-2-one ring. The patent specifies that vanadium compounds in oxidation states ranging from 0 to V are effective, with VO(acac)2 being particularly preferred, suggesting a specific coordination environment that stabilizes the reactive intermediates. This precise control over the reaction trajectory ensures that the nitrogen atoms are correctly positioned for lactamization rather than forming unwanted linear byproducts.
Impurity control is further enhanced by the selection of appropriate solvents and reaction conditions that minimize side reactions. The process tolerates a wide range of polar and non-polar solvents, including tetrahydrofuran, alcohols, and esters, allowing for flexibility in process optimization. The temperature range of 0°C to 150°C and hydrogen pressures from 1 to 100 bar provide a broad operational window, enabling chemists to balance reaction rate with selectivity. By avoiding the harsh conditions or non-selective catalysts of the past, this method significantly reduces the generation of tarry residues and colored impurities. Consequently, the final product exhibits a much cleaner impurity profile, which is a critical quality attribute for pharmaceutical intermediates intended for human use, reducing the burden on analytical quality control and ensuring consistent batch-to-batch reproducibility.
How to Synthesize Fused Piperazin-2-one Derivatives Efficiently
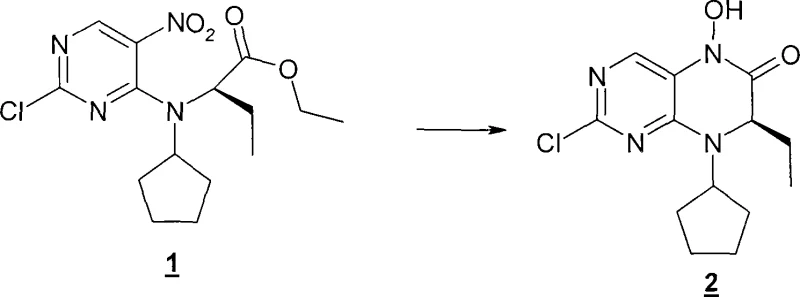
Implementing this synthesis route requires careful attention to the stoichiometry of the catalyst system and the management of hydrogen pressure. The general procedure involves dissolving the nitro-starting material in a solvent like tetrahydrofuran or ethanol, followed by the addition of the noble metal catalyst, typically 5% Pt/C. Crucially, a catalytic amount of the transition metal additive, such as vanadium(IV) acetylacetonate, is introduced either at the start or after the formation of the hydroxylamine intermediate. The reaction mixture is then subjected to hydrogen atmosphere under controlled pressure and temperature until hydrogen uptake ceases, indicating complete conversion. Following the reaction, the catalyst is removed by filtration, and the product is isolated through concentration and crystallization, often yielding high-purity solids suitable for subsequent coupling reactions in API synthesis.
- Dissolve the nitro-precursor compound (Formula II) in a suitable polar solvent such as tetrahydrofuran, ethanol, or ethyl acetate within a hydrogenation vessel.
- Add a hydrogenation catalyst, preferably Platinum on Carbon (Pt/C), along with a catalytic amount of a transition metal additive such as vanadium(IV) acetylacetonate.
- Pressurize the reactor with hydrogen gas (1 to 100 bar) and maintain the temperature between 20°C and 100°C until hydrogen uptake ceases, followed by filtration and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages regarding cost stability and supply reliability. The elimination of complex purification steps, such as extensive chromatography or multiple recrystallizations required by older methods, directly correlates to reduced manufacturing cycles and lower operational expenditures. By simplifying the downstream processing, manufacturers can achieve higher throughput rates without compromising on the stringent purity specifications demanded by the pharmaceutical sector. Furthermore, the use of commercially available and relatively inexpensive additives like vanadium acetylacetonate alongside standard noble metal catalysts ensures that the raw material costs remain predictable and manageable, shielding the supply chain from volatility associated with exotic reagents.
- Cost Reduction in Manufacturing: The streamlined nature of this hydrogenation process significantly lowers the cost of goods sold by minimizing solvent consumption and waste generation. Traditional methods that produce dark, tarry mixtures require expensive disposal protocols and additional solvent volumes for washing and purification, whereas this novel route yields cleaner crude products. The ability to isolate the final product through simple crystallization from common solvents like acetone or water mixtures further reduces utility costs and waste treatment burdens. Additionally, the high selectivity of the catalyst system means less starting material is lost to byproducts, improving the overall atom economy and maximizing the yield per batch, which is a critical factor in cost-sensitive API intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain continuity by reducing the risk of batch failures due to impurity spikes. Because the process is less sensitive to minor variations in reaction conditions compared to non-catalyzed reductions, it offers greater consistency in production scheduling. The reliance on standard hydrogenation equipment and widely available solvents means that production can be easily transferred between different manufacturing sites without significant capital investment in specialized infrastructure. This flexibility ensures that procurement teams can secure multiple sources of supply or scale up production rapidly in response to market demand, mitigating the risks of shortages that often plague complex heterocyclic intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of stoichiometric reducing agents like metal acids, which generate large quantities of salt waste, in favor of catalytic hydrogenation significantly lowers the environmental footprint. The patent examples demonstrate successful scaling from gram-scale laboratory trials to multi-hundred gram batches, proving the feasibility of commercial scale-up. This scalability, combined with the use of recyclable heterogeneous catalysts, supports sustainable manufacturing practices and simplifies compliance with increasingly stringent environmental regulations governing pharmaceutical production facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the versatility of the catalyst system and the practical benefits observed during the development of this synthetic methodology.
Q: What is the primary advantage of this new hydrogenation process over conventional methods?
A: Unlike prior art methods which often produce deeply colored mixtures requiring laborious purification, this novel process utilizing vanadium or iron additives yields high-purity products with significantly simplified downstream processing.
Q: Which catalysts and additives are essential for this transformation?
A: The process relies on noble metal catalysts such as Platinum, Palladium, or Raney Nickel, critically enhanced by the presence of copper, iron, or vanadium compounds, with vanadium(IV) acetylacetonate being particularly preferred for optimal selectivity.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the patent demonstrates successful scalability from gram-scale laboratory experiments to multi-hundred gram batches, utilizing standard hydrogenation equipment and commercially available solvents like THF and ethanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Piperazin-2-one Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the drug development pipeline and are uniquely positioned to support your projects with this advanced technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped to handle the complex analysis required for heterocyclic compounds, guaranteeing that every batch meets the exacting standards of the global pharmaceutical industry. Our commitment to technical excellence allows us to optimize processes like the vanadium-promoted hydrogenation described here, delivering cost-effective solutions without compromising on quality or safety.
We invite you to collaborate with us to leverage these process innovations for your specific therapeutic programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthetic route can improve your project economics. Please contact us to request specific COA data for our pteridinone intermediates and to discuss route feasibility assessments for your custom synthesis needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to driving innovation and efficiency in the production of complex pharmaceutical building blocks.
