Scalable Manufacturing Of Levocetirizine Via Optimized Chiral Resolution And Alkylation
Scalable Manufacturing Of Levocetirizine Via Optimized Chiral Resolution And Alkylation
The global demand for third-generation antihistamines continues to drive innovation in process chemistry, specifically for high-value chiral intermediates like Levocetirizine Dihydrochloride. Patent CN101100462A presents a robust and economically viable synthetic pathway that addresses the historical bottlenecks of cost and raw material availability associated with this blockbuster drug. Unlike previous methodologies that relied on complex asymmetric syntheses or prohibitively expensive chiral building blocks, this disclosed method leverages a classical yet highly effective resolution strategy combined with efficient N-alkylation sequences. For procurement directors and supply chain managers, this represents a critical shift towards a more sustainable and cost-controllable manufacturing model. The technology transforms simple, commodity-grade starting materials into high-purity active pharmaceutical ingredients through a series of well-defined chemical transformations that are inherently scalable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Levocetirizine has been hampered by the limitations outlined in early intellectual property, such as European patent EP 0617028. These conventional routes often necessitated the use of specialized chiral intermediates that were not only difficult to source commercially but also commanded premium pricing due to limited supplier competition. Furthermore, alternative asymmetric synthesis routes reported in academic literature, such as those by D.A. Pflum, frequently suffered from poor stereoselectivity and required severe reaction conditions that posed significant safety and engineering challenges at scale. The reliance on exotic reagents and the generation of complex impurity profiles in these older methods created substantial downstream purification burdens, ultimately inflating the cost of goods sold and introducing volatility into the supply chain for pharmaceutical manufacturers.
The Novel Approach
The methodology described in CN101100462A circumvents these issues by adopting a 'build-and-resolve' strategy rather than a 'buy-chiral' approach. By initiating the synthesis with achiral p-chlorobenzophenone and formamide, the process utilizes ubiquitous raw materials that are available from multiple global suppliers, thereby mitigating supply risk. The core innovation lies in the efficient resolution of the intermediate benzhydrylamine using L-o-chloromandelic acid, a resolving agent that offers excellent selectivity and ease of recovery. Subsequent steps involve the construction of the piperazine ring and functionalization using standard alkylation agents like chloroethanol and sodium chloroacetate. This linear progression minimizes the number of isolation steps and avoids the use of transition metal catalysts, resulting in a cleaner process that is ideally suited for GMP manufacturing environments.
Mechanistic Insights into Chiral Resolution and Sequential Alkylation
The heart of this synthetic route is the stereochemical control achieved during the resolution phase. The reaction begins with the formation of a racemic amine via a Leuckart-Wallach type reduction using formamide, followed by hydrolysis. The resulting DL-p-chlorobenzhydrylamine is then subjected to diastereomeric salt formation with L-o-chloromandelic acid in a methanol-water solvent system. The differential solubility of the diastereomeric salts allows for the selective crystallization of the desired L-enantiomer salt. Upon basification and extraction, the free L-amine is obtained with high optical purity. This step is critical, as it sets the stereochemistry for the entire molecule, ensuring that the final product meets the stringent enantiomeric excess requirements necessary for biological activity and regulatory approval without the need for costly chiral chromatography.
Following the establishment of chirality, the synthesis proceeds through a sophisticated alkylation sequence to construct the piperazine scaffold and side chains. As illustrated in the reaction scheme below, the chiral amine reacts with a bis-functionalized sulfonamide reagent to close the piperazine ring while simultaneously installing a protecting group. Subsequent deprotection reveals the secondary amine, which is then sequentially alkylated. First, reaction with chloroethanol introduces the hydroxyethyl moiety, and finally, reaction with sodium chloroacetate under basic conditions installs the acetic acid side chain. This modular approach allows for precise control over regioselectivity and minimizes the formation of over-alkylated byproducts.
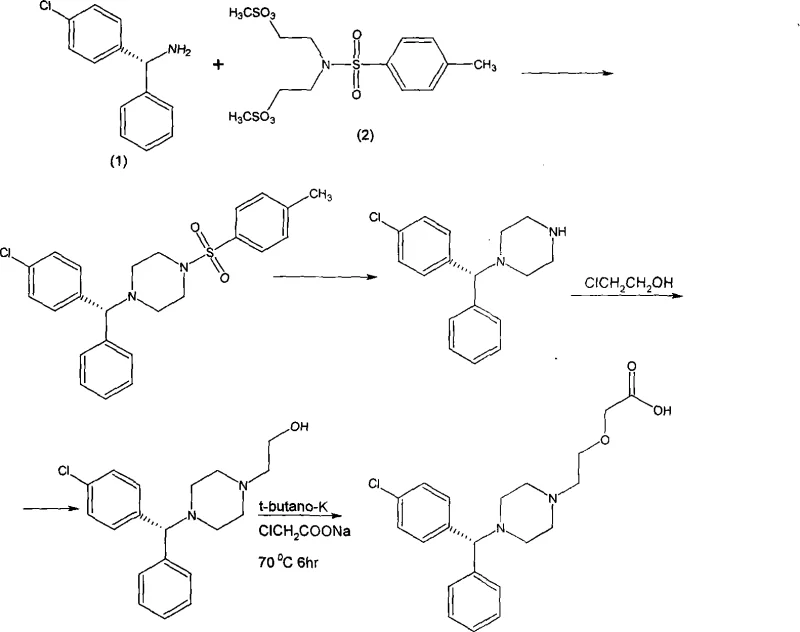
The final transformation involves the nucleophilic substitution of the piperazine nitrogen on the chloroacetate species, facilitated by a strong base such as potassium tert-butoxide. The choice of base and solvent system, typically tert-butanol or DMF, is optimized to maximize conversion while minimizing elimination side reactions. The crude product is then converted to the dihydrochloride salt through treatment with hydrochloric acid and recrystallization from acetone-water mixtures. This final salt formation step not only stabilizes the molecule for storage but also serves as a final purification stage, removing residual organic impurities and ensuring the physical properties, such as flowability and particle size, are suitable for tablet formulation.
How to Synthesize Levocetirizine Dihydrochloride Efficiently
Executing this synthesis requires careful attention to temperature control and stoichiometry, particularly during the resolution and final alkylation steps. The process is designed to be operationally simple, relying on standard reactor configurations available in most fine chemical facilities. The initial condensation and hydrolysis steps require elevated temperatures, while the resolution is performed under mild conditions to preserve crystal integrity.
- Synthesize DL-p-chlorobenzhydrylamine from p-chlorobenzophenone and formamide, followed by resolution using L-o-chloromandelic acid.
- React the resolved chiral amine with a bis-sulfonamide reagent to form the protected piperazine ring, followed by deprotection.
- Perform sequential alkylation with chloroethanol and sodium chloroacetate under basic conditions to yield the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101100462A offers distinct strategic advantages centered around cost stability and supply security. The primary driver of value is the drastic simplification of the raw material portfolio. By eliminating the dependency on proprietary chiral intermediates that are often controlled by a single supplier, manufacturers can leverage competitive bidding for commodity chemicals like p-chlorobenzophenone and formamide. This diversification of the supply base significantly reduces the risk of production stoppages due to raw material shortages and provides substantial leverage in price negotiations, leading to a more resilient and cost-effective supply chain structure.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from the elimination of expensive chiral catalysts and the reduction of complex purification steps. Traditional asymmetric synthesis often requires precious metal ligands or enzymes that add significant cost per kilogram; in contrast, this resolution-based approach uses inexpensive organic acids that can potentially be recovered and recycled. Furthermore, the avoidance of transition metals simplifies the workup procedure, removing the need for expensive scavenging resins or extensive washing protocols to meet heavy metal specifications, which directly translates to lower operational expenditures and higher overall process yields.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures that the production of Levocetirizine is not bottlenecked by niche supply markets. Materials such as sodium chloroacetate, chloroethanol, and toluene are produced on a massive global scale for various industries, guaranteeing consistent availability and stable pricing even during market fluctuations. This reliability allows for more accurate long-term capacity planning and inventory management, enabling pharmaceutical companies to maintain continuous production schedules without the fear of sudden raw material price spikes or allocation limits that often plague specialized fine chemical sectors.
- Scalability and Environmental Compliance: From an environmental and engineering perspective, this process is highly amenable to scale-up. The reaction conditions utilize standard solvents and moderate temperatures that do not require specialized high-pressure or cryogenic equipment, reducing capital expenditure for new production lines. Additionally, the process generates fewer hazardous waste streams compared to routes involving heavy metals or exotic reagents. The simplicity of the isolation steps, primarily crystallization and extraction, facilitates easier waste treatment and solvent recovery, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations without compromising output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. Understanding these details is crucial for R&D teams evaluating technology transfer and for quality assurance teams establishing control strategies.
Q: What is the primary advantage of the resolution method in CN101100462A?
A: The patent utilizes L-o-chloromandelic acid for resolution, which avoids the need for expensive chiral catalysts or hard-to-source chiral starting materials found in earlier European patents, significantly lowering raw material costs.
Q: How does this process ensure high optical purity?
A: Through careful crystallization of the diastereomeric salt formed between the racemic amine and the chiral resolving agent, the process achieves an optical purity (ee) of greater than 99%, meeting strict pharmacopeial standards.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the process relies on commodity chemicals like p-chlorobenzophenone and formamide, and utilizes standard unit operations such as reflux, crystallization, and extraction, making it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levocetirizine Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust process chemistry in the competitive landscape of antihistamine manufacturing. Our technical team has extensively analyzed the pathway detailed in CN101100462A and possesses the expertise to optimize every step, from the initial chiral resolution to the final salt formation. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess and residual solvent levels, guaranteeing that every batch meets international pharmacopeial standards.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods.
